1 Introduction
Date palm (Phoenix dactylifera L.) is a monocotyledonous, dioecious, perennial tree that belongs to the Arecaceae family. It has a long history of cultivation and utilization in North Africa and the Middle East and is highly valued in many parts of the world. This plant has immense socio-economic, environmental and ecological values, particularly in the arid and semi-arid regions of the world. It also has several other nutritional and health benefits that have triggered a lot of biotechnological research pertaining to its propagation, improvement, and preservation.
Date palm has traditionally been propagated either through seeds or offshoots. Due to the long life cycle [1] and strong heterozygous nature of this plant species [2], seed-based propagation has often been undesirable. The vegetative cloning method of date palm has also been reported to suffer from troublesome inadequacies, particularly those pertaining to the limited number of offshoots produced during the tree's lifetime [3]. Considering the inherent limitations associated with conventional propagation, several research efforts have been made to look for practical and efficient alternative techniques.
The alternative cloning techniques were based on the in vitro multiplication of young tissues through organogenesis [4–11] and somatic embryogenesis [12–26]. These methods involved the sacrifice of the entire plant and remained, therefore, limited and insufficient. No method is therefore currently available to provide adequate solutions for the cloning and amplification of date palm while maintaining the mother plant, the source of sampling and without the risk of a definitive loss, particularly of the head clone genotypes limited only to one plant. This is because nothing guarantees the possibility of obtaining organogenic/embryogenic strains, which leads sine qua none for the effective start of the production system of vitro plants from a very limited plant material (tissue from a single bud) from a single plant.
Accordingly, the present study was undertaken to demonstrate the feasibility and efficiency of a novel in vitro culture approach based on the use of the mature female flowers. These plant materials are abundantly available every year and can, therefore, be used as cheap and potent micropropagation sources. They are also isolated easily without touching the vegetative tissue and menacing the mother plant. This is because, at the mature stage, they are fully emerged, separated from the apical bud tissues, and whose collection would not present any menace to the mother plant.
Up to date and to the best of the authors’ knowledge, no previous studies reported on plant regeneration via mature female inflorescences in date palm, nor in any other plant species. This is partly due to the high recalcitrance of this arborescent and monocotyledonous plant species during the cell proliferation process, the low reactivity of the mature tissue, the specificities of the genotype, and the relatively long phase of callus formation preceding the embryogenesis process [27]. Accordingly, this study reports, for the first time, on the induction of multiple morphogenetic capacities using a novel multiplication method based on mature female date palm flowers. The findings revealed that this method represents an efficient tool for the large-scale propagation and preservation of various high quality, and particularly rare, date palm genotypes.
2 Materials and methods
2.1 Plant material
The experimental assays in the present study were conducted on mature female inflorescences collected from nine date palm cultivars (seven located and two introduced) growing in continental and coastal regions. The different cultivars tested and their origins are presented in Table 1. DNm1, DNm2 (new date palm strains), and HR are three new, interesting and rare date palm genotypes. For each variety, there existed only one tree with no offshoots, and the possibility of their multiplication via the traditional offshoot method was, therefore, not feasible. Their cloning through the in vitro culture of vegetative tissues taken from the mother plant apex also presented the risk of losing uniquely available genotypes. DNm1 was the first new strain of Deglet Noor variety and was characterised by a large fruit size (the fruit size was bigger than the normal one by 30%); DNm2 was the second new strain and was characterised by a small seed size (1/30 of the total fruit size) compared to the normal seed size (1/10 of the total fruit size). In addition to maintaining all the characteristics of the Deglet Noor variety, the new characteristics (large fruit size and small seed size) added to DNm1 and DNm2 genotypes would secure them a higher economic position, particularly when propagated at a large scale since they met the international market requirements. The HR variety was issued from seed germination and had a good taste.
The date palm cultivars studied and their origins.
| Variety | Abbreviation | Origin | |
| Deglet Noor | DN | Degache | Tunisia |
| Mutant of Deglet Noora (large size fruit) |
DNm1 | Degache | |
| Mutant of Deglet Noora (small size seed) |
DNm2 | Degache | |
| Hamma Rgayaa | HR | Degache | |
| Menakher | Mkr | Nefzaoua | |
| Gondi | Gon | Kebili | |
| Makni | Mkn | Kerkena | |
| Barhee | Ba | Iraq | |
| Blombek | Blo | Libya |
a Variety existing only in one tree in the field and lacking offshoots.
All the female inflorescences used in this study were collected in March immediately after the total emergence of the spathe, whose removal could be done easily without inflicting undesirable impacts on the subsequent growth of the mother plant. At this state, the lengths of the spathes and their relative inflorescences ranged from 33 to 40 cm and 26 to 37 cm, respectively.
This indicated that the cultured inflorescences were totally mature and belonged to the last stage of floral ontogeny [27,28].
