1 Introduction
We have surveyed the Cambridge Structural Database [1] seeking unusual supramolecular assemblies. It is important to point out that there is no current software, which automatically assembles a hydrogen-bonded network. Molecular graphics program Mercury (version 3.1) (http://www.ccdc.cam.ac.uk/Solutions/CSDSystem/Pages/Mercury.aspx) provides a “Network” command as an excellent starting point, but complete assembly requires of the crystallographer considerable time and expertise to manipulate the structural data contained in the crystallographically independent unit.
Described in this paper are suprahalide anions of the form HnXn + 1− (X = F, Cl) found in 45 crystal structures, each retrieved from the CSD and identified by its REFCODE. Two equivalent and complementary search criteria were used to screen candidates for this study:
- • quantitative – retrieved single crystal structures must have no disorder, no errors, R values less than 5%, and H..X internuclear distances significantly less than the sum of van der Waals radii;
- • visual – supramolecular assemblies must show significant interpenetration of van der Waals envelopes in molecular graphic models.
The van der Waals radii adopted for this study are those implemented in Mercury 3.1: H = 1.20, F = 1.47, Cl = 1.75 Å. Each H..X internuclear distance was assigned an estimated standard deviation of 0.05 Å, and the maximum H..F and H..Cl values accepted for this study are 1.99(5) and 2.50(5) Å respectively, significantly (99% CI) less than the sum of radii.
The three well-known and well-characterized halide ions are monatomic fluoride, F−, monatomic chloride, Cl−, and triatomic bifluoride FHF−. Each suprafluoride anion described below contains a central fluoride or bifluoride anion hydrogen bonded to one or more HF molecules. The suprachloride anions are structured in similar fashion, except that we could find no instances of HCl molecules hydrogen bonded to bichloride, ClHCl−. Since there is no widely accepted symbolism for supramolecular assemblies, we propose to formulate the supramolecular halide anions described herein as follows: (F)(HF)x−, (FHF)(HF)x−, (Cl)(HCl)x− and (ClHCl)−.
There is a theoretical distinction between the two triatomic anions HF2−. In molecular bifluoride (FHF)−, the three atoms are strongly bound by 3-center-4-electron covalent bonds [2]. In supramolecular (F)(HF)−, there is one formal H–F 2-center-2-electron covalent bond and one F..H hydrogen bond, distinguished from one another by their H..F internuclear distances. Every observed F..H..F triplet consists of two proximate internuclear H..F distances L1 and L2, and we define the triplet asymmetry parameter to be the ratio of the absolute difference to the average: δ = 2|L1–L2|/(L1 + L2). By measuring L1 and L2 for all F..H..F triplets described below, we have found that δ is less than 0.05 for all moieties which can be described as 3-center-4-electron molecule ions, while δ is greater than 0.20 for all H-bonded moieties (Fig. 1, Table 1). We estimate the standard deviation of δ to be 0.06. Thus, the unpopulated gap between 0.05 and 0.20 is significant at the 99% confidence level and provides an experimental boundary between molecular (FHF)− and supramolecular (F)(HF)−.

Triplet asymmetry parameter δ in HnFn + 1− anions.
HnFn + 1− anions.
| REFCODE | Reference | L1 | L2 | δ | Form |
| BOCKEK | [3] | 1.13 | 1.09 | 0.036 | (FHF) |
| CETPIB | [4] | 1.10 | 1.10 | 0.000 | Cu(FHF)Cu |
| CIHJOT | [5] | 1.20 | 1.16 | 0.038 | (FHF)(HF) |
| CIHJOT | [5] | 1.23 | 1.17 | 0.047 | (FHF) |
| CIHJOT | [5] | 1.30 | 1.02 | 0.237 | (FHF)(HF) |
| DEHSOY10 | [6] | 1.34 | 0.99 | 0.302 | (F)(HF) |
| DEHSUE10 | [6] | 1.52 | 0.82 | 0.595 | (F)(HF)2 |
| DEHSUE10 | [6] | 1.55 | 0.80 | 0.634 | (F)(HF)2 |
| DEHTAL10 | [6] | 1.52 | 0.87 | 0.538 | (F)(HF)3 |
| DEHTAL10 | [6] | 1.52 | 0.86 | 0.549 | (F)(HF)3 |
| GEPYUV | [7] | 1.15 | 1.13 | 0.018 | (FHF)(HF)3 |
| GEPYUV | [7] | 1.37 | 1.06 | 0.256 | (FHF)(HF)3 |
| GEPYUV | [7] | 1.40 | 0.98 | 0.354 | (FHF)(HF)3 |
| GEPYUV | [7] | 1.56 | 0.88 | 0.557 | (FHF)(HF)3 |
| GEPZIK | [7] | 1.33 | 0.84 | 0.447 | (F)(HF)6 |
| GEPZIK | [7] | 1.48 | 0.84 | 0.551 | (F)(HF)6 |
| GEQBEJ | [7] | 1.50 | 0.86 | 0.538 | (F)(HF)3 |
| GEQBEJ | [7] | 1.61 | 0.77 | 0.711 | (F)(HF)3 |
| GEQBEJ | [7] | 1.64 | 0.77 | 0.723 | (F)(HF)3 |
| GEQBIN | [7] | 1.47 | 0.89 | 0.484 | (F)(HF)5 |
| GEQBIN | [7] | 1.53 | 0.89 | 0.532 | (F)(HF)5 |
| GEQBIN | [7] | 1.51 | 0.83 | 0.585 | (F)(HF)5 |
| GEQBIN | [7] | 1.64 | 0.86 | 0.627 | (F)(HF)5 |
| GEQBIN | [7] | 1.67 | 0.80 | 0.708 | (F)(HF)5 |
| GIPGOB01 | [8] | 1.43 | 0.89 | 0.467 | (F)(HF)2 |
| GIPGOB01 | [8] | 1.44 | 0.89 | 0.469 | (F)(HF)2 |
| GIPHAO | [9] | 1.13 | 1.13 | 0.000 | (FHF)(HF)4 |
| GIPHAO | [9] | 1.72 | 0.76 | 0.773 | (FHF)(HF)4 |
| HITBIW | [10] | 1.32 | 1.07 | 0.211 | (F)(HF)2 |
| HITBIW | [10] | 1.36 | 0.96 | 0.350 | (F)(HF)2 |
| OCAZUN | [11] | 1.51 | 0.89 | 0.514 | Pd(F)(HF) |
| OLIYEO | [12] | 1.16 | 1.16 | 0.000 | Cu(FHF)Cu |
| OLIYEO | [12] | 1.47 | 0.92 | 0.465 | Cu(F)(HF)Cu |
| QIWPUH | [13] | 1.30 | 1.05 | 0.214 | Ru(F)(HF) |
| VEMKAZ | [14] | 1.66 | 1.08 | 0.427 | Nb(F)(HF)Nb |
| WEGPOO | [15] | 1.20 | 0.89 | 0.302 | (F)(HF)2 |
| XAYMOB | [16] | 1.99 | 0.63 | 1.041 | Sb(F)(HF) |
However, while both (ClHCl)− and (Cl)(HCl)− anions clearly exist at the extremes (Fig. 2, Table 2), δ values for the observed Cl…H..Cl triplets vary almost continuously, so, the triplet asymmetry parameter does not offer a clear distinction between molecular and supramolecular HCl2−.
