1 Introduction
Habitat loss and the consecutive fragmentation of the landscape are considered as the main threats to biodiversity world wide [1–3]. The prediction of population survival and evolution in small, more or less isolated suitable habitat pieces remaining in highly fragmented landscapes is a challenge for conservation biology (e.g. [4]). The metapopulation paradigm [5–8] provides an operational framework to address this crucial issue. In its essence, metapopulation biology considers the persistence of a given species in a given landscape as a balance between extinction of local populations and (re)colonisation of empty habitat patches. In such a system, dispersal rates of individuals between habitat patches are considered to be the key factor, because dispersal determines the relative importance of landscape processes (extinction and re-colonisation) vs. local demographic processes (birth and death) [7,9,10]. As species extinction in fragmented landscapes clearly depends on dispersal rates [11], we can therefore expect that strong selective pressures on dispersal are associated with habitat fragmentation.
Two different theoretical modelling approaches address this question: (1) a landscape-based approach, in which simulation models of habitat fragmentation suggest that the spatial arrangement of habitat patches matters to extinction when the proportion of the landscape covered by the suitable habitat falls below a certain threshold [11–14]; and (2) an organism-based approach focusing on the evolution of dispersal under selective pressures created by local population turnover, which indicates that processes governing dispersal rates are specific to the very functioning of each metapopulation and will result in the adaptation of the metapopulation to its landscape [15,16]. However, the validation of both these theoretical approaches remains an open question, as only a few empirical studies document the effect of fragmentation on dispersal rates and their results are not consistent [17].
Butterflies have turned out to be excellent model organisms in metapopulation biology: changes in agricultural practices in Western Europe have led to drastic transformations of man-shaped landscapes for the last 80 years and most natural habitats have been abandoned, destroyed or subdivided into small patches isolated within a matrix of cultivated fields. As many butterfly species are specialists restricted to natural habitats, they are confronted to habitat shrinking in this part of their geographical distribution (e.g. [18,19]) and they persist in metapopulations consisting of small, discrete patches of suitable habitat connected by individual movements across the landscape matrix ([20] and references therein). Considerable information is available on factors affecting butterfly dispersal and their consequence on metapopulation spatial structure and dynamics: capture-mark-recapture studies were used to estimate the dispersal rates and the maximum distance that individuals can travel (e.g. [21–24]), providing evidence that dispersal rates were constrained by the spatial configuration of the habitat patch network, i.e. patch area, shape and isolation (e.g. [9,20,25,26]).
In this paper, we investigate the effect of habitat fragmentation on dispersal in a specialist butterfly, the bog fritillary Proclossiana eunomia Esper. We take advantage from its contrasted distribution in European landscapes: continuous in the northern part of its range (Fennoscandia), vs. highly fragmented in the western and southern parts where it forms metapopulations [27]. In a first step, we stress differences between dispersal rates and their relations with patch features in a continuous and a fragmented landscape. We then turn to the analysis of land cover changes in the fragmented landscape over the last two centuries, to quantify the rate and extent of fragmentation of suitable habitats at different periods, allowing the evaluation of the time scale of the selective pressure acting on dispersal. Finally, we use a structured population model to investigate the adaptive value of the current dispersal rates in the fragmented landscape.
2 Material and methods
2.1 The species and study systems
The bog fritillary is a univoltine butterfly with adults flying in June–July, the period depending on altitude and latitude. Both adults and larvae feed on bistorts (Polygonum bistorta L.) in western and southern Europe, while larvae feed mostly on Vaccinium uliginosum L. in Fennoscandia.
The continuous patch system is situated on the eastern border of central Finland in the Kuhmo area (63°52′N, 29°10′E; Fig. 1a). The landscape is a mosaic of coniferous forest with abundant clear cuts, peat bogs and heaths. The proportion of habitats suitable to the butterfly (peat bogs) is about 16%. The study system (about 6 km2) is a dense network of 38 bogs (0.20 ha to 10.53 ha). Mean patch size is 2.46 ha±0.86 and mean connectivity index is 3.71±1.04. In the neighbourhood, bogs are very abundant with a nature reserve on the north-east and a national park on the south-west.
