1 Introduction
Aphids are widely distributed and exploit a large range of host plant species. Their reproduction dynamics often give rise to large populations, which are resources for a great number of natural enemies, including entomopathogenous fungi, predators and insect parasitoids (i.e. insects which lay eggs inside the body of an other insect, leading to the death of this host at the end of the larval development of the parasitoid). The strong selective pressure exerted by natural ennemies has led to selection for various defence mechanisms in aphids. Reciprocally, the population biology of aphids exhibits many specific traits, such as cyclic parthenogenesis, symbiotic associations or host plant specialization that influence life traits and populations of their natural enemies, especially insect parasitoids.
Primary parasitoids of aphids are found in two taxa, the sub-family Aphidiinae (Hymenoptera: Braconidae) and the genus Aphelinus (Hymenoptera: Aphelinidae). These two groups, specialized on aphids, lay their eggs in larvae and adult instars of their host. They nevertheless differ in several traits, especially those associated with reproduction [1,2]. Aphid parasitoids of the Aphidiinae sub-family have become an important model in ecological research for the following reasons.
Firstly, this group is diversified so that closely related species can be compared that range from host specific and/or host-habitat specific to generalist. This allows comparative studies on the evolution of host range in host-parasitoid interactions. Moreover, Aphidiinae, like their hosts, can be found in almost all climatic regions in the world and in a large variety of habitats including forests, fields and natural habitats from tundra to desert. This makes aphid parasitoids an ideal model for comparative studies of adaptation to both abiotic and biotic components of the environment. Secondly, a number of coexisting parasitoid species can exploit the same aphid host species, and parasitoids can differ in host specificity. This allows studying evolution of the structure of parasitoid communities and their role in the ecosystem, notably through quantitative food webs [3,4]. Thirdly, aphid parasitoids exploit aphid colonies, which have interesting dynamics: often founded by a single female, aphid colonies initially go through a phase of growth through parthenogenetic reproduction, which ensures renewal of host resources for the occurring parasitoids. This is followed by the collapse of the colony, due to the arrival of predators (like coccinellids and syrphid larvae) or to exploitation by parasitoids, and the dispersal of alate aphids away from the colony. In addition, the so-called primary aphid parasitoids are attacked by a guild of hyperparasitoids (i.e., insect parasitoids using primary parasitoids as hosts) so that aphid patches colonized by hyperparasitoids quickly lose their profitability for primary parasitoids. Accordingly, these are known to avoid aphid patches where hyperparasitoids are present. The dynamics of aphid colonies has resulted in interesting behavioural adaptations in aphid parasitoids and makes them an ideal model for comparative studies on foraging behaviour in parasitic wasps (i.e., host selection, patch time allocation, distribution of the eggs among the different aphid colonies of the habitat and life span). Finally, because of their common origin by parthenogenesis, most aphids in a colony are full sibs and the kin selection theory predicts that they may show altruistic behaviour. A well-described example is the emission of alarm pheromone by aphids once attacked by a natural enemy. This pheromone alerts siblings and elicits behavioural defence or escape from other colony members. In response, parasitoids have evolved behavioural strategies in order to reduce the emission of alarm pheromone and thus aphid defences.
All these features explain the great interest ecologists have in aphid parasitoids, especially Aphidiinae, and the abundance of recently produced data. Here, we will focus on aphid defence strategies and on the way parasitoids deal with these defences, then we will examine the responses of both aphid hosts and parasitoids to some current and future ecological constraints.
2 How aphids defend themselves against parasitoids
2.1 Behavioural defences
Individual aphids are capable of defending themselves against parasitoids with varying degrees of success [5]. Depending on the relative size of the attacking parasitoid, an aphid can respond by kicking with its legs, by quick motions of the abdomen, or through escape reactions by walking away or by dropping off the plant. Most aphid species possess a pair of cornicles, which are projections that stick out of their abdomen [6,7]. When attacked by a parasitoid, they can produce a special waxy secretion through these cornicles, as a defence mechanism (Fig. 1) [8]. A two-way defensive function of aphid cornicle secretions has been reported. Volatile (E)-β-farnesene contained in the secretion can elicit defence or escape from nearby conspecific aphids, operating as an alarm pheromone [9,10]. Also, the secretion has a sticky and hardening consistence, based on wax-like triglycerides [11] and can glue essential organs (e.g. mouthparts, antennae, ovipositor, etc.) of the attacking enemies [12]. Parasitoid wasps (Aphidius sp.) trapped in aphid cornicle secretions have been observed in the field [13] and during behavioural experiments [14]. In the Aphidius rhopalosiphi–Sitobion avenae interaction, smearing of the parasitoid results in a decreased attack rate within the aphid colony. The occurrence of this defence increases with the size of the colony, providing evidence for a case of altruistic behaviour [15].

