1 Introduction
Five-membered oligoheterocyclic compounds, such as oligthiophene, oligopyrrole, and oligofuran have raised an extensive interest in the last decade because they display fascinating semi-conductivity, photo-conductivity, and electroluminescence properties [1,2]. Intrinsic conducting polymers with conjugated double bonds are newly considered as a very important class of electroactive and photoactive materials. Some of these polymers exhibit physical properties that lend them to the development of display devices, transistors, sensors, etc. [3–5]. These applications have received a great deal of attention from both the academic and industrial research centres [6–9]. During the past decade, thiophene-based electronic materials have been extensively investigated. The ease in the chemical modification of the structures of these materials can potentially allow us to fine-tune their optical and electronic properties [10–14]. These properties strongly depend upon the degree of electronic delocalization present in such materials, effective conjugated length (ECL), and introduction of substituents at specific positions. However, a significant drawback of oligothiophenes is their low fluorescence and poor stability with respect to the oligomers consisting of substituted derivatives [15].
In order to develop a higher performance of oligothiophene, several modified oligothiophenes have been synthesized, and their electronic properties symmetrically investigated. One approach is to substitute the side-chain of the thiophene ring. Substituents at the β-position of the monomer rings prevent the undesirable α-β couplings that decrease the conjugated length and solubility of the polymers, and also the substitution plays an important role in the electrical and electro-optical properties of the polymers [16]. Conducting polymers with modified solubility in some industrially important solvents have recently been synthesized by introducing a proper substitution on the monomer ring [17,18]. It is clear that the electronic and structural properties of a substituent contribute to the delocalization of the polymer π-conjugate system, and the introduction of an electron-donating substituent at the β-position of the thiophene ring might be an interesting strategy to prevent defects, and withhold or even surpass the desirable properties of polymers [19–23].
The other approach is to synthesize the capped oligothiophene, where both ends of the thiophene chain are capped by the substituted groups [24–29]. The capped oligothiophenes have a well-defined molecular size and a high chemical stability. Recently, it has been found that phenyl groups as the end-substituents have novel light-emitting properties [24–29]. Although these features, obtained experimentally, are one of the interesting points in capped-oligoheterocyclics, the electronic structures of both neutral and doped states of the capped-oligoheterocyclics are not clearly understood due to the lack of theoretical works. However, in the last few decades, theoretical calculations have been employed to investigate different aspects related to the molecular and electronic structures of thiophene and its β-position substituted derivatives using ab initio and DFT calculations [30–37]. In our previous works [38,39], we studied a series of substituted pyrroles as potential monomers for the synthesis of conductive polymers and the corresponding oligomers with modified physical and electrical characteristics.
Theoretical investigations on a series of end-substituted oligoheterocycles are thereby desirable for designing novel functional materials, which is the main purpose of this work. In the present study, we investigated the phenyl group effect as the end-substitution on the structural and electronic properties of a series of oligothiophenes in the ground and exited forms and their doped states, as obtained with the aid of DFT calculations. To this end, we investigated characteristics of the π-conjugate system and bond length alternation pattern, HOMO–LUMO gaps, electron affinity (EA), ionization potential energy (IP), and the first excited energies of a series of PnTs including di-, tri-, tetra-, penta-, and hexamers.
2 Computational details
The ground-state geometry of each oligomer studied in this work was fully optimized using gradient procedures at the hybrid density functional B3LYP level of theory. A preliminary basis set test carried out for calculations on the electronic ground state showed that 6-31G** was the best basis set that can be used within our available hardware/software facilities within a reasonable time. Our previous studies [38,39] and reports by others [40–46] have shown that the DFT appeared notably adapted to describe oligoheterocyclics. Thus, the DFT-B3LYP level of theory was selected for the present study of PnTs using the 6-31G** basis set as implemented in the Gaussian 03 program package [47].