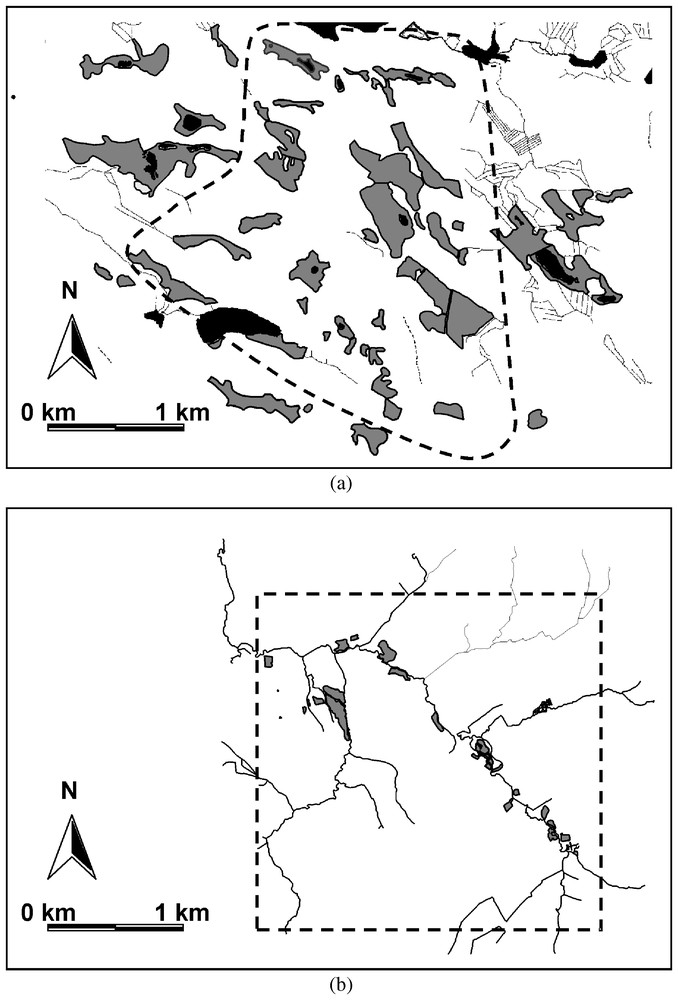
Distribution and characteristics of suitable habitat patches (grey coloured areas) for Proclossiana eunomia within the continuous (a) and the fragmented (b) landscape. Dotted lines are the limits of the study systems.
The highly fragmented patch system is located in the Ardenne region of Belgium along the Lienne river (50°18′N, 5°49′E; Fig. 1b). The study system (about 6 km2) is a network of 21 scattered patches of suitable habitat (wet hay meadows, 0.04 ha to 3.08 ha, less than 2% of the total area of the landscape), surrounded by fertilised pastures and Norway spruce plantations. Mean patch size is 0.46 ha±0.39 and mean connectivity index (as estimated by computing the area of suitable habitat within a radius of 200 m around each patch) is 0.76±0.28. No suitable habitat patches are to be found in the neighbourhood [20].
2.2 Capture-mark-recapture (CMR) study
In 2000, all patches in both systems were visited as often as possible, weather permitting, during the flight period. We performed 18 capture sessions from 24 May to 17 June in the fragmented landscape and from 21 June to 12 July in the continuous landscape. Standard transects were walked – in the continuous landscape, each patch was traversed back and forth following parallel lines – and all detected butterflies were netted and marked with an individual number on the underside of the left wing with a thin-point permanent pen. Sex was recorded at each capture, as well as coordinates of the capture location and time of the day. Butterflies were immediately released at the location of their capture. Total numbers of 293 (169 males; 124 females) and 947 (691 males; 256 females) butterflies were marked in the fragmented and continuous landscapes, respectively.
2.3 Constrained linear models
In a previous work on metapopulation dynamics of the bog fritillary [28], we used Constrained Linear Models (CLM) to estimate demographic parameters (survival, catchability, recruitment and population size) in the fragmented landscape. We applied here exactly the same methodology to derive estimates of the same parameters in the continuous landscape.
2.4 Dispersal
We analysed the effect of patch area on emigration and immigration rates within each system. Emigration and immigration rates were calculated for each patch as follow: E-rate=ER/(SPR+ER) with ER the number of recapture events involving an emigration movement from the patch i to another patch and SPR the total number of same-patch recaptures in patch i; I-rate=IR/(SPR+IR) with IR the number of recapture events involving an immigration movement to the patch i from another patch [29].