Emission of cornicular secretion by the black bean aphid Aphis fabae (Homoptera: Aphididae) after oviposition of the parasitoid Ephedrus nacheri (Hymenoptera: Braconidae). When attacked by a parasitoid, aphids often emit such exudation, containing an alarm pheromon. This emission induces various defensive behaviours in nearby individuals and can glue various parts of the parasitoid body (Photo: B. Chaubet, INRA).
Enemy-induced changes in aphid behaviour can have different associated survival costs and benefits. In some cases, the advantage of reducing the risk of parasitoid attack can be overcome by subsequent costs in terms of quantity of energy spent, loss of feeding opportunities and death probability (e.g., dessication risk or predator attack once on the ground) [16]. The population growth rate is indeed known to be decreased due to escape responses in the presence of parasitoids – in the pea aphid Acyrthosiphon pisum [17] – or of parasitism tracks in the grain aphid S. avenae [18]. The aphids’ decision as to which anti-enemy tactic should be used probably largely depends of the costs of their behaviour [19]. As predicted, A. pisum individuals are less likely to drop or walk in response to predation risk when feeding on high quality than on low quality plant hosts, and less likely to drop when the risk of desiccation once on the ground is high (i.e., under dry and hot climate conditions) [20]. The emission of cornicle secretions can also have a physiological and ecological costs for aphids. Its production is costly in terms of energy, and reduces the amount of lipids available for development [21], reproduction [22] or dispersal [23]. Furthermore, some components of aphid cornicle secretions trigger attack behaviour in predators [24] and parasitoids, obviously serving as a stimulant for host/prey finding and attacking. For instance, the release of alarm pheromone by S. avenae is attractive for A. rhopalosiphi parasitoids [25] while the emission of cornicle secretions by A. pisum stimulates a strong oviposition attack response from Aphidius ervi females [26].
2.2 An original escape tactic: the dispersal phenotypic plasticity
Many aphid species are able to produce two alternative phenotypes: winged or wingless [27] and the winged offspring are generally produced in response to adverse environmental conditions such as crowding, poor plant quality or predation risk [28]. The pea aphid, A. pisum, increases the proportion of winged morphs in its progeny when directly exposed to natural enemies [29–33] or in response to the alarm pheromone emitted by conspecifics [34]. This is one of the first reports of a natural enemy-induced morphological shift in a terrestrial antagonist system [34]. According to Weisser [35], enemy-induced or enemy cues-induced transgenerational phenotypic plasticity in aphids is adaptive because some offspring can disperse to relatively enemy-free areas. However, Fievet et al. [36] show that the grain aphid, S. avenae, can produce more wingless individuals in the offspring when it perceives the presence of conspecifics killed by parasitoids in the colony. This strategy reduces emigration and could also be adaptive because the presence of dead aphids reduces parasitoid pressure on colonies [36].
2.3 Physiological defences
Once an endoparasitoid egg has been laid inside a host, it faces a second line of defence based on immune components. The typical insect immune response against large invaders is the encapsulation process [37], largely described in lepidopteran and Drosophila host species, with a focus on the capsule-forming haemocytes and the Phenol Oxidase cascade (PO) that leads to melanin synthesis. By comparison, very little is known of the aphid immune response against endoparasitoids. This is probably because encapsulation has seldomly been reported ([38–41] and references therein), thus suggesting that observed resistance to parasitoids is most likely based on other mechanisms.
Beyond the anti-parasitic response, the aphid immune system itself is not well documented. Among the few descriptions of haemocytes available [42–45], light microscope pictures are only found for one haemocyte category, the spherulocytes. These might play a role in immune defence since they have been observed adhering to the parasitoid egg chorion [46]. Nevertheless, fine characterization of aphid cellular immune components still remains to be performed to determine their implication in aphid defences. Few data are available on the PO cascade in aphids except that many genes potentially involved in this cascade are present in the pea aphid A. pisum genome [47]. Potential involvement of the PO cascade in the antiparasitic response is suggested by reports of melanization events following parasitism [41,48] as well as by overproduction of the immature form of phenoloxidase (proPO) in aphids resistant to A. ervi [49].