2.2 In vitro culture conditions
The spathe external surfaces were rinsed with 70% (v/v) ethanol, and the inflorescences were removed and surface sterilized with 0.01% (w/v) mercuric chloride (HgCl2) for 1 h. They were then rinsed three times with sterile distilled water under aseptic conditions. The inflorescence axes were sliced into 2 cm segments, each carrying two to three flowers, and were cultured by two segment groups on the callus induction media. The latter consisted of basal Murashige and Skoog (MS) [29] salts supplemented with KH2PO4 (120 mg/l), myo-inositol (100 mg/l), glycine (2 mg/l), L-glutamine (100 mg/l), adenine (25 mg/l), nicotinic acid (0.5 mg/l), HCL-pyrodoxin (0.5 mg/l), sucrose (50 g/l), and agar (8 g/l). Several 2,4-dichlorophenoxy acetic acid (2,4-D) concentrations (0.1, 1 and 10 mg/l) were assayed (Table 4). All the cultures were maintained in darkness at 28 °C for 8 to 10 months. In some cases, however, this period must be extended up to 12 months to obtain embryogenic or organogenic calli. The latter were transferred to the culture room at 26 ± 2 °C and 16 hour day-light condition, and subcultured at 3 months intervals.
Percentage of callus formation and embryogenic or organogenic callus formation on media supplemented with 2,4-D (1 mg/l) using mature female inflorescences from different date palm cultivars (number of callus obtained/total number of flowers × 100); (number of embryogenic or organogenic callus/number of callus obtained × 100).
| Variety | 2,4-D concentration (mg/l) |
Culture age (weeks) |
Total number of flowers | Callus formation (%) | Percentage of embryogenic callus | Percentage of organogenic callus | ||||
| Hydrated (H) | Compact/Nodular (N) | Total (H + N) | From hydrated | From nodular | From hydrated | From nodular | ||||
| Ba | 1 | 19 | 108 | 20.37 (22) | – | 20.37 (22) | 18.18 (4) | – | – | – |
| Gon | 1 | 16 | 340 | 8.82 (30) | 5.88 (20) | 14.7 (50) | 18 (9) | 4 (2) | 10 (5) | 2 (1) |
| DNm2 | 1 | 16 | 231 | 21.21 (49) | 12.98 (30) | 34.19 (79) | 5.06 (4) | 2.53 (2) | 5.06 (4) | – |
| DNm1 | 1 | 11 | 340 | 10.29 (35) | 6.76 (23) | 17.05 (58) | 17.24 (10) | 6.89 (4) | 6.89 (4) | 1.72 (1) |
| HR | 1 | 18 | 312 | 9.29 (29) | 7.69 (24) | 16.98 (53) | 13.2 (7) | 3.77 (2) | 7.54 (4) | 1.88 (1) |
The embryogenic calli obtained were incubated in a half strength MS liquid medium supplemented with activated charcoal (0.3 g/l) and 2,4-D (1 mg/l) [23]. They were then kept on a rotary shaker set at 120 rpm. The organogenic calli were transferred on a hormone-free medium (MS) or on a medium supplemented with 0.5 mg/l concentrations of BAP (6-benzylaminopurine) and α-naphthaleneacetic acid (NAA) to regenerate shoots which were finally transferred on a medium supplemented with 2 mg/l of Indole-3-butyric acid (IBA) for root formation.
2.3 Histological analysis
The mature female flowers were fixed in Svaloff Navashine solution (chromic acid 0.5%, glacial acetic acid 5%, formaldehyde 15%, and ethanol 5%) for 48 h. The samples were then extensively washed with running water and dehydrated in ethanol solution series (from 50 to 100%). They were subsequently immersed in xylene-ethanol baths, followed by paraffin inclusion. Several 10-μm thick serial sections were prepared using a microtome, stained with Regaud ferric hematoxylin solution (Regaud hematoxylin solution 10%, glycerol 10%) [28], and then analyzed under a light microscope.
2.4 Statistical analysis
A X2 test [30] was performed to analyze the statistical significance of the differences observed for the genotype and 2,4-D concentration effects on the percentages of callus formation. The different responses were determined once at the end of the culture period and the calculations were based on the initial number of explants.
3 Results
The findings revealed that various remarkable morphogenetic transformations, such as root and callus formations, occurred with the different types of mature flower organs. After about 2 weeks of culture, the mature flowers, particularly those cultured on media containing a high concentration (10 mg/l) of 2,4-D, were noted to undergo a slight increase in term of size. After about 8 weeks of culture, white tissue formations were observed on the explants. The nature and frequency of the tissues were noted to depend on the type and concentration of the hormone used as well as on the cultivar.
The majority of the new tissues obtained, particularly those grown on culture media containing auxins NAA at 5 mg/l or 2,4-D at 1 mg/l, were observed to proliferate into roots that appeared at the flower base and inside the floral axes (Table 2). Most of the floral axes necrosed but the flowers, even those cultured on media containing higher 2,4-D concentration (10 mg/l), remained alive for longer periods of time. The mature inflorescences grown on MS basal medium, on the other hand, did not show any reactivity and were noted to shrink after 4 weeks of treatment. This strongly suggested that the endogenous hormone levels of those inflorescences were not sufficient to sustain their growth on just a basal medium.
Effect of the hormonal composition of the media on the percentage of root induction.
| Variety | 2,4-D 1 mg/l (%) | 2,4-D 10 mg/l (%) | NAA 5 mg/l (%) |
| Gondi | 11 | 4 | 41 |
| Deglet Noor | 16.66 | 6.25 | 41.66 |
| Makni | 27 | 8.33 | 45.83 |
Callus induction was achieved after 3 to 8 months of culture at different frequencies depending on the variety, hormone concentration and subculture period.
