1. Introduction
Learning is common in animals from small invertebrates to mammals and plays an important role in term of adaptations [1]. Associative learning that consists in the association of two different elements [2] is particularly relevant for the study of animal cognition [3].
Cognition research in marine mammals is well documented, particularly for cetaceans, with studies predominantly focused on bottlenose dolphins and pinnipeds, principally Californian sea lions [4, 5]. During recent years animal cognition research has often been combined with animal welfare studies as suggested by several authors [4, 6, 7]. Environmental enrichment, associated with the well-being concept in animals, first consisted of improvements to the captive animal environment [8]. This relatively new concept was well studied in aquatic mammals, particularly dolphins [9]. Several studies suggest that environmental enrichment for animal welfare allows the animal to express behaviours and use abilities, such as cognition [7]. The stimulation of cognitive capacities in animals, through enrichment in captivity, is a technique that can increase animal welfare and decrease, or eliminate, stereotype behaviours in a large number of animals [10]. Sensory enrichment by environmental or occupational enrichment in animals may reduce or eliminate stereotypical behaviours, and increase welfare that can be measured through behavioural pattern enhancement [11]. Regarding cognitive capacities, numerous studies have demonstrated that animals, invertebrates or vertebrates possess learning capacities that are used when they have to escape from predators or danger [12], or when foraging to increase their chance of success [13, 14]. These capacities are well studied in aquatic mammals, such as dolphins or killer whales [5]. Learning is generally related to the ability of animals to discriminate between cues, particularly visual ones, as demonstrated in insects [15], birds [16] or mammals [17, 18]. In aquatic mammals, discrimination between cues is well studied in a social context, e.g. f Tursiops truncatus Montagu 1821 (Cetaceae, Delphinidae) that uses whistle contours to discriminate between conspecifics [19]. Despite dolphins being considered as acoustic specialists, visual cues may have also a prominent role and have been used in several studies on dolphin cognition. For example, Kilian et al. [20] studied the ability of one female dolphin to discriminate visual stimuli differing in numerosity. Another study shows that a female beluga was also able to visually discriminate between quantities of food [21].
Behavioural studies on Antillean manatee (Trichechus manatus manatus, Linnaeus 1785; Sirenia, Trichechidae) are uncommon, despite their importance for conservation and their presence in several zoos and parks. Hartman [22] carried out the first large-scale study of manatee behaviour and stated that manatees do not need complex behavioural patterns because of the lack of predators, the abundance of food and the stable water temperatures where they live. Koelsch [23] suggested a higher level of sociability than described by Hartman [22] in Bahía de Sarasota (Florida), while Harper and Schulte [24] observed that social interactions occurred only between females. The most recent study by Hénaut et al. [25] reported that in captivity, social interactions also occurred between males and females and particularly between a calf and the various members of the manatee group, irrespective of sex and age. Only one recent work has focused on manatee welfare, describing their stereotypical behaviours [26]. As observed in several marine mammals, the “circle swimming behaviour” seems to be the typical behavioural characteristic of manatee’s unease in captivity [26]. As yet, no studies have adopted a behavioural approach to manatee visual perception and cognitive capacities, despite the importance that this type of study may have on their management and in particular on the welfare of captive animals and their successful reintroduction to the wild.
Vision in sirenians is considered limited when compared with other aquatic mammals [27]. However, Hartman [21] reported that vision may be important for manatees and according to Reynolds and Oddel [28] manatee’s eyes are able to see in broad daylight and half-light. Reep and Bonde [27] described that manatees have dichromatic vision as observed in pinnipeds, with the detection of blues and greens. Manatees have tactile hairs that provide good discrimination (e.g. to explore vegetation) and are also able to use their facial bristles to detect all directions of their surroundings when swimming in a circular pattern [29]. Although manatee brain size is small compared with body size, the cerebral cortex is similar to those of mammals with large brains, which may indicate considerable cognitive abilities [30].
Few studies have focused on manatee welfare in captivity. These mammals are generally captive in a reduced space, especially when compared with their natural environment, and most of the time they may be observed swimming around the aquarium [26]. This stereotypical behaviour in manatees seems associated with a reduced behavioural repertoire (Hénaut, personal observation). This reduced repertoire may also be the consequence of poor captive manatee management, with a lack of stimulating activities and few social interactions with conspecifics or humans, even when taking into account veterinary care [31], or when fed by tourists [25]. Furthermore, the low number of studies related to manatee welfare and their cognitive capacities, ensures that new information on manatee behaviour, particularly if focused on cognition and well-being, is not only fascinating in terms of a unique scientific contribution, but also valuable for their management in captivity and for achieving successful reintroductions.
Behavioural studies of manatees generally occur with captive individuals in zoological or tourist centres. Associated captivity constraints do not provide appropriate conditions for behavioural observations, particularly for cognitive studies, because the predominant form of management limits the possibilities of using devices or introducing objects into their pool and the public often disturb individual animals. Moreover, manatees in captivity generally live in groups with interaction between individual animals [25]. It is also difficult to gain access to a large number of manatees for individual observation without the possibility of social perturbation, particularly on social learning, an effect we would expect for many animals species [32].