Clonal resistance to braconid parasitoids has been described in populations of A. pisum [50–53], Myzus persicae [54,55] and Aphis fabae [56], although it is currently considered quite rare. In a resistant aphid, failure of the parasitoid can occur either at an early stage when the egg fails to develop [38,50,52] or at the larval stage [57]. Comparative studies between resistant and susceptible clones of A. pisum have linked failure of the parasitoid development to various causes. It may be due either to incomplete deployment of teratocytes, i.e. giant cells originating from the dissociation of the serosal membrane that surrounds the developing parasitoid embryo [58], to temperature [46] or to the presence of secondary symbionts members of the Enterobacteriaceae, Serratia symbiotica and especially Hamiltonella defensa [57,59,60]. H. defensa has been reported to provide resistance to A. pisum against both A. ervi and Aphidius eadyi [46,57,59,60], and more recently to A. fabae against Lysiphlebus fabarum [56]. Resistance appears to be largely correlated to the presence of a bacteriophage of H. defensa, APSE (A. pisum secondary endosymbiont) [61] which encodes homologs of bacterial eukaryote-targeting toxins from three protein families. Some of these toxins might be responsible for prematurely arresting the development of parasitoid larvae [62–64].
Symbiont-mediated protection is currently described as the main mechanism of resistance in aphids but clones without secondary symbionts also exhibit significant clonal variation in resistance suggesting existence of an aphid innate resistance [55,56]. Vorburger et al. [56] suggest that aphid parasitoids may be confronted with two lines of defence: “aphid innate defences” and “acquired defences” provided by secondary endosymbionts, which likely differ in their effectiveness and specificity. Understanding resistance to parasitoids in aphids will thus require the assessement of aphid immune defences as well as that of parasitoid strategies used in exploiting the host's physiology and maybe in circumventing its immunity (see § 3.3).
3 How parasitoids exploit aphid ressources
3.1 Aphid localization: role of the plant and of aphid density
Host localization by aphid parasitoids is mediated by different cues. The aphid's host plants play a key role by emitting volatile substances used as long-range cues by foraging parasitoids. Depending on the aphidiin species, females can be attracted either by undamaged plants or by plants attacked by aphids. For example, the parasitoid Diaeretiella rapae is attracted by isothiocyanates, volatile chemicals produced in Brassica plants [65], whether attacked or not by its aphid host, the woolly cabbage aphid Brevicoryne brassicae. In contrast, the plant-host complex is necessary for A. ervi attraction by broad bean plants infested with A. pisum [66]. Attacked plants often produce odours attractive for parasitoids [67], which A. ervi can learn and memorized for 3 days [68]. A review of aphid-induced plant volatiles and their effects on habitat and host location by parasitoids can be found in Hatano et al. [69].
Recognition of the host itself by the parasitoid occurs through its cuticular pheromones, as shown by the fact that aphid exuviae induce parasitoid attacks [70,71], or through its cornicle secretions, which contain different pheromones [68]. Glinwood et al. [72] have indeed demonstrated the role of aphid sexual pheromones in attracting parasitoids. Finally, host recognition can occur using combinated visual and chemical cues as demonstrated for A. ervi [71,73].
Increases in host density are classically known to increase the attractivity of a colony for several aphid parasitoid species [74] possibly because of increases in the release of pheromones. A counter example is Aphidius funebris whose host colonies become less attractive after parasitoid attack due to the rapid increase of alarm pheromone concentration when densities bypass a given threshold [75].
3.2 Patch exploitation strategies and host resource sharing in aphid parasitoids
A particularity common to aphid-parasitoid systems is the fact that aphids of all instars show active defensive behaviour against parasitoids (see § 2.1). Not all parasitoids react in the same way to these defences and this has resulted in different patterns of ressource exploitation strategies in different parasitoid species.
One of the best studied examples is a guild of three closely related [76] solitary parasitoids belonging to the genus Aphidius (A. rhopalosiphi, A. avenae and A. ervi), attacking the cereal aphid S. avenae in Western Europe and overlapping in phenology (spring and early summer) [77]. A. rhopalosiphi is the most abundant species, present in the field all year round and already abundant when the two other species arrive [77]. However, it exploits patches only partially, and leaves a patch when parasitized aphids start emitting cornicular secretions [14,78]. The two other species have developed different strategies to avoid the defensive behaviour of aphids: A. ervi oviposits very quickly and has an efficient way to wash out the cornicle secretion from its body, while A. avenae oviposits at a rate 40 times slower than that of A. rhopalosiphi. By doing so, A. avenae rarely elicits defense behaviour from the host [77,79,80]. Hence, A. ervi and A. avenae can use a part of the host population not exploited by A. rhopalosiphi.