2.5 Dynamics of the highly fragmented landscape
The study area is the municipality of Lierneux, ca. 9000 ha in the Ardenne region of Belgium, where the highly fragmented patch system is located. We collected 7 high resolution (1:25 000) topographic maps on this area, ranging from 1775 to 1985. Using these 7 topographic maps, we identified changes in the landscape composition. Each map was digitalised and corrected. We then computed on each map the proportion of the landscape covered by 6 different habitat types: (1) improved grasslands and crops, (2) wet meadows, (3) mixed forests, (4) deciduous forests, (5) coniferous plantations and (6) heaths. As wet meadows are assumed to represent the suitable habitat for the butterfly, we realised various pattern measures (number of patches, size, shape, distance to the nearest patch) on this particular habitat type using the FRAGSTAT software.
2.6 Structured population model
We used comprehensive data from a long-term study in which the highly fragmented patch system was monitored during ten consecutive years to parameterise a spatially and demographic structured metapopulation model using the software RAMASGIS as a platform [30,31]. Population size was modelled as density-dependent and modulated by various climatic variables acting on different developmental stages of the butterfly. Dispersal was modelled as the mean observed proportion of movements between patches, taking into account the probability of emigration out of a given patch as estimated on the same data set with the virtual migration model [32] by Petit et al. [20].
3 Results
Total population size (±95% confidence interval) was estimated to 144±30 and 1632±791 for females and 187±25 and 1752±353 for males in the fragmented and the continuous landscape, respectively. Densities were higher in the fragmented landscape and this difference was significant for males (64 vs. 38 individuals/ha, z=4.34, P<0.001) but not for females (49 vs. 36 individuals/ha, z=1.28, P=0.100), probably because of the lower precision on females population size estimates, especially in the continuous landscape. The total population size in P. eunomia showed high yearly variations in the fragmented landscape [28], with fluctuations from 21 to 197 females/ha and 19 to 160 males/ha. More details on the constrained linear models used and the differences between landscapes are to be found in [29].
A total of 112 (87 for males and 25 for females) and 122 (103 for males and 19 for females) inter-patch recaptures (IPR) were recorded in the fragmented and the continuous landscapes, on 286 (216 for males, 70 for females) and 190 (163 for males, 27 for females) total recapture events (TR). The comparison of dispersal rates (IPR/TR) between the two systems reveals that butterflies were significantly more mobile in the continuous landscape (Chi-2=28.66, df=1, P<0.001). This effect was significant for both sexes (males: Chi-2=19.51, df=1, P<0.001; females: Chi-2=9.44, df=1, P=0.002). The dispersal rates were neither significantly different between sexes in the fragmented landscape (Chi-2=0.46, df=1, P=0.500) or in the continuous landscape (Chi-2=0.52, df=1, P=0.471).
The effect of patch size on dispersal rates was tested by pooling (1) dispersal data for males and females and (2) the patches in 4 classes according to their size (Fig. 2). Patterns of emigration and immigration with patch size were different in both systems. In the fragmented landscape, emigration and immigration rates decreased significantly with increasing patch size (emigration: Chi-2=19.72, df=3, P<0.001; immigration: Chi-2=16.03, df=3, P=0.001). Smaller patches showed higher emigration and immigration rates relative to other size classes. In the continuous landscape, patch size had no significant effect on emigration and immigration patterns (emigration: Chi-2=5.06, df=2, P=0.080; immigration: Chi-2=2.44, df=2, P=0.295).