Fortunately, we were able to study the behaviour of one single manatee, called Daniel, reared in semi-captivity. The aim of our work is to study learning abilities related to food association with visual cues and the memory capacity of the manatee Daniel.
2. Material and methods
2.1. Animal and housing
Observations were conducted in the “Centro de Atención y Rehabilitación de Mamíferos Acuáticos” (CARMA) (Centre for the Attention and Rehabilitation of Aquatic Mammals) located in a lagoon (Laguna Guerrero,) near to Chetumal, in the state of Quintana Roo, south-eastern Mexico. In this centre, a manatee called Daniel, a 7-year-old male, lives in “semi-free” conditions. Daniel, an orphan calf rescued when recently born, lives in an enclosed area where food is provided, and can regularly go out into the lagoon at will, generally during the afternoon. Virtually every day, Daniel returns to the area before nightfall. The area is rectangular (17.30 m × 15 m) surrounded by an enclosure made of wood and wire mesh. The depth of the water was around three meters. The water is clear enough to allow good observation of Daniel.
2.2. Material
We used four geometrical objects (square, equilateral triangle, disc and a hexagon) made of white acrylic (Figure 2). The square and triangle had 50 cm sides while the disc and hexagon were 50 cm in diameter; therefore, all the objects were similarly sized. Each shape had a 2.5 cm white PVC tube along its bottom edge so it could be submerged vertically. The shape was hung from the enclosure with a rope so that the top part was just 1 cm under the surface of the water (see Supplementary Videos, 1, 2, 3 and 4).
When observations were made outside the enclosed area, we made a small 1 cm diameter hole in the centre of each shape so that the food could be attached using a fishing line. The shape was hung from an air tube fixed onto a wooden baton hammered into the bottom of the lagoon.
The Cos lettuce (Lactuca sativa L. var. longifolia) was used as food as previous observations suggest it was his favourite.
The shapes were always placed at least 2.5 m apart and their positions changed after each observation, using the entire enclosed area. The observer was always the same person and changed their position so that they were always at a distance from the shapes to prevent disturbing Daniel. In effect, Daniel is accustomed to the presence of an observer and did not present any particular behaviours oriented towards the observer during our experiments after at least one day’s presence. We recorded videos of Daniel to clearly illustrate behaviours.