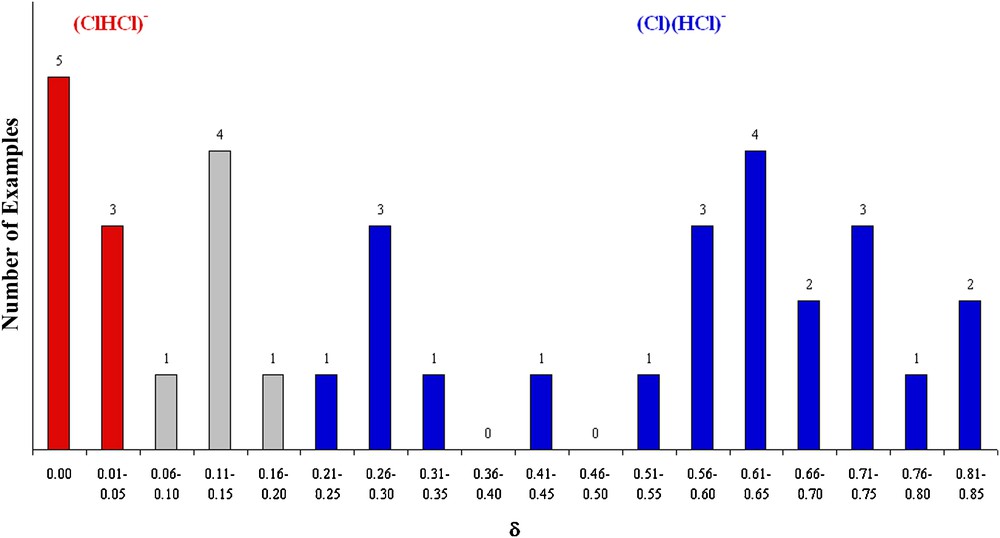
Triplet asymmetry parameter δ in HnCln + 1− anions.
HnCln + 1− anions.
| REFCODE | Reference | L1 | L2 | δ | Type |
| ATEYEE | [17] | 1.43 | 1.43 | 0.000 | Bi(ClHCl)Bi |
| AZOLUX | [18] | 1.56 | 1.56 | 0.000 | (ClHCl) |
| BAMSAJ | [19] | 1.81 | 1.39 | 0.264 | (Cl)(HCl) |
| BEMJAE | [20] | 1.55 | 1.55 | 0.000 | (ClHCl) |
| CERVAW | [21] | 1.56 | 1.55 | 0.002 | (ClHCl) |
| GABTIM10 | [22] | 1.65 | 1.47 | 0.114 | (ClHCl) |
| GETQIF | [23] | 1.79 | 1.38 | 0.254 | (Cl)(HCl) |
| JAVFUH | [24] | 2.32 | 1.21 | 0.627 | (Cl)(HCl)5 |
| JAVFUH | [24] | 2.35 | 1.22 | 0.630 | (Cl)(HCl)5 |
| JAVFUH | [24] | 2.35 | 1.16 | 0.674 | (Cl)(HCl)5 |
| JAVFUH | [24] | 2.41 | 1.14 | 0.719 | (Cl)(HCl)5 |
| JAVFUH | [24] | 2.50 | 1.04 | 0.823 | (Cl)(HCl)5 |
| JUJDAT | [25] | 2.13 | 1.25 | 0.518 | (Cl)(HCl)3 |
| JUJDAT | [25] | 2.17 | 1.21 | 0.570 | (Cl)(HCl)3 |
| JUJDEX | [25] | 2.23 | 1.24 | 0.568 | (Cl)(HCl)4 |
| JUJDEX | [25] | 2.26 | 1.24 | 0.580 | (Cl)(HCl)4 |
| JUJDEX | [25] | 2.33 | 1.10 | 0.713 | (Cl)(HCl)4 |
| JUJDEX | [25] | 2.41 | 1.01 | 0.820 | (Cl)(HCl)4 |
| KEGXOJ | [22] | 1.88 | 1.45 | 0.260 | Mg(Cl)(HCl) |
| KEGXOJ | [22] | 2.02 | 1.31 | 0.432 | Mg(Cl)(HCl) |
| NITSUF | [26] | 1.61 | 1.48 | 0.087 | (ClHCl) |
| OJALUG | [27] | 1.65 | 1.49 | 0.104 | (ClHCl) |
| OKAMES | [28] | 1.79 | 1.36 | 0.269 | (Cl)(HCl) |
| OROCEE | [29] | 1.59 | 1.56 | 0.024 | (ClHCl) |
| PINQOS | [30] | 1.86 | 1.35 | 0.321 | (Cl)(HCl) |
| PINQUY | [30] | 1.70 | 1.46 | 0.155 | (ClHCl) |
| PITLAG | [31] | 1.54 | 1.54 | 0.000 | (ClHCl) |
| PITLAG | [31] | 1.55 | 1.55 | 0.000 | (ClHCl) |
| PUMRUK | [32] | 1.65 | 1.47 | 0.114 | (ClHCl) |
| SANPAZ | [33] | 1.55 | 1.55 | 0.000 | (ClHCl) |
| TARFEX | [34] | 1.89 | 1.22 | 0.437 | (Cl)(HCl) |
| TIPGIJ | [35] | 1.70 | 1.36 | 0.221 | (Cl)(HCl) |
| TIYRID | [36] | 1.58 | 1.54 | 0.024 | (ClHCl) |
| UGIDAP | [37] | 2.15 | 1.16 | 0.603 | (Cl)(HCl)2 |
| VEDXUX | [38] | 2.26 | 1.18 | 0.628 | (Cl)(HCl)4 |
| VEDXUX | [38] | 2.24 | 1.12 | 0.666 | (Cl)(HCl)4 |
| VEDXUX | [38] | 2.43 | 1.11 | 0.743 | (Cl)(HCl)4 |
| VEDXUX | [38] | 2.42 | 1.06 | 0.777 | (Cl)(HCl)4 |
| YITPAS | [39] | 1.69 | 1.46 | 0.148 | (ClHCl) |
2 HX2−: (FHF)−, (F)(HF)− and HCl2−
Crystal structures BOCKEK ([3], Fig. 3) and CIHJOT ([5], Fig. 4) contain structurally different bifluoride ions, while the crystal lattice of DEHSOY10 ([6], Fig. 5) contains individual supramolecular (F)(HF)− anions.
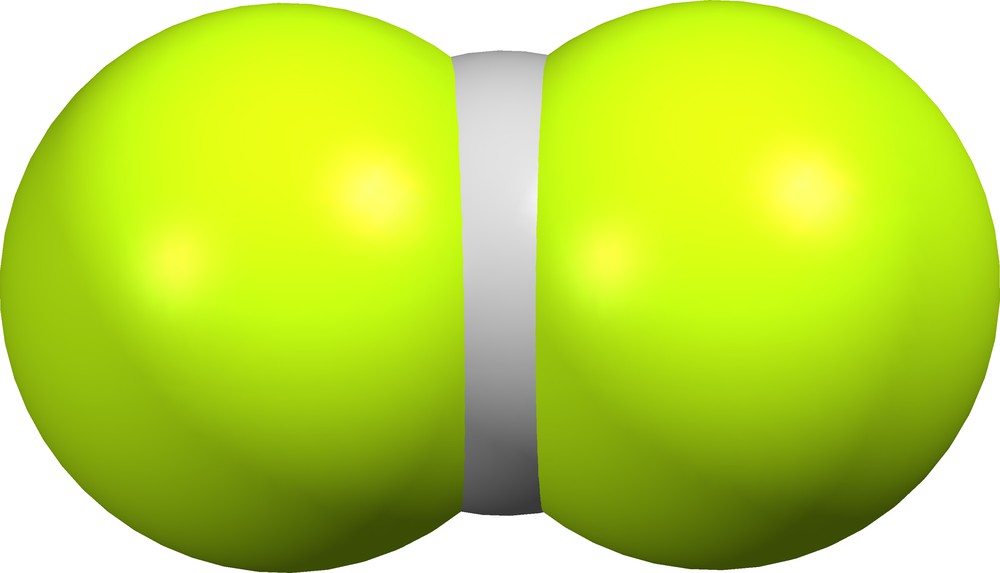
Quasi-linear bifluoride (FHF)− in BOCKEK ([3], δ = 0.036).

Bent bifluoride (FHF)− in CIHJOT ([5], δ = 0.047).