The optimized geometries were calculated for both the neutral and doped states of the oligomers. There was no symmetric constraint on geometry optimization. The nature of the optimized stationary point was characterized by frequency calculations at the same level of theory with the same basis set. The analysis of the obtained results showed that all the optimized structures were obtained as minima on the potential energy surface without any imaginary mode. The energies of the different states in the relevant geometries were obtained for calculating the IP values using the Koopman theorem [48]. The time-dependent density functional theory (TD-DFT) [49,50] was employed to calculate the optical properties on the basis of B3LYP/6-31G** optimized geometries. Ten lowest excitation states of each compound were computed, from which the excitation spectra were theoretically simulated with a Gaussian broadening of 0.6 eV of full-width half maximum.
3 Results and discussion
3.1 Molecular geometry
The sketch map of the structure for the hexamer type (n = 6), P6Ts, is depicted in Scheme 1, as an example of the PnTs. It is noticeable that the carbon atoms adjacent to the heteroatom in the thiophene ring α positions are branching centers in the electropolymerizationelectro-polymerization process, and thus controls the stereochemistry of the polymer chains. Also, the β carbon atoms are in appropriate positions for substitution. However, in this study, the phenyl substitution was fixed at the head and tail of an oligomer chain. It is known that the well-defined polymer structure contains almost exclusively the head-to-tail (HT) conformation, due to the α–α couplings between adjacent heterocyclic monomers in the backbone of polymer chains [38,39]. Thus, geometry optimization of PnTs was performed on the HT conformation at the B3LYP/6-31G** level of theory.
Structure and numbering scheme used for the phenyl-capped hexathiophene P6Ts.
One of the important factors related to the extent of the π-conjugation is the chain planarity, which can be reflected by the tortional angle D, as defined in Scheme 1. Dihedral angles defining the torsion between two adjacent building units along the oligomer chain for nTs, PnTs, and their ionic species were collected in Table 1. The calculations indicate that nTs and PnTs, in neutral states, are non-planar structures, in which the phenyl ring substituents are largely twisted from the plane of backbone structure of PnTs due to their steric hindrances. These angles are largely changed in the ionic states. With increase in the repeating units, there is a significant reduction in the inter-ring torsion angle at the centre of the oligomer, compared to the outside. It is interesting that the doped oligomers have less torsion angles than the corresponding neutral ones. It is known that the planarity of the polymer chains plays an important role in their electric conductivity. In general, crystalline oligothiophenes have been found to be nearly planar, as a result of more favorable crystal packing [51]. The DFT-optimized geometries are in agreement with the solid-state structure.
Torsion angles for neutral and ionic species of the PnTs at the B3LYP/6-31G** level of theory.
| Species | D1 | D2 | D3 | D4 | D5 | D′ | D″ |
| 2Ts | 21.9 | — | — | — | — | — | — |
| 3Ts | 18.1 | 18.2 | — | — | — | — | — |
| 4Ts | 17.1 | 14.7 | 17 | — | — | — | — |
| 5Ts | 16.4 | 9.8 | 9.7 | 16.3 | — | — | — |
| 6Ts | 16.4 | 10.3 | 0.3 | 10.7 | 16.6 | — | — |
| P2Ts | 13.8 | — | — | — | — | 25.3 | 25.5 |