2.3. Experimental procedure and associative learning task
The six steps of the experimental procedure.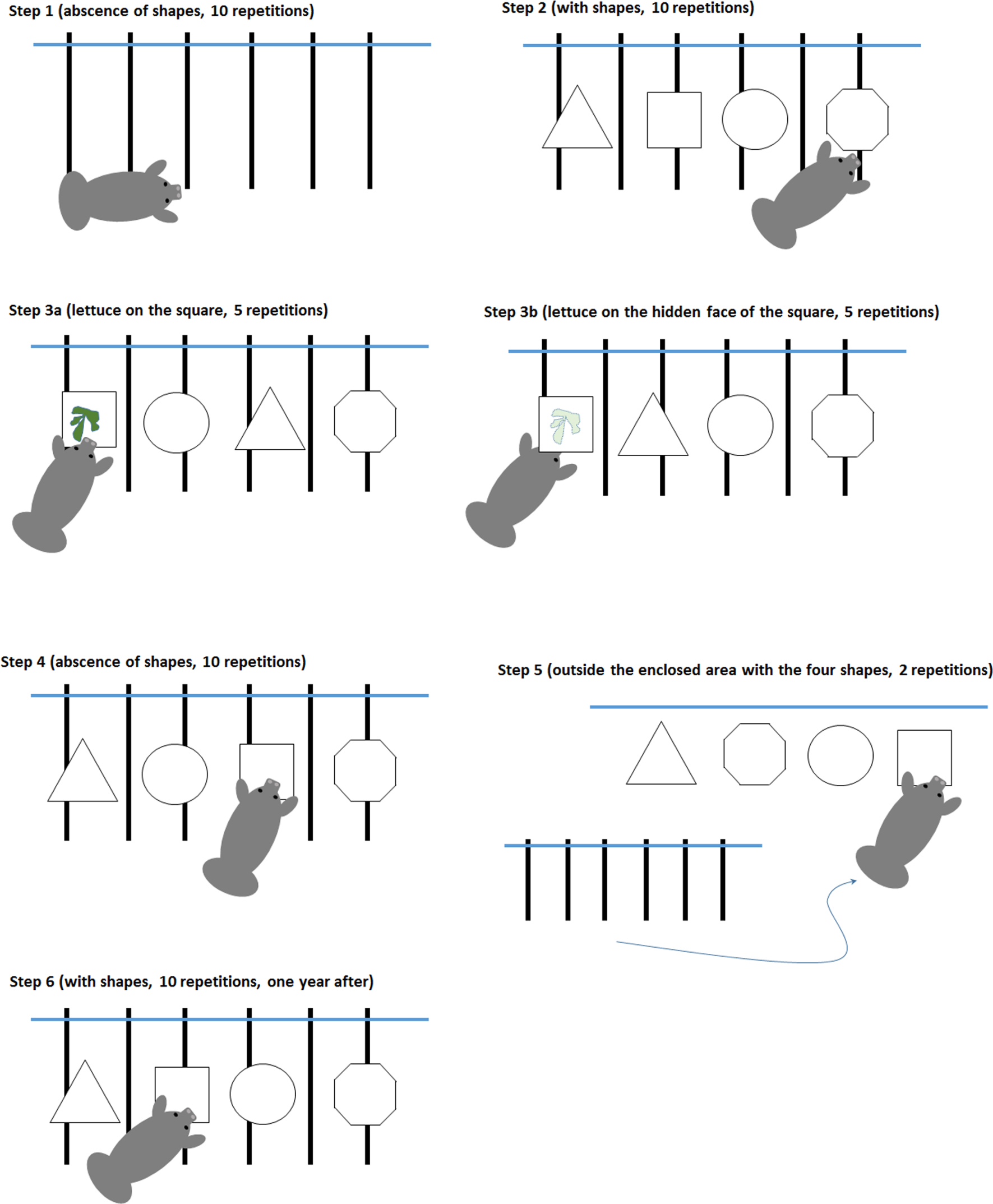
Mean and standard error of frequency for the number of paths covered in front of each shape by the manatee, before (step 2, in black) and after (step 4, in grey) training. Wilcoxon test, *: P < 0.05, NS: not significant.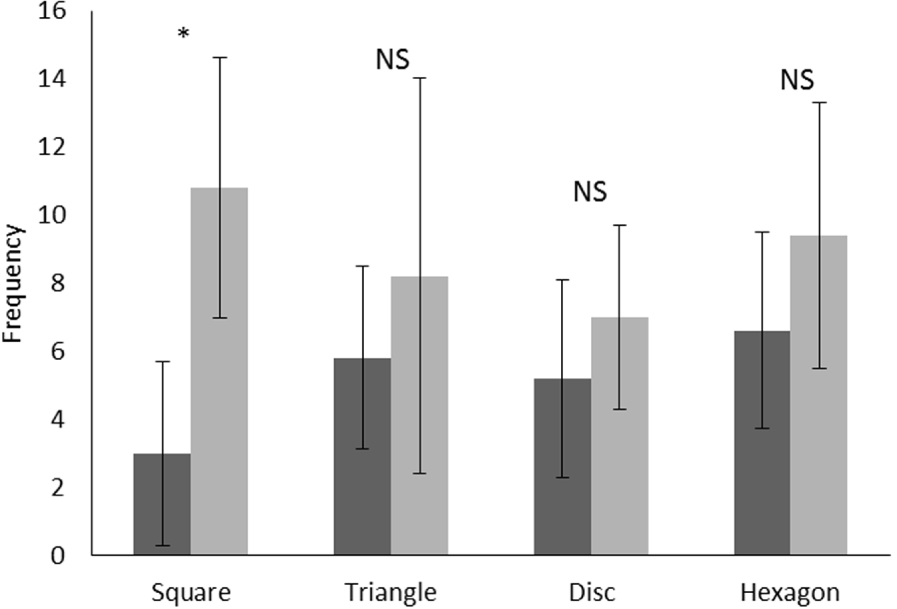
The observations took place over six steps (Figure 1).
Step 1
we observed the behaviour of Daniel in the absence of shapes, 10 repetitions were carried out.
Step 2we observed Daniel’s behaviour in the presence of the four geometric, shapes, submerged as previously described, and without food (10 repetitions).
Step 3termed the “training step” which consists in selecting the shape least preferred by Daniel during step 2 (the square) and then attaching food onto it.
Step 3a
Five repetitions were carried out with the lettuce on the open face of the shape facing the centre of the area (visible to Daniel).
Step 3b5 repetitions with the food on the hidden face of the figure.
Step 4we observed the behaviour of Daniel, once again with the four shapes, as in step 2, but without food (10 repetitions).
Step 5we present the four shapes outside the enclosed area and open the enclosure so that Daniel could leave (2 repetitions).
Step 6we observed Daniel’s behaviour in front of the four shapes without food in the enclosed area, 3 months (1 observation) and 12 months (1 observation) after the “training step” and without reinforcement or any access to the shapes during this lapse of time.
All observations took place from 8:00 am to 2:00 pm as preliminary observations indicated that this was the period of maximum activity for Daniel. For each repetition, we observed the animal during 30 min with a maximum of five repetitions per day. We recorded all behavioural events during the observation time. We divided the area surrounded by the enclosure into virtual quadrants (1 m2 each) where we noted on schematic paper Daniel’s path (or “swim by” when Daniel crossed a quadrant), behaviour and time spent in each quadrant.