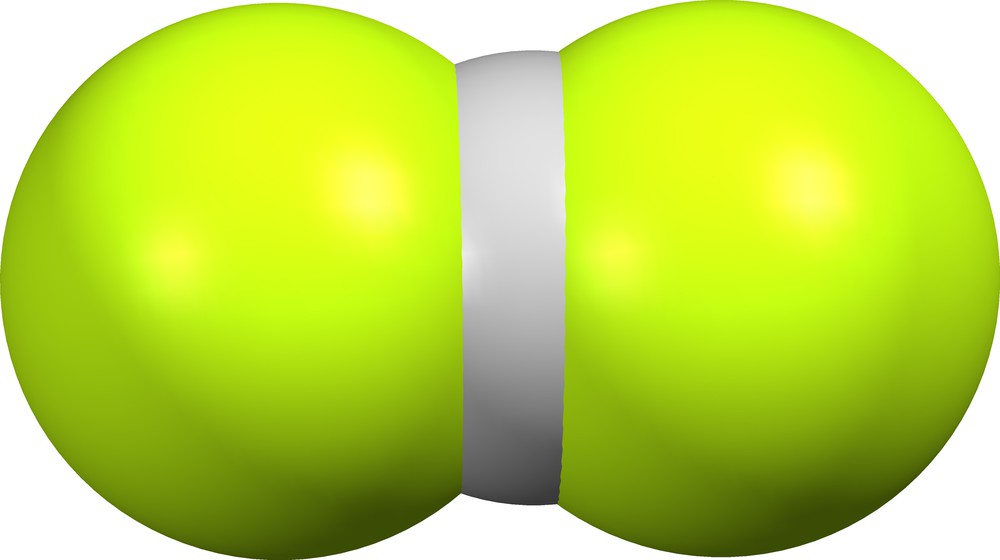
Supramolecular (F)(HF)− in DEHSOY10 ([6], δ = 0.302).
Both (FHF)− and (F)(HF)− are found as ligands in metal complexes. For example, bifluoride (δ = 0) is a bridging ligand between metal atoms in organometallic complexes CETPIB [4] and OLIYEO ([12], Fig. 6). The fluoride moiety of supramolecular (F)(HF)− also binds to metal atoms in organometallic complexes OCAZUN ([11], δ = 0.514), OLIYEO ([12], δ = 0.465), QIWPUH ([13], δ = 0.214), VEMKAZ ([14], δ = 0.427) and XAYMOB ([16], δ = 1.041).
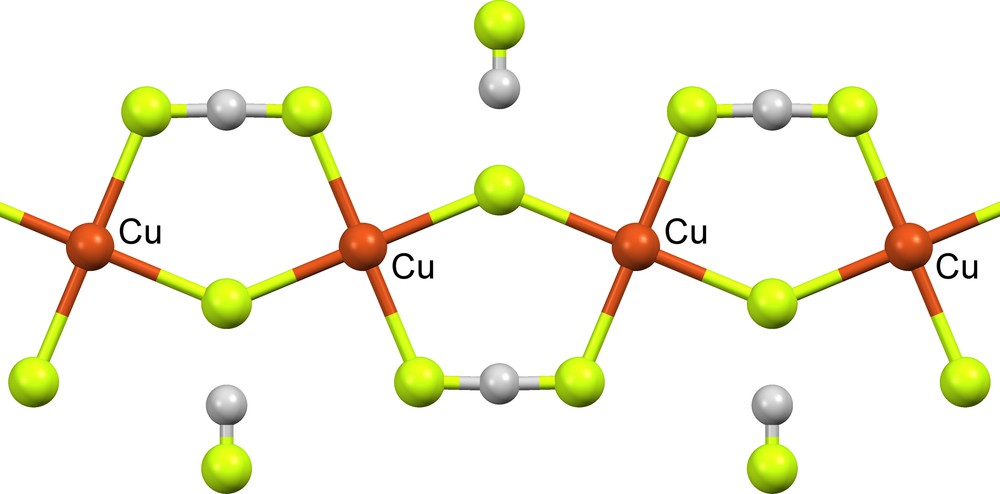
Metal ligands μ2-(FHF)− (δ = 0) and μ2-(F)(HF)− (δ = 0.465) in OLIYEO [12] (two additional organic ligands, bound to each Cu atom and perpendicular to the plane of the page, are not shown).
As noted in Table 2, HCl2− ions exist in nineteen different crystal lattices, but the δ values provide no definitive boundary between the two forms (ClHCl)− and (Cl)(HCl)−. At the extremes are the bichloride ion in AZOLUX ([18], Fig. 7) and its hydrogen-bonded counterpart in TARFEX ([34], Fig. 8).
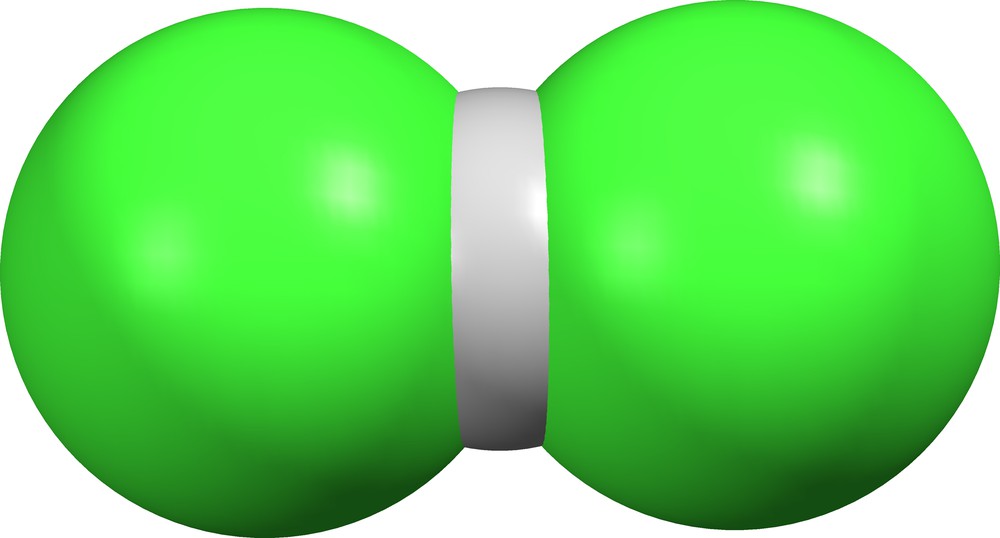
Bichloride ion (ClHCl)− in AZOLUX ([18], δ = 0).
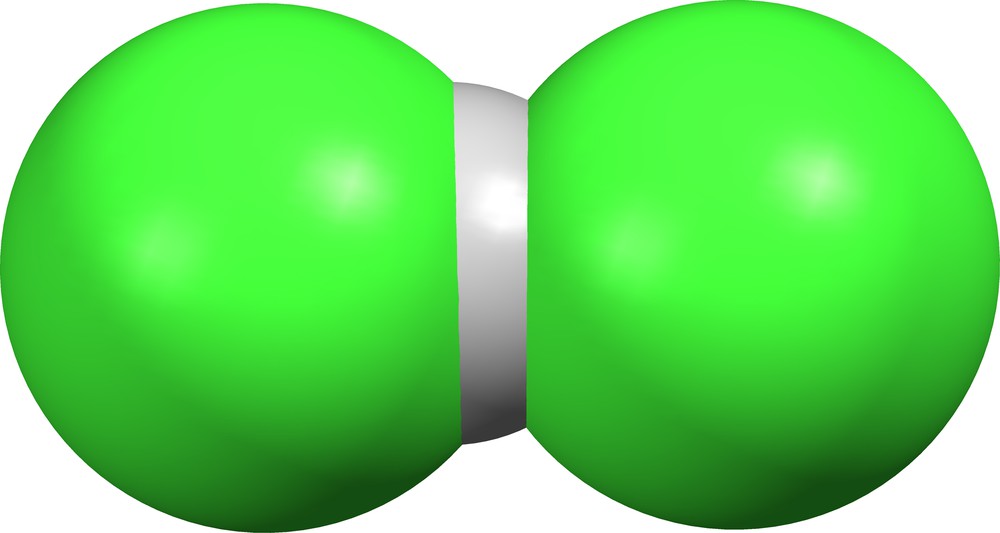
Supramolecular ion (Cl)(HCl)− in TARFEX ([34], δ = 0.437).