A preliminary study was conducted on four date palm varieties (DN, Mkn, Mkr and Blo) to optimize the new method of micropropagation using mature female inflorescences presented in the current study. Considering the long reactivity intervals of the different date palm cultivars and the responses of their different organs to 2,4-D treatment, the analysis of the effect of extreme doses, namely 0.1 and 10 mg/l, in parallel to an intermediate dose (1 mg/l), which was widely employed in date palm [23], was deemed useful.
3.1 Nature of the responses obtained
Neoformations of different types were observed at various levels of the female flower, the most abundant of which was callus proliferation at the basal level of the flower (Fig. 1), notably in sepal and petal armpits (Fig. 2) which makes them more predisposed to this type of evolution than any of the other tissues of the mature flower at an entirely differentiated state.

Mature female flower showing the development of callus at its basal zone. Scale bar: 5 mm.
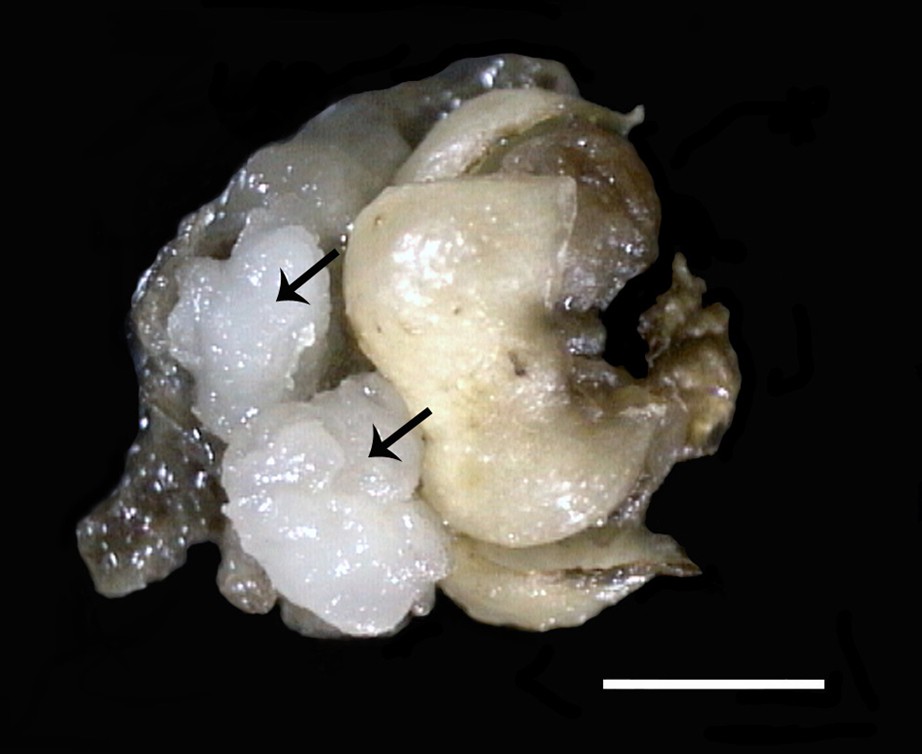
Mature female flower showing the development of two callus in the axils of sepals. Scale bar: 8 mm.
Nevertheless, some proliferations can occasionally occur at various organs of the flower (sepals, petals, carpels) to contribute to the amplification process.
In fact, the histological examination of the female flowers at their final developmental phase, where they were totally differentiated and ready for pollination (Fig. 3), revealed the presence of three welded sepals, three largely-recovered free petals, six tiny ring-shaped staminodes located at the sepal and petal armpits, and three separated ovule-harboring carpels. This analysis also showed that these flowers were at the final stages of floral development (stages 7 and 8) [27,28].

Transverse section of mature female flower of date palm variety DN stage VII revealing the presence of three sepals (S), three petals (P), six staminodes (st) and three separated carpels (C) harboring ovules (O). Scale bar: 4 mm.
The calli emerged distinctly in the armpit of one or several sepals or petals (Fig. 4) but also in the total central zone of the flower located between the carpels (Fig. 5).
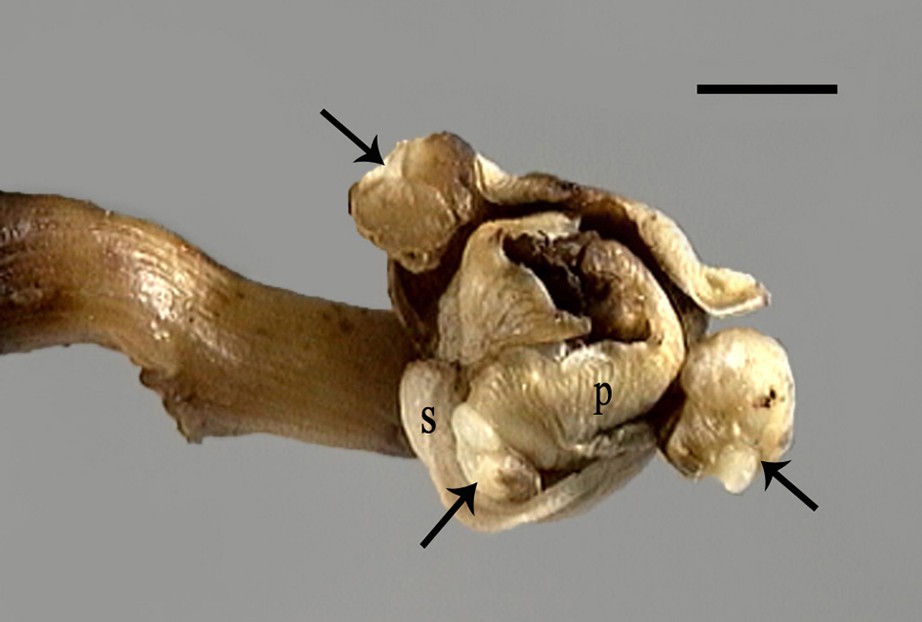
Axes having a mature female flower showing the emergence of three callus in the armpit of sepals (S). Scale bar: 6 mm.