2.4. Data and analysis
During step 1, without any shapes, we noted the number of paths made by Daniel swimming and crossing a quadrant as well as the time spent in each quadrant within the enclosure where the shapes would be submerged during the subsequent step. We considered that for all steps, with the exception of the first (without shapes), a visit by Daniel was recorded when it crossed a quadrant with its head at least 50 cm from the shape present in the quadrant. When Daniel remained in a quadrant without moving, its head clearly oriented towards a shape as if starring at it, we recorded that Daniel “looked at the figure”. During step 1, we recorded the number of paths in quadrants without shapes, where they would be subsequently installed in the additional steps. We also recorded the time spent by Daniel crossing these quadrants. The frequency of visits and the “look at the shape” behaviour were recorded for step 2 (shapes without food before training), step 4 (shapes without food after training) and step 5 (shapes without food, after training but outside the enclosed area). All other behaviours, including unexpected ones, were described in the results. During the “training stage” (step 3a and 3b) we recorded the time required to “reach the figure”, “begin eating the lettuce” once the figure was reached, and “finish consuming the lettuce” until the lettuce had been completely eaten. When shapes were presented to Daniel 3 and 12 months after the training step (step 6), only the interaction of Daniel with the shapes and the behaviours associated with those interactions was recorded.
To establish whether the associative learning task was working, we compared using a Friedman test, the number of paths, visits, “look at the shape” behaviours and the time spent in each quadrant for the four shapes during step 2 (before training) and step 4 (after training). We compared the time spent by Daniel in the quadrants with a shape (step 2) and the corresponding quadrant without the shape (step 1) using a Wilcoxon matched pairs test. The same test was used to compare the number of visits for both of these steps. The time necessary to “reach the figure” with the lettuce, to “begin to eat” and “finish consuming the lettuce” for the lettuce in front of the figure and the lettuce on the hidden face of the figure (step 3a and 3b) were compared using a Wilcoxon matched pairs test. The number of paths, visits, “look at the shape ” behaviours and the time spent in front of each shape for step 2 (before training) were compared with step 4 (after training) for each shape using a Wilcoxon matched pairs test. All tests were performed using Statistica 7.0.
We also recorded and described short duration events behaviours for the four first steps. It was impossible to observe the totality of behaviours during the fifth step because the distance from Daniel made observation difficult. The occurrence of known behaviours and the appearance of new ones was compared for steps 1 to 4. These descriptive results will help us understand if and how the presence of the shapes and training may have affected Daniel activities. For step 6, (observation after 3 months and 12 months) we only described the selection of certain shapes and specific behaviours. Video clips of exemplary trials are included in the supplementary material.
3. Results
3.1. Daniel’s behaviour without and with the shapes (steps 1 and 2)
Daniel’s behaviour during step 1 consisted essentially of movement behaviours, (Table 1) mostly swimming and breathing around the enclosed area. Occasionally Daniel displayed “rotation” and “head out” behaviours. When the shapes were present in the area, the activity of Daniel radically changed with considerably more movements than during step 1, and a series of new behaviours directed at the shapes: “look at the figure”, “hug the figure” (Supplementary Video 1), “touch the figure with its face”, “touch the figure with a flipper” and “play with the figure”; however, the “head out” behaviour disappeared during step 2 (Table 1). The “touch the figure with a flipper” behaviour demonstrated a clear interest for the new objects.
Description and codes for all behaviours described in this study
| Behaviour | Code | Description | Step 1 | Step 2 | Step 3a | Step 3b | Step 4 |
|---|---|---|---|---|---|---|---|
| Rotation | Ro | Rotating movements along the vertical/horizontal axis [25] | X | X | X | X | X |
| Swim | Sw | Normal and slow swimming in the water | X | X | X | X | X |
| Swim up | Su | Normal and slow swimming with the dorsal part of the body emerging from water | X | X | X | X | X |
| Swim out | So | Swim towards the water surface with part of the body emerging and then swimming back under the water | X | X | X | X | X |
| Breathe | B | Raise the head above water and breathe | X | X | X | X | X |
| Resting | Re | Resting, floating near the surface (described by Hartman [22]) | X | X | X | X | X |
| Head out | Ho | Raise the head above water without breathing | X | X | X | X | |
| Look at the shape | Lf | Remain in a quadrant without moving, the head clearly oriented towards a shape, as if staring at it | X | X | X | X | |
| Touch the shape with face | Tf | Contact with the shape using its face (and/or vibrissae) | X | X | X | X | |
| Touch the shape with flipper | Tfl | Contact with the shape using a flipper | X | X | X | X | |
| Hug the shape | Hf | Hug the shape with the flippers | X | X | X | X | |
| Play with the shape | Pf | Quickly succession of behaviours that always occur together: hugging the shape, touching it with one flipper, nose or body | X | ||||
| Feeding on the shape | Fef | Stay close to the shape, catching the lettuce with the nose and eating it | X | X | |||
| Rest on the bottom | Rb | Stay on the bottom of the water without moving | X | ||||
| Bite the shape | Bf | Bite the edge of the shape | X | X | X | ||
| Swim close to the shape | Sf | Swim normally but visibly alongside a shape | X | X | X | ||
| Rest on the shape | Rf | Use the shape as a support for the head and stop moving | X | X | |||
| Bite the Mesh | Bm | Bite the wire mesh close to a shape | X | X | |||
| Looking behind the shape | Lbf | Push the shape with the nose and inspect it | X | X | |||
The occurrence of each behaviour for each step is indicated by a cross.