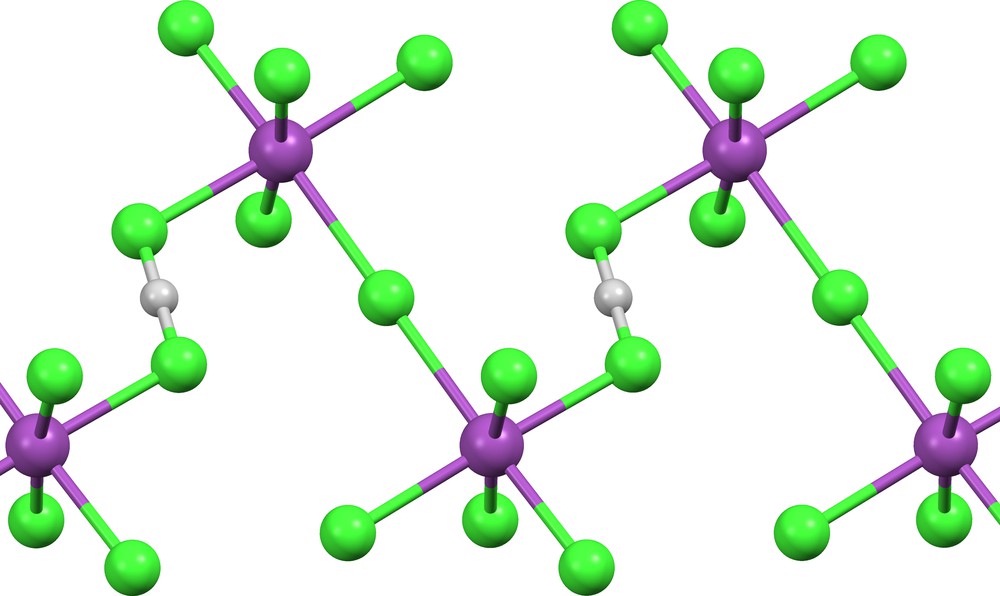
The bichloride ion is a symmetric (δ = 0) bridge between Bi atoms in ATEYEE ([17], Fig. 9), while two (Cl)(HCl)− ions (δ = 0.306 and 0.452) act as monodentate ligands in KEGXOJ ([22], Fig. 10).
Chloride and bichloride ligands bridge Bi atoms in ATEYEE [17].
(Cl)(HCl)− ligands bound to Mg in organometallic KEGXOJ [22].
3 H2X3−: (FHF)(HF)−, (F)(HF)2− and (Cl)(HCl)2−
The crystal lattice of CIHJOT [5] contains two different fluoride anions: (FHF)− as noted above, and (FHF)(HF)− with δ = 0.038 for the bifluoride moiety and δ = 0.237 for the H-bonded HF moiety (Fig. 11).
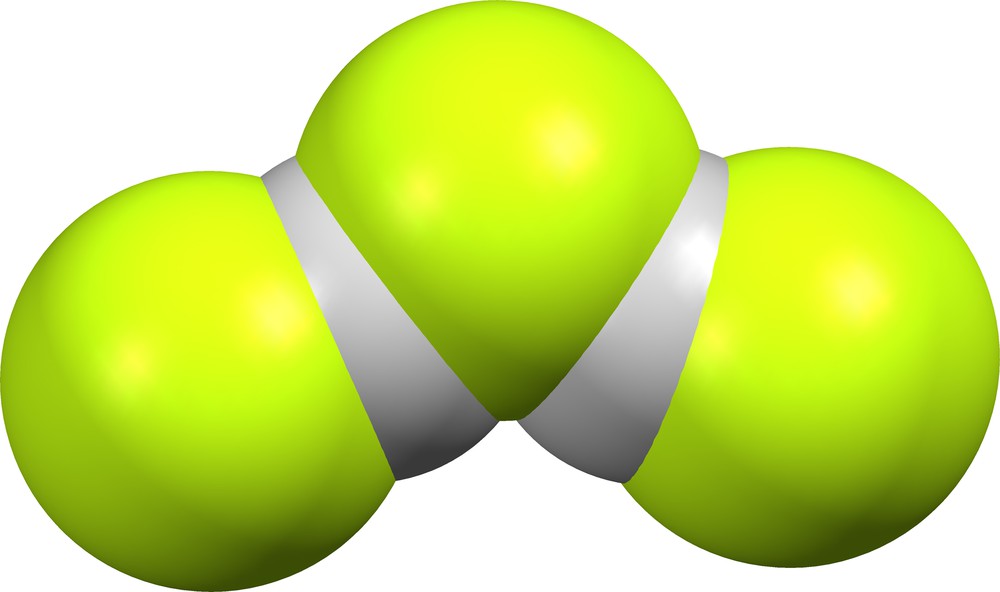
(FHF)(HF)− in CIHJOT [5].
Supramolecular anion (F)(HF)2− is contained, for example, in HITBIW ([10], Fig. 12), in which the coordination geometry of the central fluoride ion is trigonal planar with the third position occupied by a hydrogen-bonded hydroxyl ligand.

(F)(HF)2− ions in HITBIW ([10], δ = 0.211, 0.350).
The (F)(HF)2− anion takes a different form in WEGPOO ([15], Fig. 13). The central fluoride ion occupies an inversion center and its coordination geometry is linear; both positions are occupied by HF molecules and there are no other hydrogen bonds to this ion.

The (F)(HF)2− ion in WEGPOO ([15], δ = 0.302, 0.302).
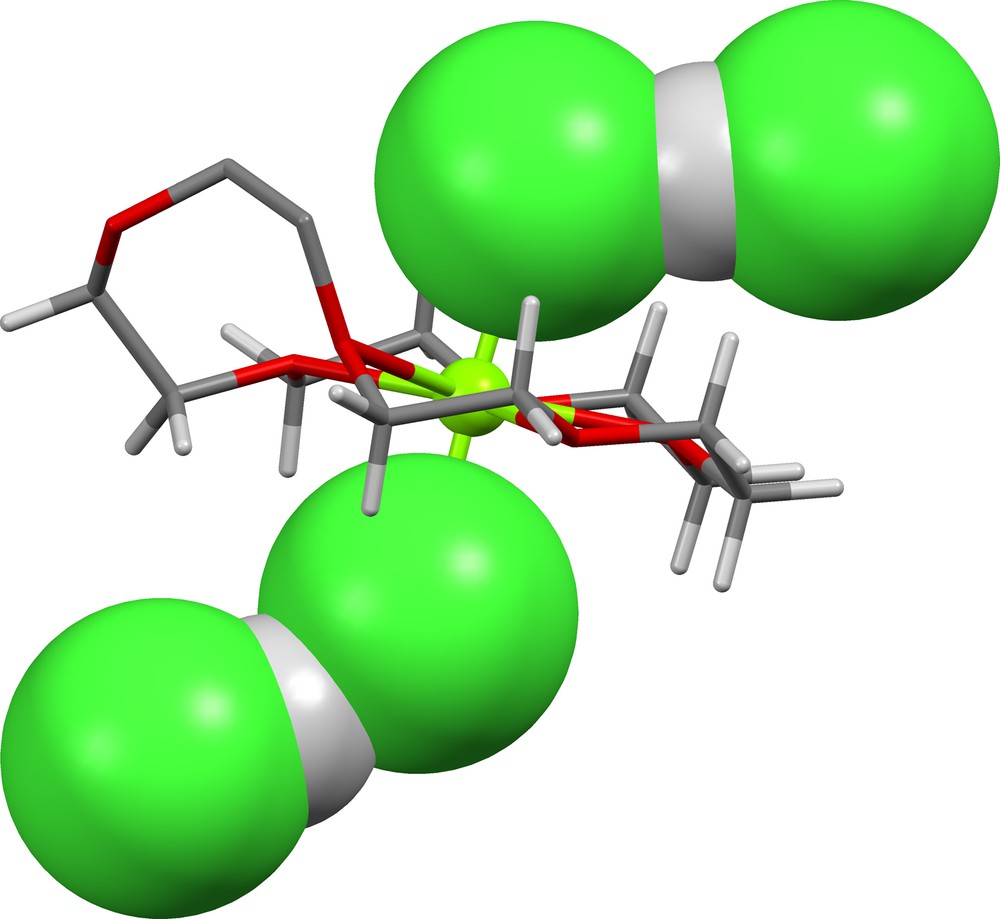
The (Cl)(HCl)2− ion found in UGIDAP ([37], Fig. 14) is bent. There is no other H-bonding in the lattice other than that between the central chloride ion and the two HCl molecules. We conclude that the shape of this ion is dictated by packing forces.