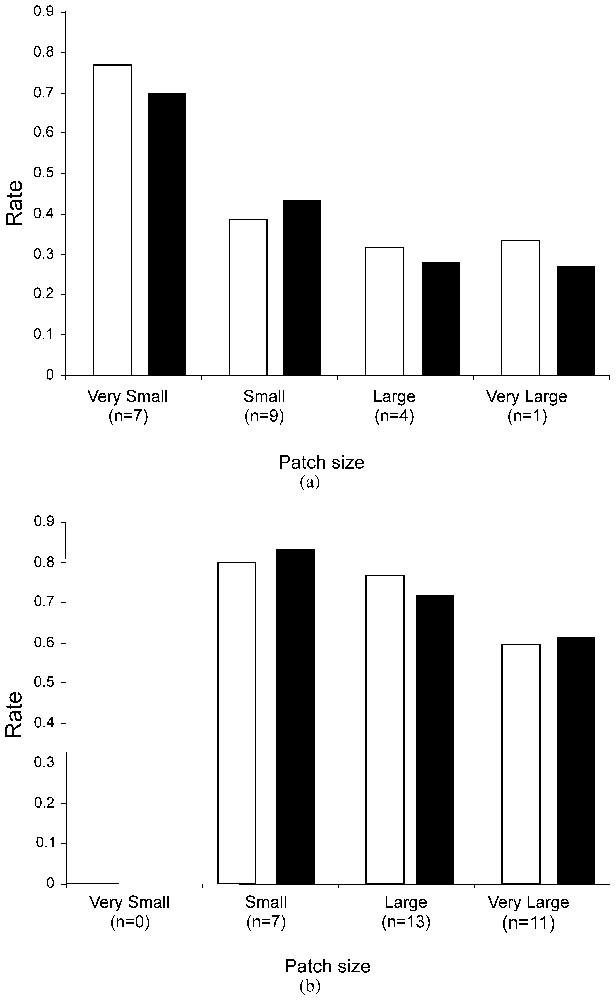
Effect of patch area on emigration (white columns) and immigration (black columns) rates in (a) the highly fragmented and (b) the continuous patch systems. According to their size, patches were pooled in four classes: very small [<0.2 ha]; small [0.2–0.5 ha]; large [0.5–3.0 ha] and very large [>3.0 ha]; n indicates the number of patches included in each size class.
Landscape dynamics in the highly fragmented patch system are shown in Fig. 3a. Our analyses show that the area of the particular habitat required by the bog fritillary butterfly (wet meadows) remained constant between 1775 and 1923; it then decreased drastically during the following 30 years and remained very low until 1985. During the same period, the spatial pattern of wet meadows was also affected in the following ways: (1) the number of patches first increased (1775–1923), due to the fragmentation of a few large patches, and then decreased (1923–1953), as the total area dropped (Fig. 3b); (2) the spatial isolation of wet meadows remained more or less constant until 1923 and then increased suddenly between 1923 and 1953, with a mean distance to the nearest patch multiplied by 3 (Fig. 3c); and (3) the shape of wet meadows changed with ribbon-like meadows found in 1775 becoming progressively small circular patches. So, we detected two important periods of changes in the butterfly landscape: (1) up to 1923, the number of patches increased, due to the break up of very large ones, very close to each other as total area and isolation were mostly unchanged; and (2) between 1923 and 1953, a sudden decrease of total area, number of patches and size of patches, resulting in a much increased spatial isolation – this situation found in 1953 still prevailed in 1985.

Landscape dynamics in the municipality of Lierneux (now the highly fragmented patch system): (a) changes in land cover between 1775 and 1985, (b) changes in wet meadows (the bog fritillary suitable habitat) mean patch size and patch number in the landscape, (c) changes in spatial isolation of wet meadows patches. Y axis is the mean distance to the nearest patch.
The adaptive value of current dispersal rates to the highly fragmented patch system was investigated by changing dispersal rates in the structured population model developed on this particular landscape [31]. We increased or decreased dispersal rates in our model and evaluated the probability that the metapopulation size falls below a given value (Fig. 4). Results indicated that dispersal rates remain far higher than the threshold value preventing recolonisations or rescue effects; this threshold value occurs when dispersal was decreased by between 99% and 100%.

Simulation of effects of changes in between patch dispersal within the fragmented landscape. The between patch dispersal matrix is increased or decreased according to the values figured in the panel. Y axis is the probability that the metapopulation falls below the corresponding size on the X axis at least once for the next 200 generations (interval quasi-extinction risk). Each curve is the mean of 1000 trajectories.
4 Discussion
Dispersal is affected by fragmentation. The comparison between the two landscapes indicates clearly that dispersal rates were lower in the highly fragmented patch system. Moreover, the effect of patch size on immigration and emigration was significant in the highly fragmented system only. As dispersal was shown to be density-dependent in P. eunomia [33], we could assume that the classical increase of density with fragmentation (e.g. [17]) should account for this difference. However, the comparison between the two systems shows that densities were similar, albeit higher in the fragmented landscape: the values observed in the continuous landscape were in the variation range of long-term estimates in the fragmented landscape [28,29]. Behavioural observations indicate that flying butterflies perceive and react to patch boundaries by flying back into the patch in the fragmented landscape (provided that the patch area reach a minimal value), while they cross much more easily patch boundaries in the continuous landscape [34]. The same differences were observed in the closely related star butterfly Melitaea cinxia, when individuals flying freely in continuous landscapes in southern France are compared to individuals circling in patches in a highly fragmented landscape in Belgium (Baguette, anecdotal unpublished observations). This change in behaviour could explain why inter-patch movements were less frequent in the fragmented landscape.