The number of paths was statistically greater (Table 2) in the quadrant with a triangle (step 2) than in the corresponding quadrant without the triangle (step 1). More paths were observed in the quadrant with the hexagon or disc (step 2) than in the same quadrants without these shapes (step 1) (Table 2); however, these differences were not significant. In the first step, only quadrants with the square showed a tendency towards fewer visits when compared with the corresponding quadrants without the square.
Mean and standard error of path numbers covered by the manatee in quadrants without a shape (step 1) and the same quadrant with a shape (step 2), for the four geometric shapes tested
| Shape | Step 1 (without shape) | Step 2 (with shape) | P |
|---|---|---|---|
| Square | 3.60 ± 1.90 | 2.80 ± 1.10 | NS |
| Triangle | 2.40 ± 0.80 | 6.20 ± 1.20 | 0.04 |
| Disc | 3.00 ± 0.90 | 3.80 ± 0.90 | NS |
| Hexagon | 3.40 ± 0.80 | 7.20 ± 1.90 | NS |
Wilcoxon test, NS = not significant.
Daniel spent significantly more time in the quadrant with the hexagon (step 2) than in the corresponding quadrant without the hexagon during step 1 (Table 3). Only in the case of the hexagon did Daniel spend significantly more time in the quadrants with the figure than without. The amount of time spent in quadrants with the triangle or the disc tended to be greater than in those without these shapes, while the time spent in the same quadrant with the square and without the square were identical. The number of paths Daniel took in front of the shapes (Figure 2) and the number of visits (Figure 3) did not present significant differences between shapes during the second step (Friedman test, d.f. = 3, F = 2.74, p = 0.43 and d.f. = 3, F = 5.90, p = 0.11 respectively).
Mean and standard error of frequency for the visits made by the manatee before (step 2, in black) and after (step 4, in grey) training. Wilcoxon Test: NS: not significant, *: P < 0.05.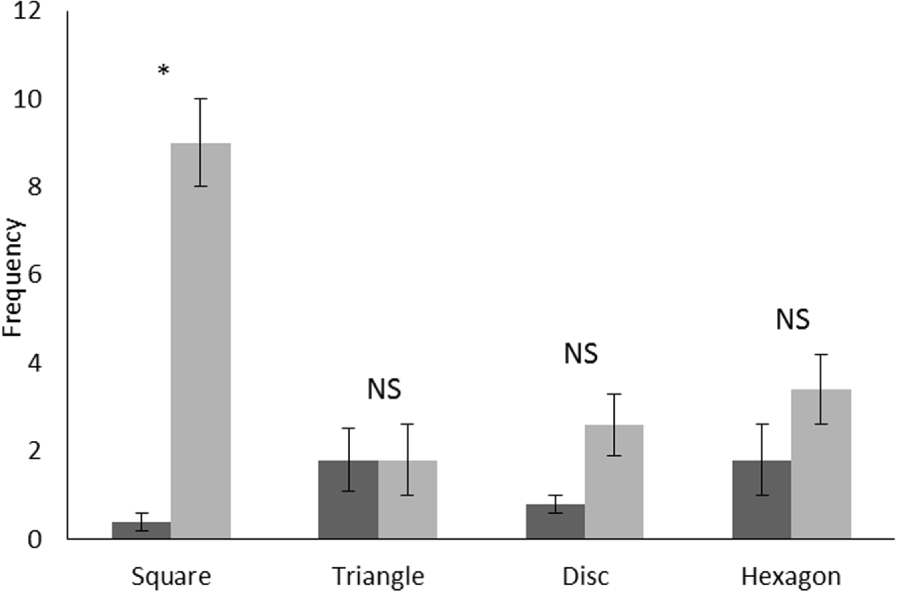
Mean time (min) spent by the manatee in quadrants without a shape (step 1) or in the corresponding quadrant with a shape (step 2), for the four geometric shapes tested
| Shape | Step 1 (without shape) | Step 2 (with shape) | P |
|---|---|---|---|
| Square | 3.60 ± 1.90 | 3.40 ± 1.40 | NS |
| Triangle | 3.20 ± 1.40 | 7.60 ± 1.40 | NS |
| Disc | 4.30 ± 0.90 | 6.00 ± 1.10 | NS |
| Hexagon | 4.00 ± 0.80 | 8.20 ± 1.70 | 0.04 |
Wilcoxon test, NS = not significant.
The “look at the figure” behaviour was only observed for the triangle, disc and hexagon during step 2 (Figure 4). When comparing the frequencies of this behaviour, between the triangle, disc and hexagon in step 2, no significant differences were revealed (Friedman test, d.f. = 2, F = 4.90, p = 0.08). Daniel never touched the square but frequently touched the other shapes, particularly the hexagon which was subject to “hugging” (Supplementary Video 1) or “playing with the figure” (Table 1). The latter behaviour was specific to this step.