Both intra- and interspecific host discrimination (i.e. the ability of female parasitoids to distinguish between unparasitized and parasitized hosts, belonging to the same or to another parasitoid species, respectively) are important parameters in host selection and patch time allocation of aphid parasitoids. Host discrimination provides information about the level of exploitation of the host population, and thus on the quality of a patch. Intra-specific host discrimination has been reported in the majority of aphid-parasitoid species studied. Two mechanisms have been described that involve the recognition of an external or an internal mark, which are primarily of chemical origin. Internal marks may result from injection of components by the ovipositing female, or from host physiological modifications induced by the delopping parasitoid larva. External marks can also be left by the ovipositing female or may be linked to host defences such as cornicular secretions. Some species use only an external mark (Lysiphlebus testaceipes on Aphis gossypii), some only use an internal one (A. avenae [77]), and some use both (A. ervi [80]). The parasitoid A. rhopalosiphi uses the host aphid's alarm pheromone to avoid already exploited patches [81].
Published evidence for interspecific host discrimination is restricted to a few cases of closely related species [82,83], or to pair of species having asymmetric competitive interactions [84,85]. In aphid-parasitoid systems, interspecific discrimination occurs for example between Aphidius smithi and A. ervi [86] and between A. ervi and Aphelinus asychis [87]. Within the above-mentioned guild of Aphidius species [80], rejection of hosts already parasitized by the other species can occur after insertion of the ovipositor with A. rhopalosiphi and A. avenae, but most often before ovipositor insertion with A. rhopalosiphi and A. ervi. Hence, interspecific host discrimination in these three Aphidius species is based on either an internal mark only or in combination with an external mark. A. rhopalosiphi uses an internal mark to detect hosts parasitized by A. avenae but an external one to recognize hosts already parasitized by A. ervi. This may be due to the fact that hosts parasitized by A. avenae are very quiet, while hosts parasitized by A. ervi have been alerted and are ready to emit cornicular secretion [80].
3.3 Host manipulation and parasitoid development success
The development of an endoparasitoid inside the body cavity of its insect host relies both on its success in avoiding the host's defences and its ability to exploit the host's resources to promote its own development. This is generally achieved using factors produced in the venom apparatus and/or in the ovaries, that are co-injected with the egg into the host [88]. In addition to these factors, teratocytes contribute to ensure larval development of braconid and some platygasterid and scelionid wasps. Teratocytes do not reproduce but they increase in size, may become polyploid and their growth is synchroneus with that of the parasitoids. They can play a role in immune suppression as well as nutrition [89,90].
To deal with host immunity, parasitoids use a variety of strategies ranging from escaping immune recognition to actively suppressing the immune response [88]. However, little is known of the strategies used by aphid parasitoids. The example of Monoctonus paludum suggests that some species might use a hiding strategy: this parasitoid inserts its ovipositor close to the ventral nerve ganglia of the host, so that the deposited eggs are embedded in the nerve ganglia and are most likely protected from circulating hemocytes [38]. To our knowledge, there is still no report of host immunosuppression by aphid parasitoids but this might simply be due to the lack of data on aphid immune responses and of analyses of venom and teratocyte-secreted proteins.
Parasitoids have developed many ways to exploit the host resources, based on the use of venom or teratocyte-secreted proteins. In Aphidiinae, host castration would allow reallocation of resources toward parasitoid development by dramatically reducing nutritional competition between host reproductive tissues and parasitoid juvenile stages. Castration of A. pisum by A. ervi has been reported and it is mediated by the combined effect of venom [91,92] and teratocytes [93]. The venom targets the upper part of the host's ovarioles, inducing the degeneration of germaria and young apical embryos [92]. This is due to the activity of a major component of the venom, a Gamma-Glutamyl Transpeptidase (GGT), which triggers apoptosis of the cells in the germaria and ovariole sheath of the aphid [94]. In very young host instars, ovarioles do not develop at all and castration is complete. In later instars that already contain developing embryos, castration is achieved by teratocytes. They selectively target embryos, performing some kind of extra-oral digestion and thus releasing nutrients available for the parasitoid larva [93]. A major protein released by the teratocytes, that belongs to the fatty acid-binding protein (FABP) family, is involved in the transport of fatty acids from the site of digestion of host tissues to the parasitoid larvae [95]. Several lines of evidence also support the hypothesis that aphid parasitoids exploit the nutrititional interaction between aphids and symbiotic bacteria of the Buchnera genus. These bacteria, found in specialized aphid cells, the bacteriocytes, provide the aphid with essential amino acids, in short supply in the aphid diet of plant phloem sap [96]. Pennacchio et al. [97] found that the parasitoid A. ervi performs poorly in aposymbiotic pea aphids (aphids made artificially nonsymbiotic), while Rahbé et al. [98] demonstrated that in parasitized aphids, essential amino acid synthesis by Buchnera is selectively preserved or promoted in the case of tyrosine. Finally, the number and biomass of bacteriocytes is significantly higher in parasitized aphids and this effect varies between aphid clones in a fashion consistent with their relative susceptibility to parasitism [99]. Although the between-clone variation in bacteriocytes features might result from other parasitoid-mediated effects, susceptibility of the aphid-Buchnera symbiosis to parasitoid-mediated manipulation might contribute to the susceptibility of aphids to parasitoid exploitation [99].