| P3Ts | 12.8 | 12.9 | — | — | — | 25.9 | 25.8 |
| P4Ts | 15.3 | 13.9 | 15.4 | — | — | 26.4 | 25.8 |
| P5Ts | 15.5 | 12.6 | 12.7 | 15.6 | — | 25.5 | 25.5 |
| P6Ts | 15.6 | 12.5 | 12.6 | 12.5 | 15.5 | 26.3 | 25.6 |
| P2Ts(+) | 0.1 | — | — | — | — | 7.7 | 7.5 |
| P3Ts(+) | 1 | 1 | — | — | — | 16.7 | 16.6 |
| P4Ts(+) | 1.5 | 0.6 | 1.7 | — | — | 19.8 | 19.5 |
| P5Ts(+) | 2.8 | 0.5 | 0.6 | 2.9 | — | 21.2 | 20.8 |
| P6Ts(+) | 3.2 | 0.4 | 0 | 0.4 | 3.2 | 22.1 | 22.6 |
| P2Ts(–) | 0 | — | — | — | — | 0 | 0 |
| P3Ts(–) | 0 | 0 | — | — | — | 0.1 | 0.1 |
| P4Ts(–) | 0 | 0 | 0.1 | — | — | 0.1 | 0.3 |
| P5Ts(–) | 1.3 | 0.1 | 0.2 | 1.4 | — | 6.8 | 7 |
| P6Ts(–) | 2.3 | 0.1 | 0 | 0.1 | 2.4 | 11.4 | 11.3 |
Another important parameter of interest with respect to the π-conjugation is the bond alternation parameter, δ, defined here as the bond length difference between two adjacent single and double bonds. A small δ value may correspond to a good π conjugation [52]. Here, the two bond alternation parameters, δintra and δinter, are defined as:
| δintra = rββ – rαβ and δinter = rαα − rαβ | (1) |
Bond alternation parameters and quinoid coefficients, fn, for neutral and ionic species of the PnTs at B3LYP/6-31G** level of theory.
| Species | /Å | /Å | fn |
| 2Ts | 0.052 | 0.073 | 1.0441 |
| 3Ts | 0.046 | 0.069 | 1.0411 |
| 4Ts | 0.042 | 0.066 | 1.0391 |
| 5Ts | 0.04 | 0.066 | 1.0377 |
| 6Ts | 0.038 | 0.063 | 1.0367 |
| P2Ts | 0.038 | 0.066 | 1.0347 |
| P3Ts | 0.036 | 0.064 | 1.0344 |
| P4Ts | 0.035 | 0.062 | 1.0342 |
| P5Ts | 0.034 | 0.062 | 1.0339 |
| P6Ts | 0.034 | 0.061 | 1.0337 |
| P2Ts(+) | 0.021 | 0 | 0.9902 |
| P3Ts(+) | –0.013 | 0.008 | 0.997 |
| P4Ts(+) | –0.008 | 0.014 | 1.0016 |
| P5Ts(+) | –0.003 | 0.019 | 1.0052 |
| P6Ts(+) | 0 | 0.023 | 1.0081 |
| P2Ts(–) | 0.006 | 0.066 | 0.9987 |
| P3Ts(–) | 0 | 0.064 | 1.0037 |
| P4Ts(–) | 0.004 | 0.062 | 1.0074 |
| P5Ts(–) | 0.007 | 0.062 | 1.0103 |
| P6Ts(–) | 0.009 | 0.061 | 1.0127 |
Furthermore, the quinoid term refers to a structure in which the inter-ring bond has a greater double bond character than that in the standard aromatic configuration, and it is determined by the quinoid coefficient, fn [53–56]. This geometry parameter is defined as:
| (2) |
3.2 Electronic properties
It is useful to examine the highest occupied orbitals and the lowest virtual orbitals for these oligomers and polymers because the relative ordering of the occupied and virtual orbitals provides a reasonable qualitative indication of the excitation properties and the ability of electron or hole transport [57]. Since the first dipole allowed electron transitions as well as the strongest electron transitions with largest oscillator strength correspond almost exclusively to the promotion of an electron from HOMO to LUMO, we calculated the HLGs for all the studied oligomers in both the neutral and doped states. The HLG values for the neutral form are illustrated in Fig. 1 schematically. As we can see in this figure, reduction of HLGs for all oligomers becomes more obvious with increasing the conjugation length. As shown in this figure, the HLG values for the PnT species are less than those for the corresponding nT species. According to this figure, closing the end-sides of oligothiophenes by phenyl groups destabilizes both the HOMO and LUMO levels. The results obtained show an increase in the HOMO orbital level and a decrease in the LUMO orbital level after closing the end-sides of oligothiophenes by phenyl groups, and then, a reduction value for the HOMO–LUMO gap for phenyl-capped oligothiophenes.