Mean and standard error of frequency for the “look at the shape” behaviour performed by the manatee before (step 2, in black) and after (step 4, in grey) training. Wilcoxon Test, *: P < 0.05, NA: not available, NS: not significant.
During step 2, no statistical differences (Friedman test: d.f. = 3, F = 6.81, p = 0.78) were observed in relation to the time spent in front of each shape (mean in min ± SE; square: 0.40 ± 0.30; triangle: 0.50 ± 0.20; disk: 0.60 ± 0.30; hexagon: 1.40 ± 0.70).
We concluded that Daniel was more attracted to the hexagon and the triangle but less attracted to the square, which we therefore decided to use for training. The presence of geometrical shapes elicited a notable increase of Daniel’s activities.
3.2. Training (step 3)
During training, Daniel displayed the same behaviours as step 1 and step 2 but also new ones. Daniel displayed “feeding” behaviour (Supplementary Video 2) when he ate the lettuce (Table 1). When the lettuce was on the open face of the shape, after eating, he “rested at the bottom of the water”; this behaviour only occurred during this step (step 3a). Daniel also “bit the figure” during the step with lettuce on the open face (step 3a) and on the hidden face (step 3b). A new “swim close to the figure” behaviour appeared during training and occurred during both phases. The “swim close to the figure” behaviour was recorded when Daniel swam normally but clearly alongside the shapes (Supplementary Video 3), or specifically alongside only one shape. When the lettuce was on the hidden face of the figure Daniel displayed new behaviours such as “bite the mesh”, “look behind the shape” and “rest on the figure”. During the step 4 we observed an impressive increase of activity.
The time it took Daniel to reach the square when the lettuce was hung in front of it was 1.26 ± 0.70 min (mean ± SE). When the food was hung behind the shape the time recorded was 6.22 ± 4.00 min (mean ± SE). Despite a clear tendency to take longer to reach the shape when the food was behind, no significant difference was observed (Wilcoxon matched pairs test: Z = 1.82, p = 0.06). Furthermore, no significant difference was observed (Wilcoxon matched pairs test: Z = 0.94, p = 0.34) in the time needed to begin eating the lettuce when in front of the figure (mean ± SE: 5.00 ± 3.00 min) or behind (mean ± SE: 8.40 ± 1.20 min) (Supplementary Video 2). The time required to consume the lettuce was identical (Wilcoxon matched pairs test: Z = 1.09, p = 0.27) whatever the position of the lettuce on the shape (front: 2.10 ± 0.70 min; behind: 1.00 ± 0.30 min).
3.3. After training (step 4)
During this step Daniel displayed the same behaviours than during training, except for “feeding” (Table 1). The “rest on the shape” behaviour, when Daniel used the figure as a support for its head and stopped moving, was observed twice and was only associated with the square. The “look behind the figure” behaviour, when Daniel used its nose to push the figure from behind and inspect it, occurred with the square (5 observations) and once with the hexagon. The “bite the shape” behaviour, when Daniel bit the edge of the shape, was observed once on the hexagon. The “swim close to the shape” behaviour occurred for all the shapes (number of observations: square: 62, triangle: 39, disk: 43 and hexagon: 46) with a clear preference for the square, the least preferred figure prior to training (Supplementary Video 3). During this step, Daniel also presented important activity, comparable to that presented during training. Two behaviours appear to be more apparent during this step: “swim close to the shape” and “look at the shape”. Both of these behaviours as well as the “look behind the shape” behaviour clearly show that the manatee is actively searching for food, particularly on the square.
Pooling the results for four shapes used during the experiment, the mean of the number of paths taken in front of each shape before training (step 2) (mean ± SE: 5.10 ± 0.60) is significantly lower (Wilcoxon matched pairs test: Z = 2.70, p = 0.006, N = 20) than after training (step 4) (mean ± SE: 9.50 ± 0.80). Figure 2 clearly shows an increase in the mean number of paths for each geometric shape with the exception of the square; there are no statistically significant differences between before and after training. No differences were observed for the frequencies of the mean number of paths between the four shapes after training (Friedman test: d.f. = 3, F = 3.00, p = 0.40). Regarding this behaviour, it appears that Daniel increased the number of paths after training, although the results do not allow a distinction to be made between shapes.
Pooling the results for all shapes used during the experiment, the number of the “look at the shape” behaviours before training (mean ± SE: 0.40 ± 0.10 min) is significantly lower (Wilcoxon matched pairs test: Z = 3.40, p = 0.0007, N = 20) than after (mean ± SE: 3.60 ± 0.70 min). Figure 4 shows clearly that the “look at the shape” behaviour only appeared after training with the square and never before. In regards to the other shapes, the frequency increased significantly with training for the disc and the hexagon, but not for the triangle. Furthermore, we observed significant differences between frequencies of the “look at the shape” behaviours (Figure 4) for the four shapes after training (Friedman test: d.f. = 3, F = 12.40, p = 0.006) with approximately 7.5 behaviours per repetition for the square while the frequencies for other shapes was always less than 4.