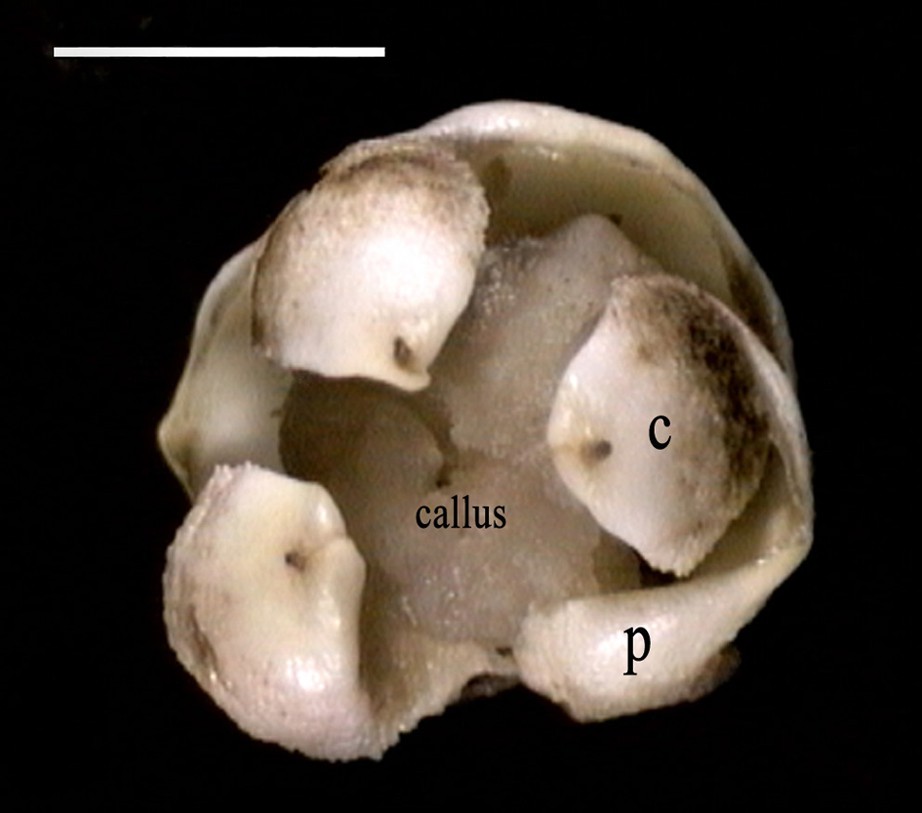
Mature female flower showing three petals (P) and the development of callus in the total central zone located between the carpels (C). Scale bar: 8 mm.
A limited number of vegetative buds or embryos also appeared early from the flower tissues without an apparent manifestation of callus structure (Fig. 6). The careful examination of these structures confirmed the transit through the callus phase with a swift neoformation of buds and/or somatic embryos from undifferentiated tissues. This type of evolution occurred more frequently on media containing low 2,4-D concentrations (0.1 mg/l) where these structures remained for a sufficiently long period of time exceeding of more than 2 months.

The development of vegetative buds (vb) within the flower tissues without the appearance of a clear callus structure. Scale bar: 8 mm.
3.2 Genotype, 2,4-dichlorophenoxy acetic acid concentration and subculture period effects on callus formation
While low 2,4-D concentration (0.1 mg/l) was noted to result in low percentages of callus formations for the DN and Mkn cultivars with no potential for plant regeneration, higher 2,4-D doses of 1 and 10 mg/l were observed to be much more efficient in the induction of high percentages of callus for most of the varieties tested, with a relatively important capacity of plant regeneration (Table 3).
Percentage of callus formation and embryogenic or organogenic callus formation on media supplemented with different 2,4-D concentrations using mature female inflorescences from different date palm cultivars (number of callus obtained/total number of flowers × 100); (number of embryogenic or organogenic callus/number of callus obtained × 100).