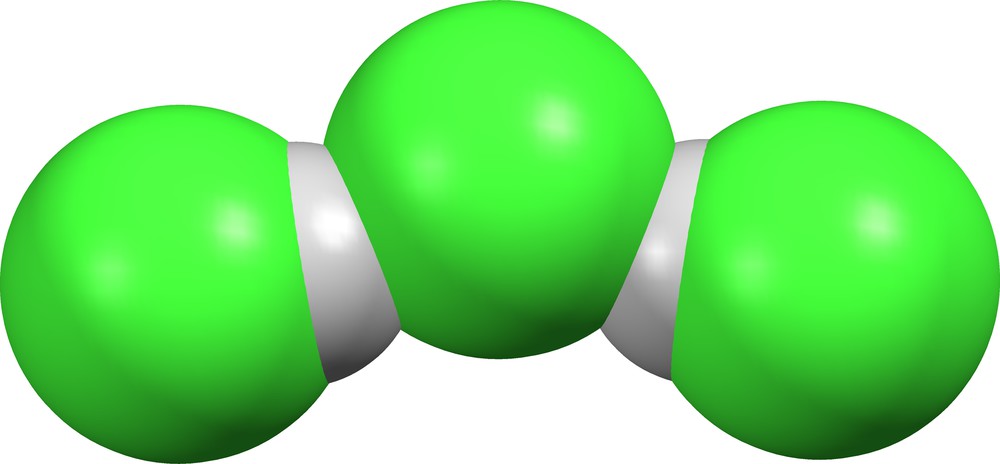
(Cl)(HCl)2− in UGIDAP ([37], δ = 0.603, 0.603).
4 H3X4−: (F)(HF)3− and (Cl)(HCl)3−
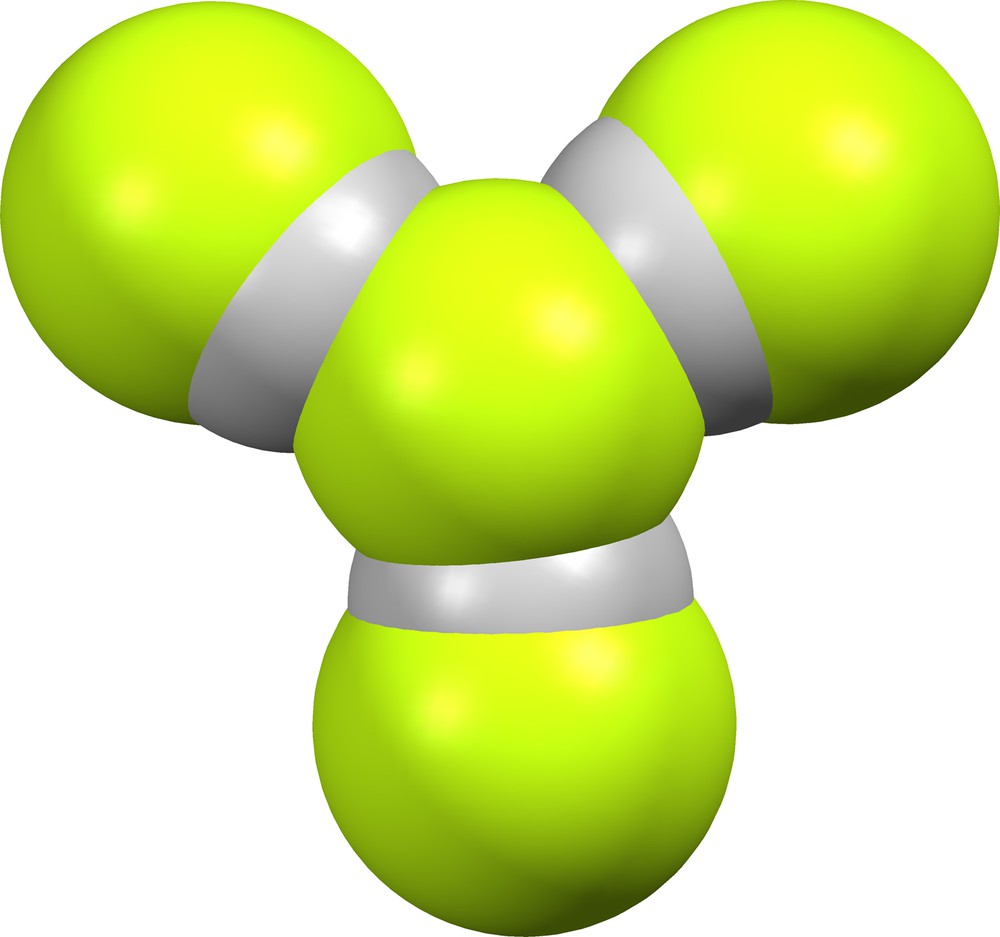
We have found no crystal structures that contain supramolecular anions (FHF)(HF)2−. Two crystal lattices contain the quasi-trigonal planar (F)(HF)3− supramolecular anion (Fig. 15): GEQBEJ ([7], δ = 0.538, 0.711, 723) and DEHTAL10 ([6], δ = 0.538, 0.538, 0.549).
The suprachloride ion (Cl)(HCl)3− is found in JUJDAT ([25], δ = 0.518, 0.518, 0.570; Fig. 16). The coordination geometry of the central chloride ion is quasi-tetrahedral with the fourth position occupied by a hydrogen bonded dimethylsulfonium cation.
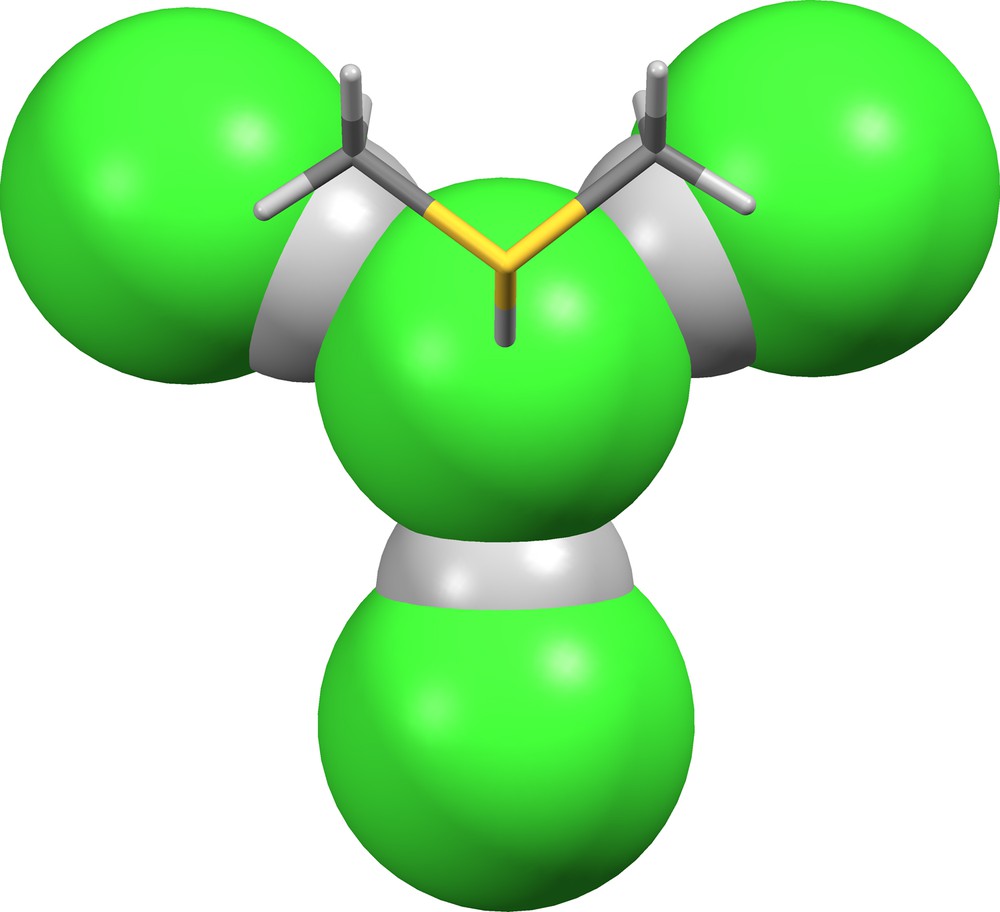
The (Cl)(HCl)3− anion in JUJDAT [25].