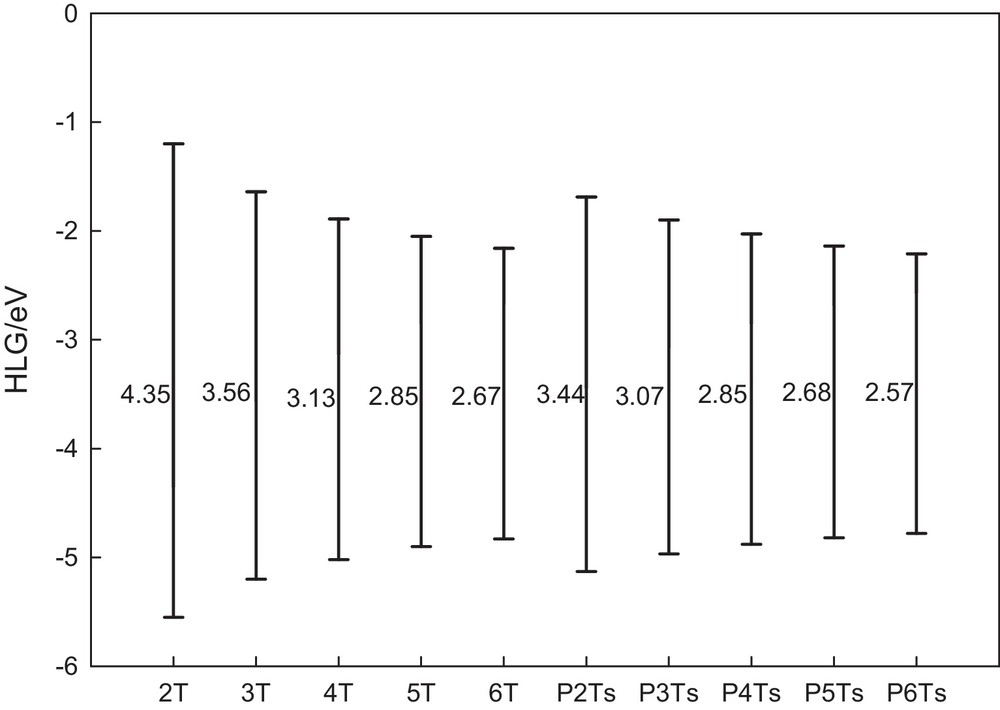
Variation in HOMO, LUMO, and HLG with the number of units (n) for PnTs and nTs, schematically.
In addition, another important tool to qualitatively judge the excitation state is the TD-DFT technique. It is clear that the low-lying excited states of the polymer, oligomer, and organic semi-conductors are correlated with the electron and hole conductivities. In particular, the first excited state is strongly correlated with these transport processes. Therefore, studying the excited states is important in the elucidation of the mechanism of the electron and hole conductivities in organic semi-conductors. Here, the excitation energies from the first to 6th excited states of each oligomer are calculated at the B3LYP/6-31G(d,p) level of theory. For instance, the simulated excitation spectra obtained using a Gaussian broadening for six lowest excitation states of hexamers are described in Fig. 2. The results obtained for the TD-DFT excitation energy (Eg) and the longest wavelength (λmax) of the maximum absorption peaks with corresponding oscillator strengths (f) of each oligomer are collected in Table 3. For all the oligothiophene derivatives, the calculated λmax values show the increasing order of dimer < trimer < tetramer < pentamer < hexamer, which is in good accordance with the corresponding reverse order of Eg values. The obtained results show that the attachment of phenyl groups to the end-sides of oligothiophenes leads to an evident bathochromic shift and increase in the intensity of the maximum absorption peak. This indicates that PnTs will have large λmax values in the solid state due to higher conjugation length and flat structures. Moreover, as expected, the simulated absorption spectra and behavior of the maximum absorption peaks are qualitatively in agreement with the results obtained in studying the HOMO and LUMO orbitals.