| Variety | 2,4-D concentration (mg/l) |
Culture age (weeks) |
Total number of flowers | Callus formation (%) | Percentage of embryogenic callus | Percentage of organogenic callus | |||||
| Hyper-hydrated | Hydrated (H) |
Compact/Nodular (N) | Total (H + N) |
From hydrated | From nodular | From hydrated | From nodular | ||||
| DN | 0.1 | 15 | 202 | 0.99 (2) | 1.48 (3) | 0.99 (2) | 2.47 (5) | – | – | – | – |
| 1 | 15 | 340 | 2.94 (10) | 5.88 (20) | 14.11 (48) | 20 (68) | 17.65 (12) | 7.35 (5) | 10.29 (7) | 2.94 (2) | |
| 10 | 13 | 680 | 5.14 (35) | 17.94 (122) | 17.05 (116) | 35 (238) | 18.48 (44) | 3.36 (8) | 12.18 (29) | 3.78 (9) | |
| Mkn | 0.1 | 16 | 240 | 0.88 (2) | 1.66 (4) | 1.25 (3) | 2.91 (7) | – | – | – | – |
| 1 | 16 | 300 | 2 (6) | 6.66 (20) | 3.33 (10) | 10 (30) | 10 (3) | – | 10 (3) | 3.33 (1) | |
| 10 | 13 | 182 | 6.04 (11) | 21.42 (39) | 16.48 (30) | 37.91 (69) | 8.69 (6) | 2.89 (2) | 23.18 (16) | 5.79 (4) | |
| Blo | 0.1 | 16 | – | – | – | – | – | – | – | – | – |
| 1 | 16 | 268 | 1.49 (4) | 5.59 (15) | 9.32 (25) | 14.92 (40) | 15 (6) | 2.5 (1) | 5 (2) | – | |
| 10 | 16 | 360 | 2.77 (10) | 9.72 (35) | 11.38 (41) | 21.11 (76) | 27.63 (21) | 5.26 (4) | 7.89 (6) | 1.31 (1) | |
| MKr | 0.1 | – | – | – | – | – | – | – | – | – | – |
| 1 | 32 | 300 | 0.33 (1) | 1 (3) | 0.66 (2) | 1.66 (5) | – | – | – | – | |
| 10 | 32 | 216 | 1.85 (4) | 5.55 (12) | 5.09 (11) | 10.64 (23) | 8.69 (2) | – | – | – |
In fact, when compared to the percentages obtained for callus formation using the medium supplemented with 2,4-D at a concentration of 1 mg/l, those obtained with 2,4-D at 10 mg/l were noted to be significantly higher at P < 0.0001 for the DN and Mkn varieties and at P < 0.001 for the Blo and Mkr varieties. Those concentrations were also noted to exert similarly significant effects in terms of the differences observed for the percentages of embryogenic/organogenic callus formations, which were statistically significant at P < 0.0001 for DN and at P < 0.0004 for Mkn and Blo varieties.
In addition to the effects attributed to 2,4-D concentration, a second factor that was equally implied in tissue response related to the source plant genotype of the inflorescences (Tables 3 and 4). In fact, several differences in terms of response were observed between the cultures maintained in the same conditions for the DNS, Ba, DN, HR, Blo, Gon, DNG and Mkn cultivars cultivated on MS + 2,4-D (1 mg/l) and the ones for the Mkn, Blo, DN and Mkr cultivars cultured on MS + 2,4-D (10 mg/l). A X2 contingency test revealed that these differences were statistically significant (P < 0.001).
The data presented in Table 3 showed that for the initiation phase, the culture of explants on media containing 2,4-D at a concentration of 10 mg/l had a beneficial effect for it enhanced the percentages of induced calli and their future acquisition of new embryogenic or organogenic capacities. Several varieties were noted to show considerable flexibility even at 1 mg/l of 2,4-D, which was the case for the DN and Mkn varieties whose floral organs revealed high potencies that had never been reported before in the literature, namely the appearance of callus between the carpels and at petal axils, the transformation of the carpel into callus, and even the direct induction of the shoot.
Other varieties, such as Mkr, did not show any multiplication response only when 2,4-D was used at a concentration of 10 mg/l. The recalcitrance of this cultivar was confirmed by its very low reactivity and need for a longer period of culture on a 2,4-D rich medium (10 mg/l) (Table 3). In fact, Mkr was the sole variety among all the other varieties tested that did not show any reactivity to 2,4-D at 1 mg/l. Moreover, when 2,4-D was used at 10 mg/l, the Mkr cultivar showed the lowest percentage of callus formation of only 10.64 against the 35, 37.91 and 21.11% for the DN, Mkn, and Blo varieties, respectively (Table 3). The callus initiation process underwent by this variety was also observed to be very late (after 6 month) and to extend over a very long period (10 months to 1.5 years).
Once controlled, the multiplication technique based on mature female flowers was eventually selected for further application as a multiplication strategy and to avoid problems related to somaclonal variation, the medium for callus induction was used with 2,4-D at a concentration 1 mg/l (Table 4). In fact, 2,4-D at a concentration of 0.1 mg/l proved inefficient in terms of plant regeneration. At a concentration of 0.5 mg/l, on the other hand, it induced the formation of new callus and plants at relatively low rates when compared to those attained at 1 and 10 mg/l concentrations, especially for the DN and Mkn cultivars.
The findings demonstrated that maintaining the mature flowers on the same culture medium for a long period (3 months) before their transfer to another one, was beneficial and sufficient to achieve good new tissue evolutions specifically before the first subculture (Table 5). While this process was observed to limit the necrotic tissue rates, it was later noted to ensure a better evolution for the derived callus; otherwise the explants were noted to shrivel. In fact, previous research indicated that this period must be shortened only when the explants produce high amounts of phenolic compound oxidants that are highly toxic to the plant tissues [31,32].