5 H4X5−: (FHF)(HF)3− and (Cl)(HCl)4−
We have found no crystal structures that contain (F)(HF)4−. Supramolecular anion (FHF)(HF)3− (Fig. 17) is contained in the GEPYUV [7] lattice, with δ = 0.018 for the FHF moiety and δ = 0.256, 0.354 and 0.557 for the H-bonded HF moieties.
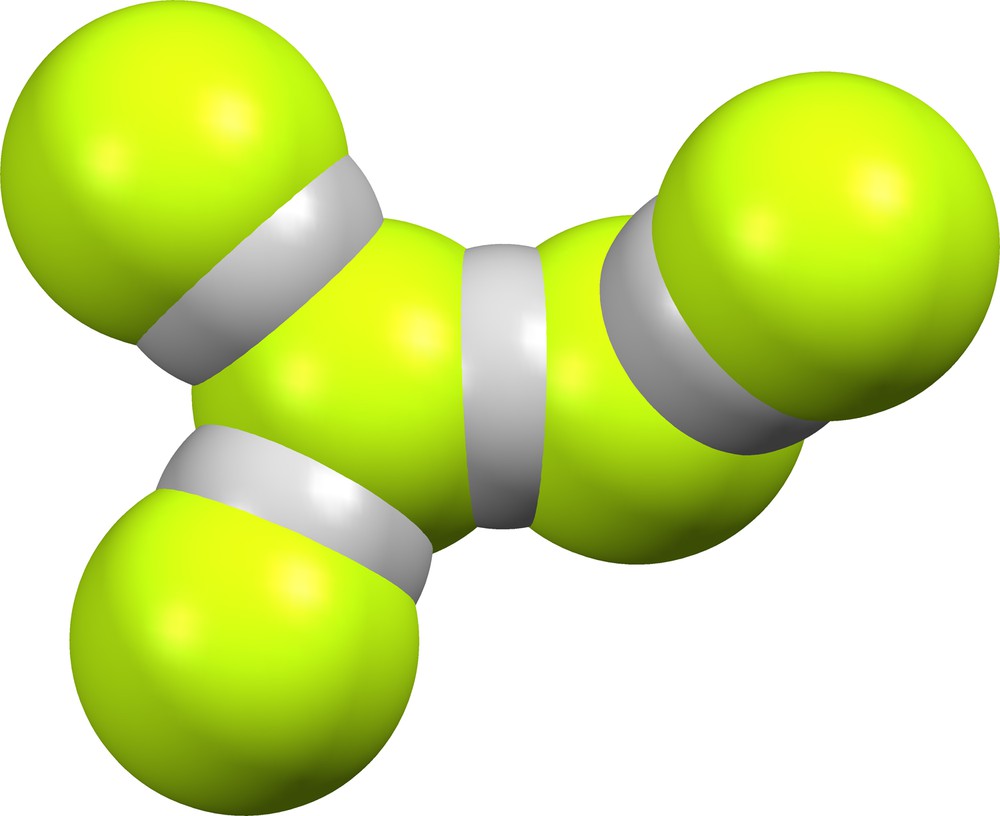
Supramolecular anion (FHF)(HF)3− in GEPYUV [7].
There are two different forms of (Cl)(HCl)4– in the CSD. In JUJDEX [25], the coordination geometry of the central chloride ion is quasi-square pyramidal (Fig. 18), with basal positions occupied by hydrogen bonded HCl molecules (δ = 0.568, 0.580, 0.713, 0.820) and the apical position occupied by a hydrogen-bonded dimethylsulfonium cation.
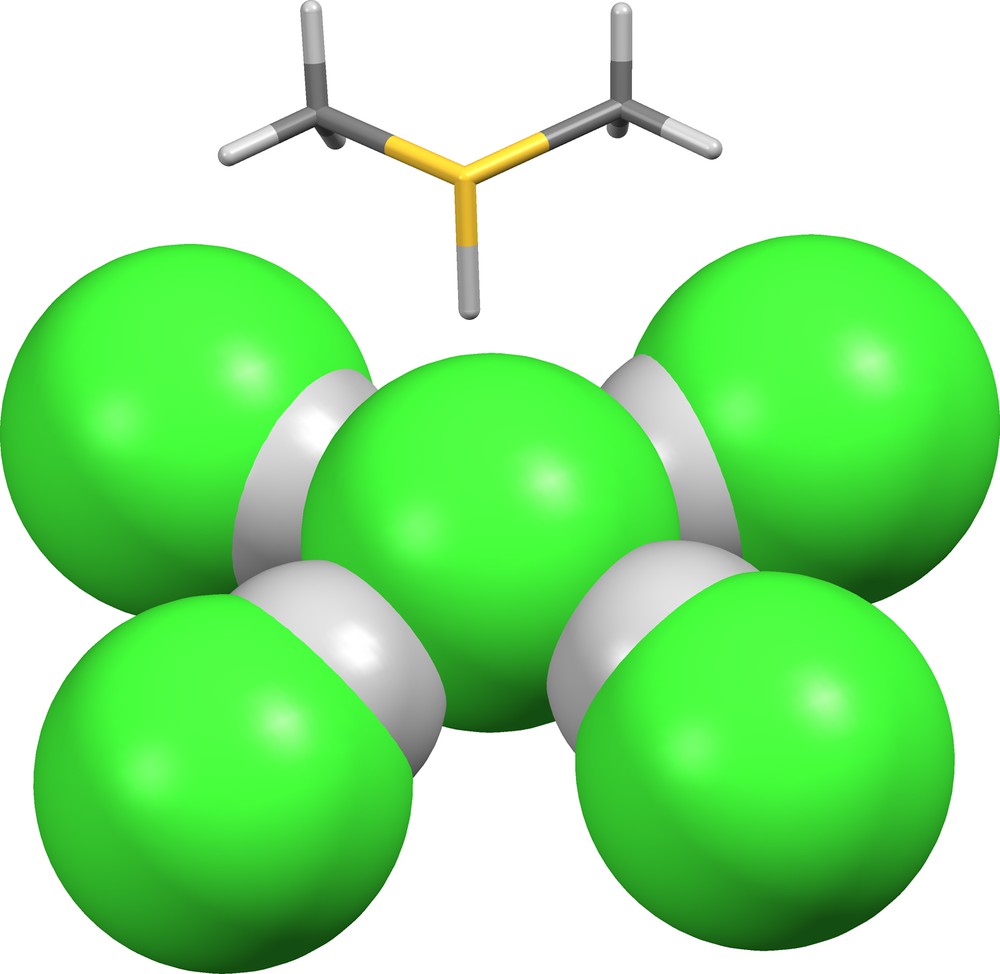
Supramolecular ion (Cl)(HCl)4− in JUJDEX [25].
In VEDXUX [38], the coordination geometry of the central chloride ion is quasi-trigonal bipyramidal (Fig. 19). Both of the axial and two of the equatorial positions are occupied by hydrogen-bonded HCl molecules (δ = 0.628, 0.666, 0.743, 0.777), while the third equatorial position is occupied by a hydrogen-bonded trimethylammonium cation.