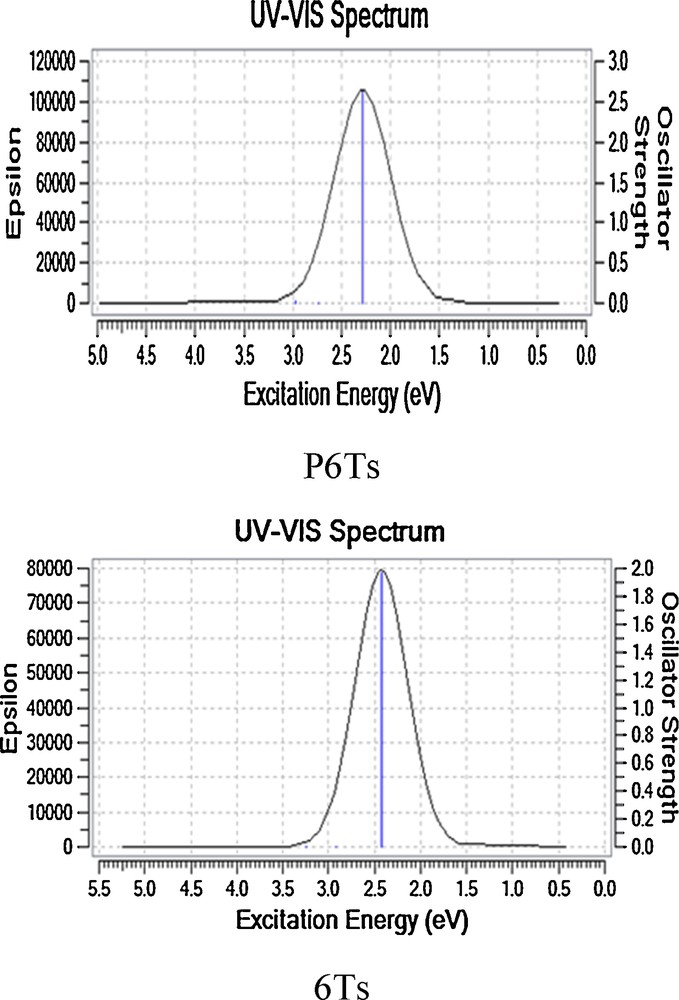
Simulated absorption spectra for both hexamers 6Ts and P6Ts calculated at the Td-B3LYP/6-31G(d,p) level of theory.
The values of λmax, Eg and oscillator strength (f) of simulated absorbtion spectra for the PnTs at B3LYP/6-31G** level of theory.
| Compound | λmax/nm | Eg/eV | f |
| 2Ts | 301.5 | 4.11 | 0.41 |
| 3Ts | 369.88 | 3.35 | 0.8 |
| 4Ts | 424.78 | 2.92 | 1.2 |
| 5Ts | 472.66 | 2.62 | 1.58 |
| 6Ts | 511.2 | 2.42 | 1.96 |
| P2Ts | 390.66 | 3.17 | 1.2 |
| P3Ts | 441.61 | 2.81 | 1.57 |
| P4Ts | 480.63 | 2.58 | 1.94 |
| P5Ts | 515.17 | 2.41 | 2.28 |
| P6Ts | 542.96 | 2.28 | 2.62 |
As mentioned in the introduction section, efficient injection and transport of both holes and electrons are important parameters for the rational design of optimized light-emitting diodes, and lead to the good performance of conductive polymers in device, such as organic light-emitting diodes (OLEDs) [52]. The ionization potential and electron affinity are closely related to the degree of electron extraction (hole injection) to form p-type semi-conductors and electron injection to form n-type semi-conductors [44]. Electronic energies for the neutral, cationic, and anionic species of the oligomers were calculated at the B3LYP/6-31G** level of theory, and used for calculation of the ionization potential (IP) and electron affinity (EA) of each oligomer, which are listed in Table 4. The results obtained for IPs and EAs show a decrease in the energy barrier for the hole and electron injection after closing the end-sides of oligothiophenes by phenyl groups, and show better p-type and n-type conducting for phenyl-capped oligothiophenes than the corresponding oligothiophenes. It was found that as the fraction of thiophene increases, the IP values decrease, which corresponds to an improvement in the hole injection properties, and this is also consistent with the trend of changes in the case of HLGs. With increase in the conjugated length, influence of the substituent type is not considerable. It is known that the formation of the intermediate radical cations from the monomers, as the first step in the polymerization mechanism of conductive polymers, has an important role in their polymerization process. This reaction can be considered as an ionization reaction [58–60]. In general, it may be concluded that the initiation step of electropolymerization of PnT species requires a lower applied potential. In other words, this potential is reduced when the electron conjugation of molecules increases. This behavior is in agreement with the lower oxidation potentials measured experimentally [60].