Effect of the subculture period on the percentage of callus induction for DN variety during 12 months of culture. Experiments were performed in triplicate and were submitted to the Student's t-test which was used to indicate significant differences (P < 0.001).
| Number of subculture cycles | Callus percentage (%) |
| 3 (every 4 months) | 75.77 ± 1.17 |
| 4 (every 3 months) | 78.16 ± 1.16ns |
| 6 (every 2 months) | 55.70 ± 0.88a |
| 12 (every 1 month) | 31.83 ± 0.92a |
a These values are significantly different according to the Student's t-test (P < 0.001).
3.3 The different types of calli obtained and their subsequent development
The first key factor of the technique presented in the current study, which is based on completely differentiated flowers, related to the attainment of the callus. In fact, the return to the meristematic level is a highly sophisticated process that depends heavily on a complex of various physico-chemical factors, among which the hormones used are particularly important. In this context, the 2,4-D hormone has often been described as a potent auxin that has the ability to stimulate differentiated tissues to reach meristematic status without causing tissue damage or mutations and that should be used at relatively high concentrations [14,33].
3.3.1 Hyper-hydrated calli
Hydrated calli refer to non-nodular calli that have irregular borders and that are already characterized by good proliferation capacities. Based on their hydration degrees, two types of calli could be distinguished. The hyper-hydrated calli, which were strongly translucent (water content: 90.12%), were noted to undergo necrosis with whatever type of treatment being performed. The formation rates of this type of calli were constantly very low and hardly did they exceed minute percentages (2-10%) (Table 3). Except for the hyper-hydrated calli that were formed at the basal limit of the flower, which is the attachment site to the pedicel in a direct contact to the medium, the other types of calli, including the nodular one, were noted to originate from all floral organs.
3.3.2 Hydrated calli
This type of calli consisted of hydrated and partially separated cell groups that started to appear at various rates 2 to 3 months after the first culture, which was attributable to the 2,4-D hormone effect. The percentage generated for this type of calli revealed that it was the most abundant type among all the cultivars investigated, except for the DN and Blo varieties (42–62%) (Fig. 7a) (Tables 3 and 4). These calli showed normal growth (water content: 87.63%) and were able to regenerate plants.
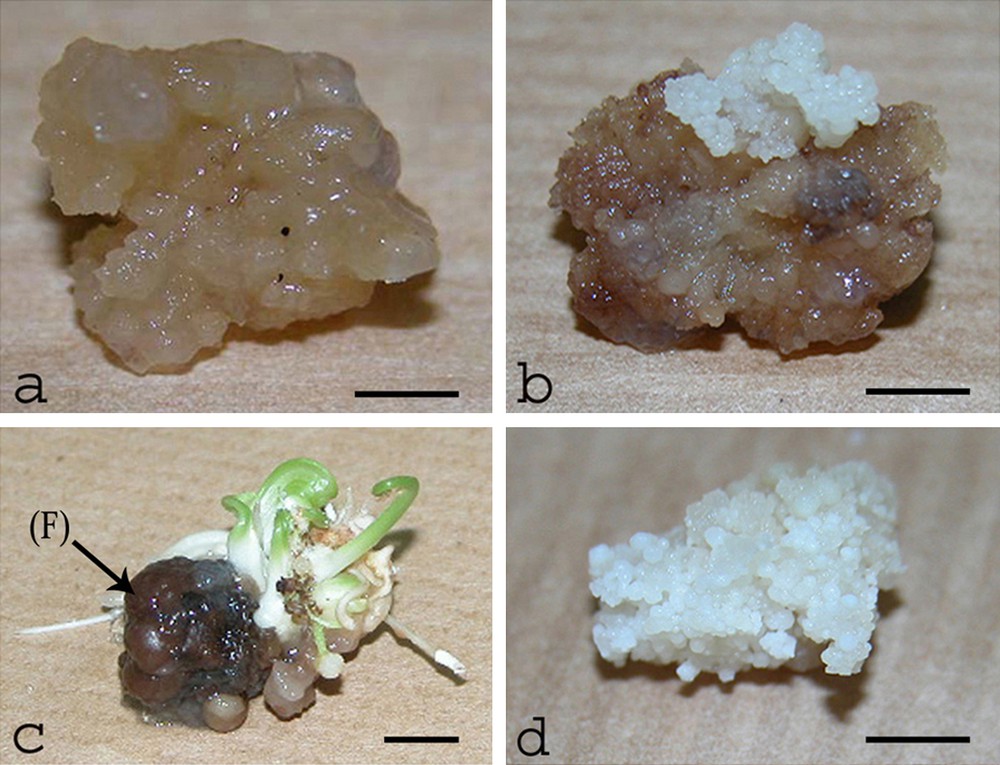
Aspects of a hydrated callus (a), the evolution of small parts of the hydrated calli to be differentiated into embryogenic calli (b), the evolution of the hydrated calli located at the base of flower (F) into white tissues (c), embryogenic callus (d). Scale bar: (a, b, c) 4 mm; (d) 5 mm.