The procedure we used here is based on the comparison of two landscapes and fragmentation effects on dispersal are assessed using the continuous landscape as a reference. The two landscapes are manifestly different in various factors (e.g. structure, history, biogeographical context). Such comparative studies are therefore less powerful than experimental approaches advocated for by McGarigal and Cushman [35]. However, this comparative approach is the only way to detect adaptive response to fragmentation. In the present case, the assumption that the population very functioning before habitat fragmentation in Belgium was similar to what we observed in Finland is highly plausible, as no other significant difference in the biological parameters was detected (except from the host plant shift) [29].
Two different periods are identifiable in the landscape dynamics leading to the highly fragmented patch system. Between 1775 and 1868, the mean size of habitat patches decreased while their number increased. However, the spatial isolation of the patches in the landscape, as estimated by the mean distance to the nearest neighbour remained almost unaffected. As the only effect of patch area on the bog fritillary butterfly biology (increase of immigration and emigration rates) was detected when patch area falls below 0.2 ha, we can safely assume that the butterfly was not affected by this first change in its landscape. The second period started between 1923 and 1953 and corresponds to the time of a large change in agricultural practices everywhere in Western Europe. Both severe habitat loss and spatial isolation of remaining patches occurred simultaneously. The effect of spatial isolation on the bog fritillary butterfly populations may be estimated using data from capture-mark-recapture studies, by comparing the frequencies of individual dispersing a certain distance between successive recapture events, for each of the mean between patch distances to the nearest patch, ca. 100 m in 1923, 300 m in 1985: corresponding frequencies in the dispersal kernel are 40% and 10%, respectively. It means that the mean probability of encounter between a butterfly dispersing in the landscape and a suitable habitat patch dropped from 0.4 to 0.1 in 30 years, due to the change in patch spatial configuration only.
Modelling changes in dispersal rates in the highly fragmented patch system has no effect on metapopulation extinction, unless dispersal rates were decreased to 99% of its observed value (remember that dispersal was modelled as the mean observed proportion of movements between patches). Therefore, even if immigration and emigration are reduced by fragmentation, the dispersal rates remain far above the critical threshold leading to extinction. We must emphasise that the metapopulation spatial structure in the fragmented landscape is rather particular, with a central large patchy population and several small peripheral local populations (e.g. [28]). The presence of the patchy population explains why dispersal has to be reduced to a very low value before affecting metapopulation extinction: as distances between local population in the patchy population are usually short (mean distance 26 m), even extremely low dispersal rates allow recolonisations or rescue effects to occur. We can therefore conclude that the present dispersal rate is well adapted to the spatial configuration of habitat patches in the fragmented landscape.
5 Conclusion
Dispersal mortality was identified by theoretical models as the key factor for metapopulation persistence in fragmented landscapes [11]. As our comparisons between continuous and fragmented landscapes revealed lower dispersal rates in the highly fragmented patch system, we can expect that changes in individual behaviour leading to this decrease in dispersal were selected for rapidly (max. 30 generations) when patch spatial isolation increased. Our study, by providing evidences of the effect of habitat fragmentation on movements between habitat patches and supporting predictions from dispersal-based theories, indicates that landscape spatial configuration and connectivity have to be taken into account in the debate about large-scale conservation [36].
Acknowledgements
This paper is dedicated to the late J.M. Dumont for his pioneering work on land cover changes. GM and SP were funded by the European Commission “Training and Mobility of Researchers” programme (TMR-FRAGLAND 1998–2002) and by a grant from the Office of Scientific and Cultural Affairs (Belgian Federal Government) (contract OSTC-PADD II EV10/16A, 2000–2004) to MB. NS acknowledges a PhD grant from the National Fund for Scientific Research (FNRS). Special capture licenses for P. eunomia and site access were provided by the “Ministère de la Région Wallonne”. This is contribution BRC006 of the Biodiversity Research Center (UCL).