At an interspecific level, the host range of aphid parasitoids is partly shaped by their ability to interact with the metabolism of their various hosts. This might explain, for example, many cases of parasitoid development failure before mummification, associated with strong effects on aphid fecundity and longevity [100]. These effects are very similar to those observed in resistant clones at an intraspecific level (see § 2.3), such as described for populations of the parasitoid D. rapae on one of its hosts, Rhopalosiphum padi [Le Ralec, unpublished data]. In this case, host castration seems only partial, and the parasitoid larvae are smaller and teratocytes less numerous than in more suitable hosts, B. brassicae and M. persicae. Whether mechanisms implied in clonal resistance and in host suitability at the species level are identical remain to be demonstrated.
4 How to deal with ecological constraints
4.1 Diapause strategies in parasitoids and mechanisms of diapause induction
A particularity of most of the aphid species is that they are holocyclic with two alternating modes of reproduction: sexual reproduction, with females laying eggs after being inseminated by males (oviparous morphs) and parthenogenetic reproduction with females that reproduce without insemination and produce larvae (viviparous morphs). Holocyclic populations of aphids can overwinter by producing cold resistant eggs, which are not attacked by parasitoids. In temperate regions with mild winters, anholocyclic aphid populations that only reproduce by parthenogenesis also occur and coexist with holocyclic populations [101,102]. To synchronize with the life cycle of their hosts, aphid parasitoids may thus have to enter diapause to overwinter if few hosts are available.
Parasitoid species that have only one generation a year (i.e. univoltine species) enter an obligate, genetically determined diapause, whereas multivoltine species use a variety of abiotic (temperature, photoperiod) and biotic (low host resource levels, hormonal state of the host or changes in host-plant physiology) signals for the induction of diapause [103]. Aphidiinae parasitoid species, which are in majority multivoltine species, are a typical group as they present both types of signals for diapause induction [104].
Short days and low temperatures induce the production of sexual aphids that lay diapausing eggs. Aphid parasitoids may overwinter by entering a prepupal diapause, within the body of the aphid, dead at a larval or adult instar (mummy stage), which means that the normal metabolism is stopped [105–107]. In Aphidius species, diapause can be induced by low temperature and short day length, either directly by exposing the juvenile parasitoid stages to these conditions or indirectly by exposing parasitoid females during their adult life, which will increase the proportion of diapausing offspring in the progeny [105–108]. However, climatic conditions do not always induce diapause in parasitoids. Under short day conditions and low temperatures, individuals of Aphidius uzbekistanicus Luz developing on holocyclic populations of S. avenae enter diapause, whereas those developing on anholocyclic populations do not [109].
Parasitoid diapause can also be induced by biotic cues such as the internal state of the aphid in which it develops. In the parasitoids Aphidius matricariae Haliday and Praon volucre Haliday (Hymenoptera: Aphidiinae) that develop on oviparous morphs of the black bean aphid A. fabae (Homoptera: Aphididae), 70–90% of the larvae enter diapause even under high temperatures (21 °C) and long day conditions (16 h light–8 h dark). This diapause is likely independent of environmental cues or maternal effects but may be induced by the higher levels of ecdysteroid hormones of oviparous morphs in comparison to those of the viviparous ones [106,110]. In other species, diapause can only be induced by a cold shock in oviparous or viviparous aphids [111]. Finally, a third trophic level, the plant, might be implicated in induction of parasitoid diapause. Indeed, the host plant plays a significant role in the polymorphism of aphids and low quality plants may induce longer development times in both aphids and their parasitoids [103].