Moreover, hydrated calli were noted to grow all over the tissues of mature flowers, especially at their basal tissues, and to consist of separated, white, and soft fragments that were characterized by their abilities to grow for a long period. Their transfer onto completely 2,4-D free media caused necrosis during the subsequent 2 or 3 months of culture. When cultured for a long period (more than three months) on media containing a low 2,4-D dose (0.1 mg/l), however, these hydrated calli were able to grow and develop to produce either somatic embryos or shoots. The somatic embryos appeared after small parts of the hydrated calli evolved and acquired new abilities to be differentiated into embryogenic calli (Figs. 7b and d). The latter were later transferred into a liquid medium. After 2 to 3 subcultures in a medium containing 2,4-D at 0.1 mg/l, the organo-embryogenic culture and the white tissues (Fig. 7c) were transferred to media that were either hormone-free or supplemented with BAP and NAA at 0.5 mg/l concentration each, and were exposed to light. These tissues could, accordingly, grow and develop organogenic tissues.
3.3.3 Nodular calli
This type of calli consisted of nodules of different sizes (1–4 mm), usually yellowish (Fig. 8A) and attached or separated by undefined borders. They started to appear at the third or fourth month of culture and characterized by regular surfaces and hard consistencies. They were noted to develop within the petal tissues and around the point at which the flowers and their floral axes were joined. These calli showed slow growth rates that were maintained throughout the subsequent cultures, under the same conditions or with changes in hormone level or culture period. A few percentages of this type of calli were noted to differentiate a vegetative bud (organogenic tissues) (Fig. 8B) or small granules on their surfaces. These granules later formed embryogenic callus (Fig. 8C) (Tables 3 and 4). In some cultivars (Gon and Mkn), the nodules were able to proliferate and develop into stems within about 3 months of culture, which was the case for more than 6% of all the flowers cultured on appropriate culture media (MS basal medium supplemented with BAP and NAA at 1 mg/l concentration each).
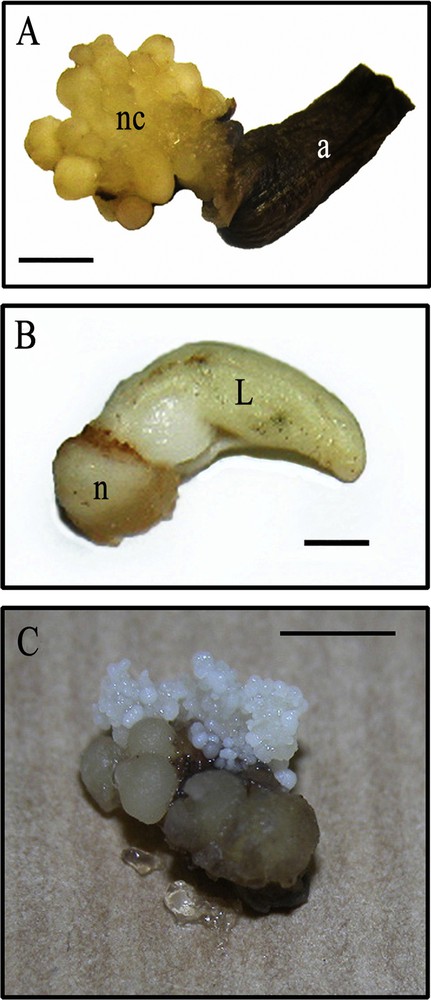
Axis having a flower totally transformed into nodular callus (A), the differentiation of vegetative bud from nodular calli (B), the evolution and differentiation of small parts of nodular calli into embryogenic calli (C). (a) axis, (nc) nodular calli; (n) nodule and (L) first leaf. Scale bar: (A) 5 mm; (B) 1 mm; (C) 4 mm.
Moreover, among the organogenic callus obtained, a quantity at the rate of 400 mg to 600 mg was transferred under 16-h photoperiod for 6 to 8 weeks, to achieve shoot regeneration. On average, five to eight regenerating shoots were scored per callus. Concerning the regeneration of plantlets from the embryogenic callus we were followed the protocol optimized in previous study [23].
The flower carpels, on the other hand, were rarely responsive. Overall, they were noted to follow one of two developmental patterns. In the first pattern, the carpels underwent an increase in size (5–10 times) but remained regular and hard. They did not show signs of development towards organogenic growth or callogenesis processes and remained at this state for a long period before an eventual necrosis. In the second developmental pattern, unlike the other flower parts, the carpels were noted to show an exceptionally significant increase in size. They also underwent a clear delay followed by a development of small granules at their external surface, which occasionally led to the formation of tissues that were able to develop into embryogenic calli (Fig. 9). This distinctive type of evolution followed by the carpel was observed starting from the 6th month using Mkn, Gon, and DNm2 varieties at a percentage of 1.64, 5.29 and 7.35, respectively. The results also indicated that the compact large-size nodules usually formed roots.

The formation of small granules proembryogenic at the external surface of the three carpels. Scale bar: 6 mm.
3.4 Histological analysis of calli origins
Longitudinal and transverse section analyses, carried out on flowers taken at different levels of callus formation process, revealed that the petals (Fig. 10) and flower bases (Fig. 11) were the most reactive organs, showing numerous granules in the tissues especially around the nutrition vessels. Those tissues were those that would later develop the abovementioned types of calli. The results indicated that different growth forms would later lead to the formation of shoots or somatic embryos, were discerned at the sepal and petal armpits.