Pooling the results for all shapes used during the experiment, the mean of the “visit” behaviour was significantly lower before training (mean ± SE: 1.10 ± 0.30) (Wilcoxon matched pairs test: Z = 3.00, p = 0.002, N = 20) than after (mean ± SE: 4.20 ± 0.70). Figure 3 shows that the frequency observed for the number of visits to the square increased significantly after training while no significant difference was observed for the remaining three shapes. There were significant differences when comparing frequencies of number of visits between the four shapes after training (Friedman test: d.f. = 3, F = 10.40, p = 0.01) with around nine visits per repetition for the square while the number of visits for other shapes was always less than four (Figure 3).
Pooling the results for all shapes used during the experiment, the time spent at the shapes was significantly lower (Wilcoxon matched pairs test: Z = 2.02, p = 0.04) before training (mean ± SE: 2.50 ± 1.71 min) than after (mean ± SE: 10.30 ± 1.78 min). Table 4 shows that this difference is predominantly due to the manatee being trained using food on the square.
Mean time (min) spent by the manatee in quadrants with shapes during step 2 and step 4 for each shape
| Shape | Step 2 (before training) | Step 4 (after training) | P |
|---|---|---|---|
| Square | 0.06 ± 0.04 | 7.20 ± 1.01 | 0.04 |
| Triangle | 1.60 ± 1.10 | 1.00 ± 0.50 | NS |
| Disc | 0.16 ± 0.05 | 0.80 ± 0.37 | NS |
| Hexagon | 0.78 ± 0.56 | 1.30 ± 0.30 | NS |
Wilcoxon test, NS = not significant.
All of these results showed that training altered Daniel’s behaviour in front of the shapes, with a tendency for a higher frequency or occurrence of new behaviours, specifically regarding the square.
3.4. Observations outside the enclosed area (in open lagoon, step 5)
We performed two observations outside the enclosed area where the experimental steps were performed (step 1 to step 4). During the first observation, Daniel carried out the “look at the shape” behaviour three times (Supplementary Video 4): twice for the square and once for the hexagon. Daniel also performed the “look behind the shape “behaviour once on the square. In this case, he touched the shape with its nose for approximately two minutes. After these interactions, Daniel left the shape zones and did not return during one hour of observation. Human activity in the lagoon clearly disturbed him once it was in the open area. During the second observation, Daniel was attracted by a small boat, then swam toward the disc to “look at the shape” but subsequently did not spend more time with any of the shapes.
3.5. Observations after 3 and 12 months (step 6)
When the four shapes without food were submerged in the enclosed area, 3 and 12 months after training, Daniel only visited and interacted with the square (Supplementary Video 5). The animal repeatedly swam close to and around the square, looking behind the object during both 3 and 12 months after training. In both observations, Daniel also touched both faces of the square with its lips as if eating from it.
4. Discussion
Daniel was clearly attracted to the visual cues used in our experiment, even before one of the geometrical shapes was associated with food. The presence of new objects resulted in an obvious change in manatee behaviour with more behavioural patterns focused on the objects and a higher level of activity, especially movement. These results coincide with reactions observed in animals, for example in mice, when visual cues are not associated with food or water [33]. Attraction to the objects was particularly high for the hexagon and triangle, demonstrating that Daniel had a preference for these shapes. It is difficult to know if this preference for a hexagon and triangle is due to previous experiences with objects with a similar geometric form. Studies on dolphins [9] demonstrated that when new objects are introduced into the pool, dolphins are visually attracted by them, manipulate some of them and may have their favourite object. A similar process could be taking place in this study with new shapes, although the square is not attractive before training, maybe because it was not a new shape for Daniel. However, we have no evidence of particular shapes the manatee was in contact with before or after the training as Daniel was always living in the same conditions. Daniel visibly perceived the shapes in the water, orientating its path towards them, and inspecting them through visits and “look at the shape” behaviour. This ability for long-distance inspection of large objects has also been described by other authors [27, 34] although manatees seem to have less visual capacity than other aquatic mammals. In our study, it appears that manatees may use vision to explore their environment underwater. This result coincides with previous observations [35] for long and intermediate distances in two manatees, which presented a disparity in results between the subjects since one individual did not present visual acuity at short distances. However, manatees do not always live in clear water and it would be interesting to study whether this ability is the same for individuals in cloudy water and if manatees are able to respond to “out of the water” stimuli.