Aphid parasitoids may also use biotic cues as an indication of declining host populations or unfavourable climatic conditions. In summer, populations of cereal aphids often crash in Western France and A. rhopalosiphi can enter diapause [112]. In this species, summer diapause does not seem to be initiated by climatic conditions but is rather induced by a reduction of the number of hosts available [112]. In specialist species, diapause is an adaptation to the life cycle of their host that enables parasitoids to survive all year in a single environment composed of the aphid primary host plants when host resources are low [113]. For example, Ephedrus persicae, which attacks the rosy apple aphid Dysaphis plantaginea, enters diapause in spring before its host has completely left the apple trees [114]. For generalist species, diapause induction via the aphid host also occurs, but entering diapause too early may prevent exploitation of available alternative host resources. For example, A. matricariae does not enter diapause in several anholocyclic aphid species such as Aulacorthum solani and A. gossypii [111] but it enters diapause in viviparae of holocyclic M. persicae [103]. In the generalist A. ervi, diapause induction in oviparous morphs of A. pisum has been explained as an adaptation of the parasitoid to the life cycle of its main host [106]. Diapause may also be initiated by host and parasitoid density dependent processes, for example to avoid a period of intense competition for hosts in the next generation [105]. In A. uzbekistanicus, when several larvae compete for the same host, which implies a high density of emerging adults and thus fewer hosts available at the next generation, the larva that wins the competition can enter diapause [115].
As a conclusion, diapause is an adaptive and often phenotypic plastic life history trait that allows parasitoids to synchronize their life cycle with seasonal climatic changes and host resources.
4.2 Aphid host plant specialization – a promoter of parasitoid diversification?
Many organisms live in heterogeneous environments and are exposed to divergent selection on traits associated with resource use. Depending on the balance between selection and gene flow, microhabitat-specific genetic structuring may occur, leading to the formation of genetically distinct and specialized populations [116]. Because of the intimate relationship with their host plants, phytophagous insects are considered to be prime candidates for such ecological specialization [117]. There are now many excellent examples of genetic differentiation among populations of phytophagous insects feeding sympatrically on different host plant species [118]. This ecological specialization leads to the formation of what is called ‘host-races’ and is often considered as the first step towards sympatric speciation. Host races have been reported in a growing number of phytophagous insects, the most famous example being certainly the shift of the apple maggot fly (Rhagoletis pomonella) from hawthorn (Crataegus spp.) to domesticated apple (Malus pumila) [119–125].
Even if host plant-associated genetic differentiation is a common phenomenon in phytophagous insects, the degree to which such associations sequentially drive diversification at higher trophic levels is not as well analysed [126]. Phytophagous insects are attacked by a diverse assemblage of insect parasitoids, but mechanisms of population and species divergence in parasitoids are largely unexplored. A particularly interesting possibility is that host-associated differentiation (HAD) may cascade across trophic levels if HAD in a herbivore leads in turn to divergence of its parasitoids. Stireman et al. [127] first reveal the existence of cascading HAD in parasitoids of two phytophagous insects, each of which consists of genetically distinct host-associated lineages on the same pair of goldenrods (Solidago sp.). Each parasitoid exhibits significant host-associated genetic divergence, and the distribution and patterns of divergence are consistent with divergence in sympatry. Although evidence for cascading HAD is currently limited, these authors suggest that it could play an important role in the diversification of parasitoids attacking phytophagous insects.
In aphids, host races have been described [27,128–131], especially in the pea aphid A. pisum, which encompasses distinct races highly specialized on various Fabaceae species [53,132–137]. Up to now, 11 pea aphid host plant races have been genetically delineated [137]. These host races differ in numerous and diverse traits such as reproductive mode [102], dispersal phenotype [138], body colour, prevalence of facultative endosymbionts [139], susceptibility to natural enemies such as parasitoids [51] and fungal pathogens [140], and defensive responses towards predation risk [141]. Until now, it is not clear to what extent HAD in aphids may affect population structure or divergence at the parasitoid level and how it may trigger codivergence in their parasitoids. Given the increasing interest in coevolutionary processes, studies examining the degree to which higher trophic levels evolve in response to genetic structure of aphids populations will be important for understanding species interactions and diversification [142].
4.3 Consequences of climate change on aphid-based multitrophic systems
In ecosystems, multitrophic interactions often result from a long co-evolutionary process specific to a particular environment including climatic conditions. Temperature changes may differentially affect each component species of the system, possibly resulting in a destabilisation of the dynamics that could lead to the extinction of part of the system [143].
Aphids have a very short generation time for insects, generally shorter than that of their parasitoids. This life history trait, as well as other parameters affecting parasitoid and host population dynamics (e.g. fecundity, longevity, parasitoid attack rate), may change with temperature. In an other type of natural enemy, Harrington et al. [144] found that below 11 °C, the reproductive rate of the pea aphid, A. pisum exceeds the rate at which the coccinellid predator Coccinella septempunctata L. can consume the population, but that the reverse occurs above 11 °C.
An increase in temperature will result in an increase in metabolic rate and subsequently in the general activity of an insect. This, in turn, could result in an increase in a parasitoid's realized fecundity if its attack rate on hosts increases. However, it may also result in a decrease in longevity due to the accelerated use of energy, which could limit the realized fecundity. The reduction in longevity is then a constraint on lifetime reproductive success of the parasitoid.