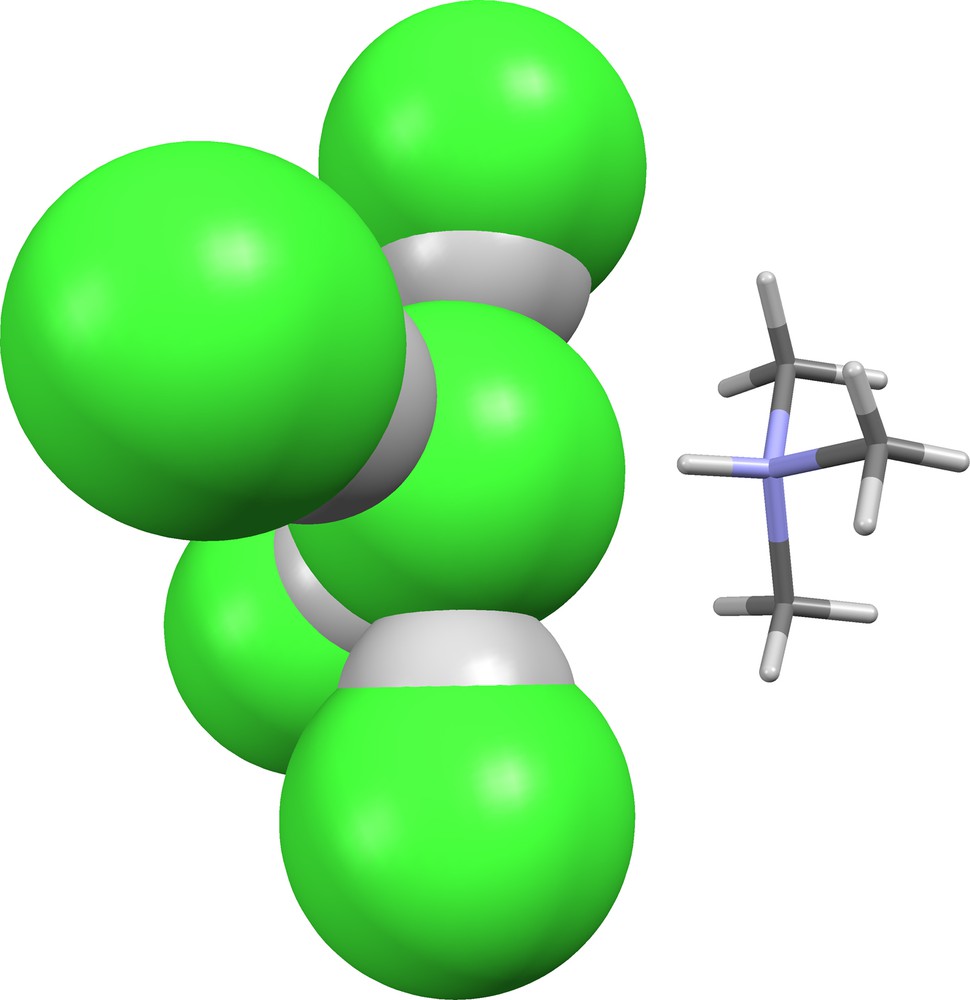
Supramolecular ion (Cl)(HCl)4− in VEDXUX [38].
6 H5X6−: (FHF)(HF)4−, (F)(HF)5− and (Cl)(HCl)5−
The lattice of GIPHAO [9] contains suprafluoride anion (FHF)(HF)4– (Fig. 20). The ion has crystal symmetry
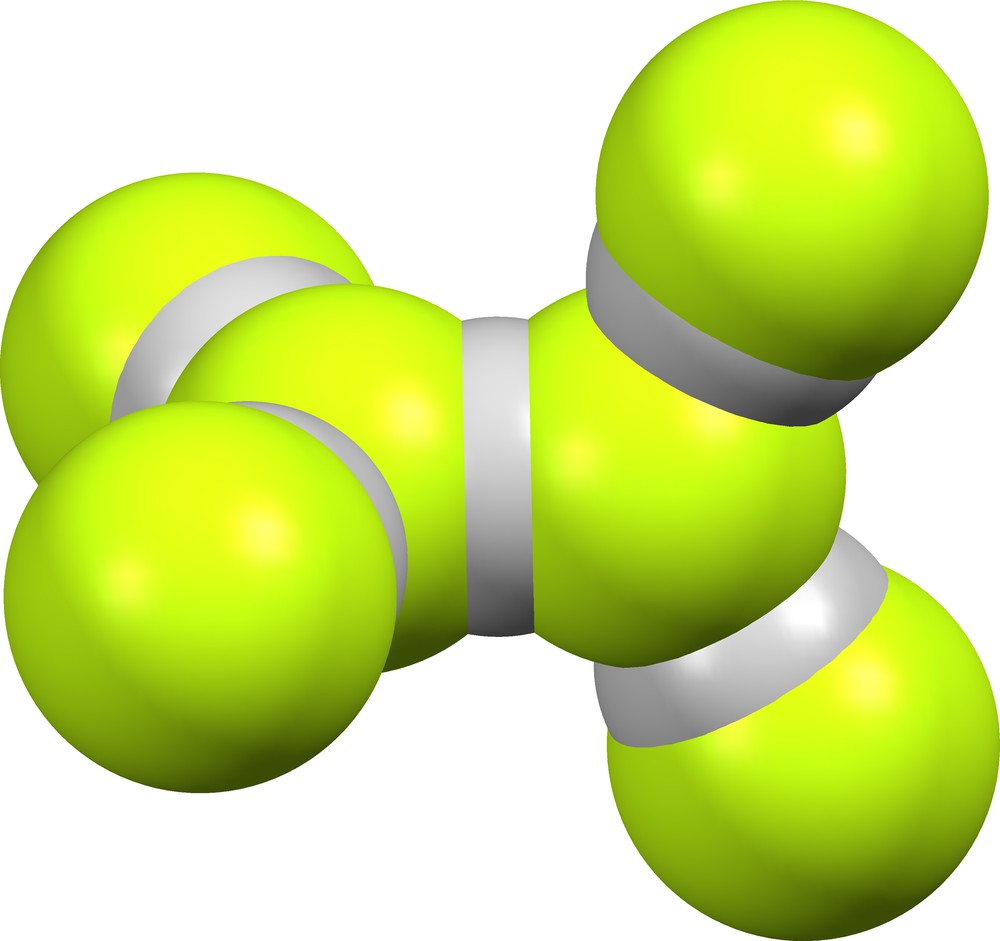
Supramolecular anion (FHF)(HF)4− in GIPHAO [9].
Supramolecular anion (F)(HF)5– (Fig. 21) is found in the lattice of GEQBIN [7], with δ values 0.484, 0.532, 0.585, 0.627 and 0.708.
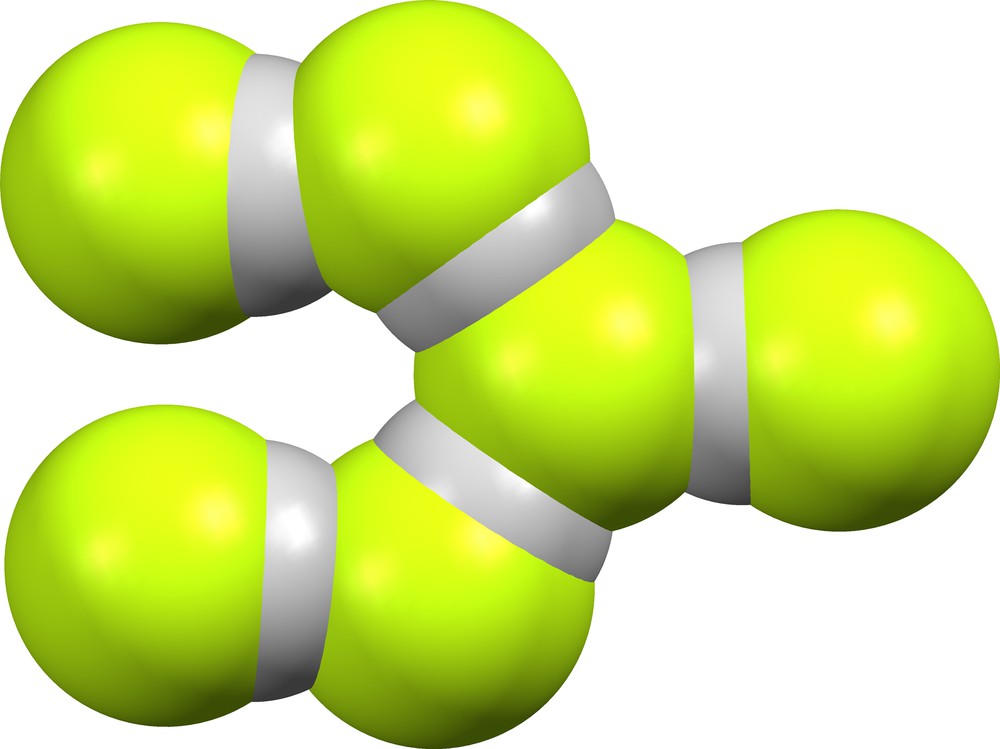
Supramolecular anion (F)(HF)5− in GEQBIN [7].