The values of ionization potential (IP) and electron affinity (EA) for the PnTs at B3LYP/6-31G** level of theory.
| Species | IP/eV | EA/ev |
| 2Ts | 7.11 | –0.34 |
| 3Ts | 6.49 | –0.36 |
| 4Ts | 6.14 | –0.78 |
| 5Ts | 5.9 | –1.05 |
| 6Ts | 5.74 | –1.25 |
| P2Ts | 6.28 | –0.63 |
| P3Ts | 6 | –0.94 |
| P4Ts | 5.81 | –1.15 |
| P5Ts | 5.67 | –1.31 |
The organic field-effect transistor and the organic light-emitting diode experiments show that a Schottky-type injection barrier for hoping-type carrier transport can satisfactorily describe the charge injection properties of electrode/organic interface [61,62]. Thus, for a given electrode, the hole and electron injection rates are mainly decided by the HOMO and LUMO levels. The relationship of the charge carrier injection rate between the unsubstituted and substituted molecules can be expressed as:
| (3) |
| (4) |
Comparison of the carrier injection rates for PnTs respect to corresponding nTs.
| Compound | n-type | p-type |
| 2Ts | 1.00 | 1.00 |
| 3Ts | 1.00 | 1.00 |
| 4Ts | 1.00 | 1.00 |
| 5Ts | 1.00 | 1.00 |
| 6Ts | 1.00 | 1.00 |
| P2Ts | 1.06 × 107 | 1.78 × 108 |
| P3Ts | 7.11 × 103 | 2.794 × 104 |
| P4Ts | 2.04 × 102 | 3.29 × 102 |
| P5Ts | 26.15 | 26.15 |
| P6Ts | 7.41 | 5.99 |
4 Conclusion
In this work, the effect of the end-substitution of phenyl group on the geometry, electron conjugation and electronic properties of oligothiophenes was investigated using DFT. The presence of the phenyl substituent at the ends of an oligothiophene chain exerts fine-tuning effects on the structures, electronic and excitation states. The calculations carried out on both neutral and doped phenyl-caped oligorthiophenes show that the doped oligomers have more satisfactory structural and electronic characteristics for conducting polymers. The conjugated π-system in the doped oligomers is expanded more, with a higher planarity, and the quinoid coefficient is close to unity. The obtained results reveal that with increasing the oligomer chain length, the bond alternation parameter in a conjugated system tends towards zero, corresponding to higher aromaticity. Also, the calculated energy gap values between the frontal orbitals, electron affinities, and ionization potentials indicate that with increasing the oligomer chain length, the conductivity band decreases. The absorption spectra were evaluated at the TD-B3LP/6-31G(d,p) level. Among the molecules investigated, those derivatives with a phenyl ring as the end-capped group display the largest λmax value, which is in good agreement with the results obtained for the HOMO–LUMO energies. In addition, all the PnTs investigated have better hole- and electron-transporting rates, and can act as nice ambipolar materials.
Acknowledgement
The authors wish to thank the Shahrood University of Technology for the financial support of this work.