Aphids defensive behaviours (see § 2.1) are also affected by temperature: when temperature increases, pea aphids show a reduced tendency to drop off the plant in response to the presence of natural enemies. This might decrease their risk of dehydration but makes them more vulnerable to parasitoids [145]. Sex allocation in aphid parasitoids might also be affected by temperature because temperature affects host size [146]. Host size is linked to sex allocation in several species of parasitoids that tend to lay fertilized eggs more often on larger hosts thus producing more female offspring. However, female offspring may be allocated to smaller hosts when the average host size decreases [147], resulting in smaller parasitoid females. To date, no empirical evidence is available that shows a relation between temperature and sex-ratio in aphid parasitoids.
Aphids and their parasitoids harbour two major groups of endosymbionts, Buchnera and several secondary endosymbiotic bacteria in aphids, and Wolbachia in parasitoids, which may be affected or even eliminated by short exposures to high temperature [148]. In field populations of A. pisum, the endosymbiont H. defensa (see § 2.3) provides variable levels of protection against parasitoids [59,61,63] and this symbiont-mediated protection fails under heat stress [46]. In contrast, Chen et al. [149] showed that under constant rearing at 25 °C, other secondary endosymbionts, S. symbiotica and Rickettsia, can confer heat-tolerance to the pea aphid A. pisum, whereas at the low temperature of 20 °C, they can decrease its fecundity, depending on the clone and the host plant.
The geographical distribution of aphids and their associated parasitoids might be less affected by climate changes than that of other insect host–parasitoid systems, because of their large dispersal capacity. Within the area where abiotic conditions are tolerable for an herbivore, aphid distribution may be limited by the availability of host plants. Likewise, the distribution of their parasitoids may be limited by the availability of their hosts. However, in aphid-based communities, both parasitized and unparasitized aphids can be dispersed by wind [Hulle, Pers. Comm.]. Dispersal of parasitized aphids could thus result in dispersal of their parasitoids.
The phenological synchrony between plants and insects, and between hosts and parasitoids, could possibly become uncoupled if the two processes are temperature-driven in different ways. This has been shown in different tritrophic systems, with heavy consequences on population dynamics [144,150], but at this moment no empirical evidence exists for aphid-parasitoid systems. Variation in the proportion of hosts and parasitoids entering diapause is also influenced by temperature: in aphid-parasitoid systems in cereal fields in Western Europe, a proportion of both aphid and parasitoids populations enter diapause, while the rest of the population continues its reproductive activity throughout the winter (see § 4.1). In cold winters, non-diapausing aphids and parasitoids will suffer a high rate of mortality and at the beginning of the spring the diapausing part of the population will be the most important, while in mild winters non-diapausing parasitoids and aphids will survive better than in cold winters, resulting in larger host and parasitoid populations in early spring. As early spring population density is a good predictor of the population dynamics during the rest of the season [151] and parasitoids are the second most important mortality factor of aphid populations in Western Europe [152], the population dynamics of aphids is strongly linked to average winter temperature in this area. Parthenogenetic aphids produce nowadays around 18 annual generations in UK and an increase of 2 °C of temperature will allow five generations more [153]. When average temperature increases, the rate of increase of parasitoid populations will grow less quickly than that of aphid populations [144]. Several models of host-parasitoid interactions have indeed predicted an increase in pest-outbreaks with climate change, mostly because of an increase in the number of generations of hosts [143,144,154–157].
5 Conclusion
Aphids exhibit many original biological and ecological features, which strongly influence their parasitoid life traits. Interestingly, aphid parasitoids are found in only two highly specialized taxa with limited species numbers. In this article, we have highlighted some specific aspects of aphid-parasitoid interactions based on recently published data, as well as the promising perspectives for the use of this model in future research (Fig. 2).