The largest suprachloride anion, (Cl)(HCl)5–, is found in JAVFUH ([24], Fig. 22). The coordination geometry of the central chloride ion is quasi-octahedral, with five positions occupied by hydrogen bonded HCl molecules (δ = 0.627, 0.630, 0.674, 0.719, 0.823), and the sixth position occupied by a hydrogen-bonded pyridinium cation.
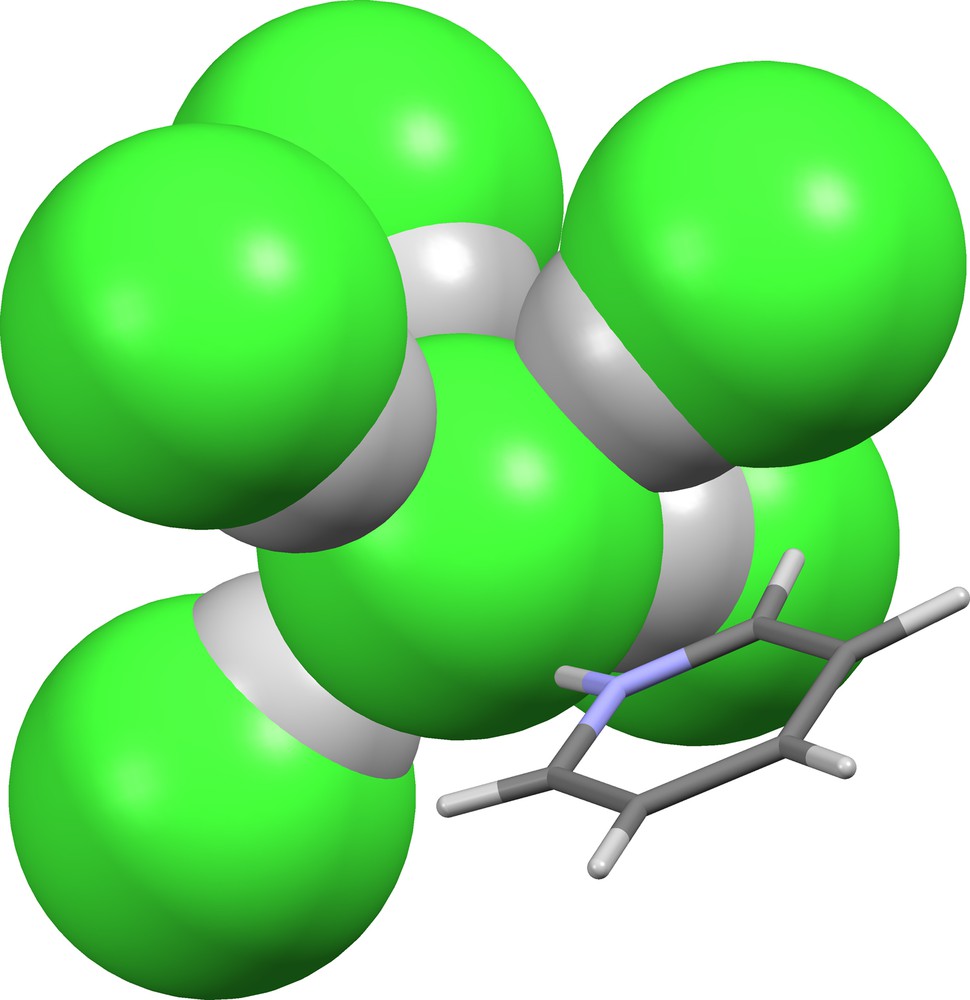
Supramolecular anion (Cl)(HCl)5− in JAVFUH [24].
7 H6X7−: (F)(HF)6−
We have found no crystal structures that contain supramolecular anions (FHF)(HF)5−. The lattice of GEPZIK [7] contains suprafluoride anion (F)(HF)6− (Fig. 23). The central fluoride ion occupies a 3-fold crystallographic axis, and each identical spiral arm has δ = 0.447 and 0.551.
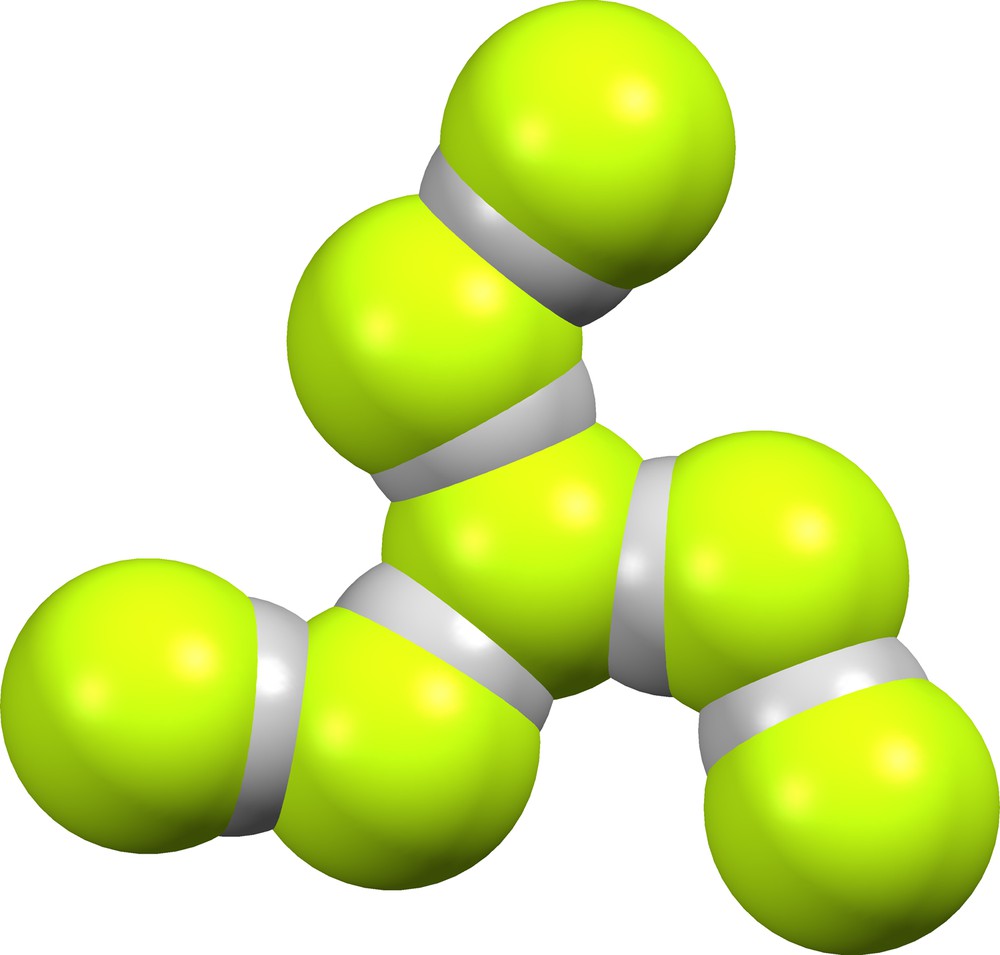
Supramolecular anion (F)(HF)6− in GEPZIK [7].
8 Conclusion
The geometric variety of suprafluoride and suprachloride anions in the solid state is astonishing. While the structural details of each are undoubtedly influenced by site symmetries, lattice packing forces, and the nature and location of other H-bonded species, we believe that the gap in δ values for HF2− represents a true demarcation between multicenter bonded and hydrogen bonded species. That there is no equivalent gap for HCl2− may be due to external forces, but we hope theoretical chemists will eventually be able to clarify this point.
During our search of the CSD, we found no suprabromide or supraiodide anions. We did find suprafluoride anions in thirteen separate crystal structures, but none of the form (F)(HF)4−, (F)(HF)n > 6−, (FHF)(HF)2− or (FHF)(HF)n > 4−. Likewise, we found suprachloride ions in twenty-six crystal structures, but did not find (Cl)(HCl)3−, (Cl)(HCl)n > 5− or (ClHCl)(HCl)n > 0−. While there may be sound theoretical reasons prohibiting certain supramolecular assemblies, these reasons are as yet unknown, and we suspect new solid-state suprahalide anions will be found in the future if crystallographers choose to look for them.
Acknowledgements
We thank Professor L. Massa (Hunter) for useful comments on the bonding issues raised in our discussions.



Vous devez vous connecter pour continuer.
S'authentifier