After training, Daniel clearly responded to the shape associated with food. The “playing with the shape” behaviour was only observed before training but disappeared after the manatee associated food with the square. We suggest that after the association between food and shapes, particularly the square, the geometric forms acquired a specific meaning and Daniel adjusted his behaviour according to the possibility of food being present. Daniel clearly displayed the ability to quickly associate visual cues with the possibility of food and to discriminate between geometrical shapes. This assertion is supported by the fact that he showed new behaviours after training, associated with searching for the presence of food in the shapes, with “look behind the shape” behaviour. This capacity for learning in manatees was previously observed in veterinary training [31, 34]. We did not expect that after three months a manatee would still be able to discriminate and Daniel still visibly demonstrated attraction to the square. Surprisingly, this behaviour was repeated after 12 months, accompanied by typical food research behaviour and tool manipulation. Although these results are new for manatees, the possibility of long term memory is common in mammals. For example, a border-collie named “Chaser” can memorize the name of more than a thousand objects [36], and there are bottlenose dolphins that can memorize other dolphin’s characteristics for decades [37]. This long-term memory capacity is not surprising if we consider that phylogenetically manatees are relatives of the elephant [38, 39] which is known for its excellent memory [40]. Furthermore, the capacity of a manatee to use the information learned outside the enclosed area, even if results are limited, could indicate the ability to generalize information as cues which are used to find resources in a natural area. In this context when manatees are trained before being released it may be important not to train them with shapes or object that could attract them toward human activities or constructions, but instead use natural elements found in the wild. However, in the case of Daniel, reared and imprinted by humans we consider that this is not a real problem because this manatee is always looking for human contact. This capacity, which consists in using information in different contexts, has already been described in birds and several mammals that may use learned information in their search for food areas or for refuge [41]. Several works report that manatees are able to cover large distances looking for food, freshwater, warm water or a sexual partner [22]. The ability to memorize and associate food with environmental cues has been observed in several species of invertebrates [12, 15] and vertebrates [41], and is described as an adaptive strategy to optimize foraging [12, 42]. Considering our results, it is reasonable to suggest that manatees may learn a wide range of information, allowing them to increase their subsistence ability from birth, on account of strong mother-calf interactions, interactions with other congeners [25] and individual or social experiences. We expect that if a manatee is able to use visual information underwater during foraging, manatees may also use visual information from their environment, under or outside the water, during their displacements that can cover large distances [43]. However, further studies are necessary to assess this possibility.
Because of their appearance and slow swimming, manatees are generally considered as being slow in understanding or unintelligent when compared with other aquatic mammals. This view of the manatee should change in the light of this first formal study about their learning and memory capacity. In fact, their cognitive ability, with their specificities, may be closer to that of other aquatic mammals with a large brain [30]. Some potential practical applications of our results are worth mentioning. First, after this pioneering study, more studies on manatees learning capacities are essential, using different cues to test different aspects of their environment, such as social requirements. Second, the possibility of using visual cues to train manatees could be used to manage, train and prepare them without human interaction for release into the wild and reintroduction programs. The possibilities of sensory and environmental enrichment for the welfare of the manatee in an aquarium environment could be paired with the possibility to prepare some animals for reintroduction into a new environment. In fact, our study clearly shows that the introduction of new objects in its environment resulted in increased activity and new behaviours that may help to break the stereotypical “circle swimming behaviour” described in manatees in captivity [26]. Furthermore, the association between objects and food that elicited more activities, object manipulation and active food research using short and long term memory or cues in different contexts may be a convenient way to increase manatees welfare, considering the importance of using cognition and occupational devices for this aim [4, 6]. We hope that similar studies will contribute to improving management programs for the manatees, as well as encouraging further research on manatee behaviour with the aim of improving their welfare.
Animals released into the wild have often proved to be unable to adapt to the new conditions because animals reared in unnatural conditions have to learn about predators, how to find food and how to survive in different natural conditions [44]. The vital importance of learning and preparation before being released is well documented in invertebrates [13, 15] and vertebrates [45]. In the case of manatees, several attempts at reintroduction have not been as successful as expected [46]; therefore, we consider that this study could contribute to improving the pre-release preparation of these endangered aquatic mammals and thus increase the chances of successful release and survival in their natural habitat. For example, manatees need to find freshwater and specific food items within large areas; the fact that calves live with their mother over long periods of time where they may learn the location of food and water sources, may be clearly linked with visual and other cues associated with presence of resources. This learning necessity must be taken into account in reintroduction programs.
This study provided new information about learning capacities and long term memory in manatees. However, the research focused on one solitary manatee reared by humans and it would be interesting to develop similar studies with wild manatees, using more natural items. In this context, our study also demonstrates how manatees increase their activity and number of behaviours in the presence of new objects. Therefore, introducing new objects to stimulate associative learning with visual cues provides an interesting new method to increase the welfare of manatees in captivity. Furthermore, our findings may contribute to the preparation and successful reintroduction of manatee to their natural environment. This pioneer study offers new perspectives for behavioural studies on manatees in particular those associated with cognition, management and welfare.
Conflicts of interest
The authors declare that they have no conflicts of interest concerning this article.
Acknowledgments
We thank the Ministry of Urban Development and Environment for the facilities provided for carrying out the work in the Care and Rehabilitation Centre for Aquatic Mammals. Many thanks to CONACYT and ECOSUR for providing an MSc study grant to Lara-Sánchez. We are grateful to Olivier Velázquez for the video work and also Mr. Eladio Juarez and his family for support and services offered.
Supplementary data
Supporting information (videos) for this article is available on the journal’s website under https://doi.org/10.5802/crbiol.6 or from the author.




 CC-BY 4.0
CC-BY 4.0