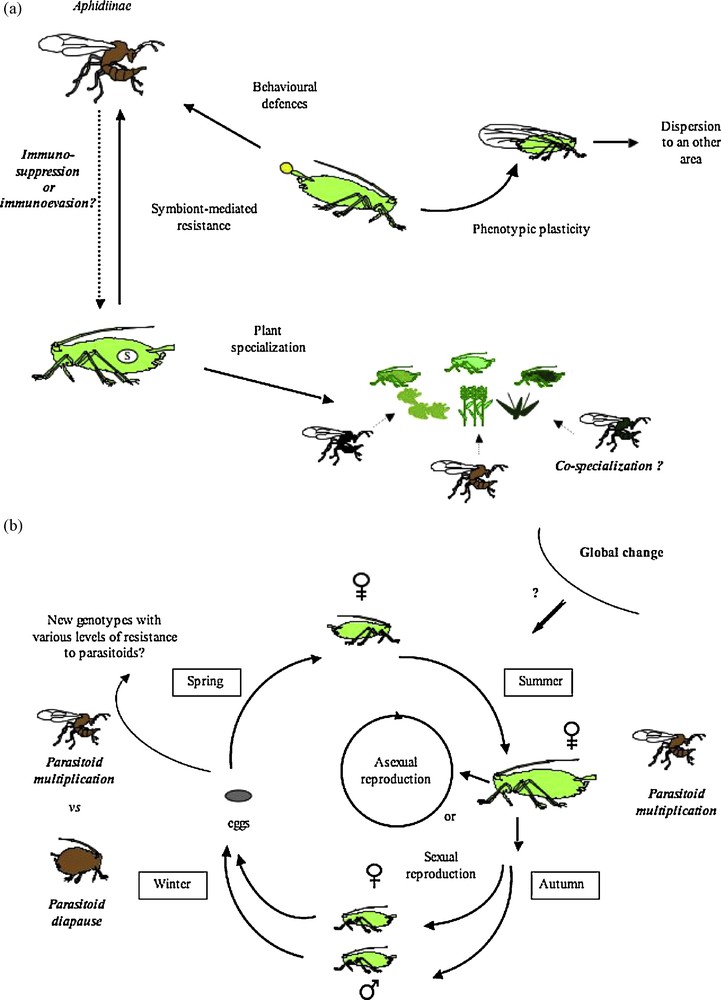
a: biotic interactions of aphids (see § 2.1, 2.2, 2.3, 3.3, 3.2 for more explanations) (S: symbionts); b: aphid life cycle (see § 4.1, 4.3 for more explanations).
Aphids have developed both behavioural and physiological defences to parasitoid attacks. The way parasitoids manage to deal with the behavioural defences is already known to influence the exploitation of hosts as ressources and to contribute to ressource sharing by various parasitoid species. In contrast, the understanding of the mechanisms involved in physiological defences is only at its beginning. However, the apparent lack of parasitoid encapsulation in aphids and the importance of secondary symbionts in aphid resistance suggest original mechanisms of resistance compared to what is known of other insect–parasitoid associations.
Our knowledge of the physiological interactions between aphids and aphidiines during parasitoid development including that of the parasitoid factors involved in regulating host metabolism, has increased during the last 10 years [90,97,158]. However, much remains to be done to characterize the interactions between aphids and parasitoids of the Aphelinidae family, despite their interesting features such as the lack of teratocytes.
At the population level, the evidence of plant-associated genetic differentiation in several aphid species now allows testing the hypothesis of a corresponding specialization in associated parasitoids. Other hypotheses in the field of co-evolution arise from the co-existence of sexual and asexual lineages in a single aphid species [159], such as the advantage of sexual reproduction in the arms race between aphid resistance and parasitoid virulence.
At the community level, the ability of a parasitoid to find hosts using plant and/or host cues, to deal with various aphid defence mechanisms and to manipulate the host's metabolism for its own development will shape its host range. The great majority of aphidiine parasitoids have a limited host-range, restricted to only a few host species, while a minority of species are generalist. Aphids developing on different host plants may share one or more parasitoid species, which might lead to apparent competition between aphid species, i.e. competition mediated by their natural enemies [160,161]. Geographic variation in the parasitoid host range may also occur, due to variation in the local pool of available host species, e.g. following changes in local climate [162].
Beyond the interest of aphid parasitoids as models for ecological and evolutionary research, they are important auxiliaries for biological control of crop pests. Inundative releases of A. ervi, for instance, are used in several European countries [163, this issue]. Fundamental studies on aphid-parasitoid interactions might thus help to enhance the efficiency of aphid parasitoids in biological control. Similarly, multitrophic approaches on aphids, host plants, endosymbionts, parasitoids, parasitoids predators and hyperparasitoids could result in new strategies for pest control. For instance, natural parasitoid control of a pest aphid species might be enhanced by favouring a non-pest host species to create an apparent competition between the two hosts [164,165]. Again, favouring host races of aphids specialized on non-crop plants will allow developing of parasitoid populations which will latter attack aphids on cultivated plants.
Aphid–parasitoid associations are thus suitable biological models to explore ecological and evolutionary theories, and their study could result in new ways to manage populations of aphid pest species.
Acknowledgements
The authors acknowledge Dr Jacqueline Pierre for fruitful comments.