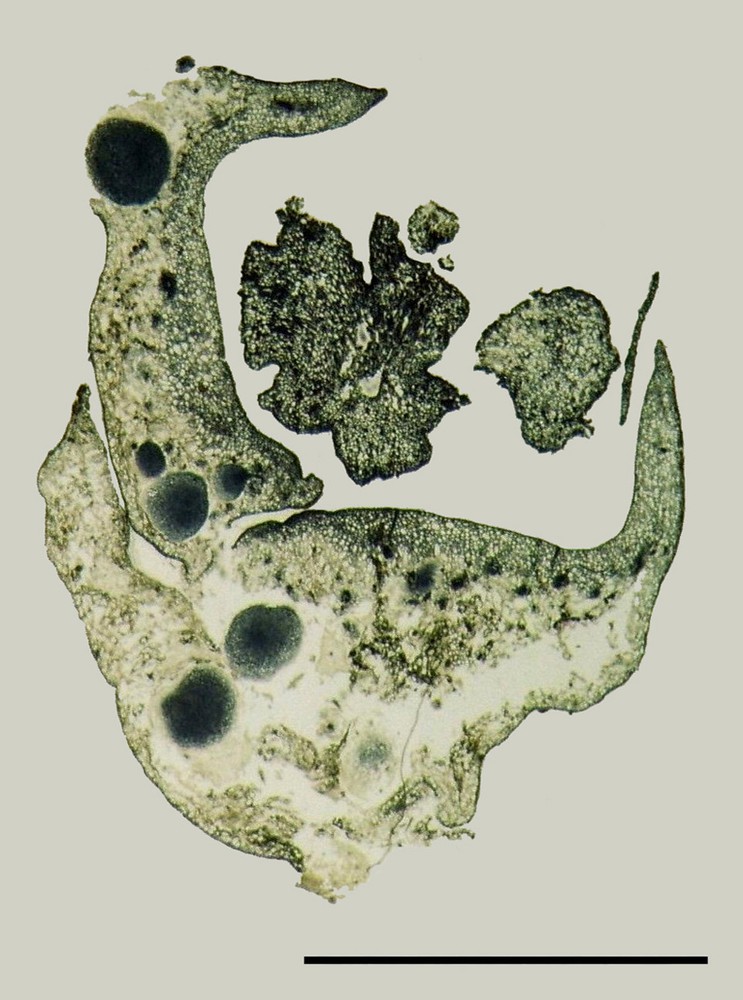
Longitudinal section of mature female flower of date palm showing the presence of a nodular calli in the petals (P). Scale bar: 3 mm.
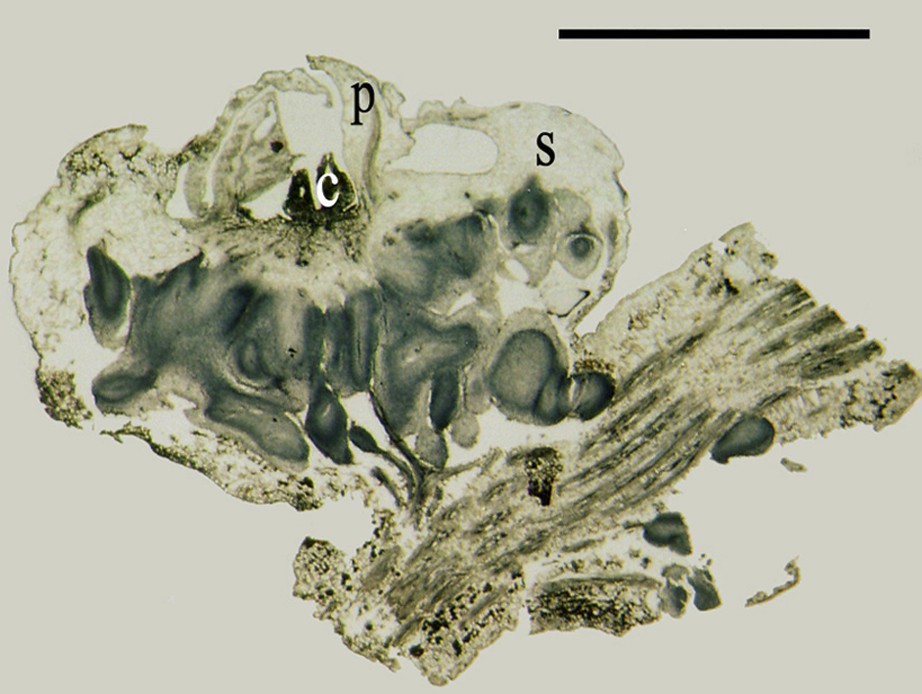
Longitudinal section of a mature female flower of date palm attached at their axe showing the presence of sepals (S), petals (P), carpels (C) and initiates a callus at the base of the flower. Scale bar: 4 mm.
4 Discussion
The present study was undertaken with the aim of investigating the feasibility and possible gain benefits of using mature female flowers for the micropropagation of date palm so as to contribute to the continuous search for alternative techniques that would help overcome some of the troublesome inadequacies associated with the conventional and currently employed ones. The findings indicated that date palm propagation using mature female flowers is both possible and promising. The success of this technique depend on the synergistic interaction of different basic factors particularly those pertaining to the hormonal composition of the medium, the physiological state of flowers at the time of culture, the physico-chemical conditions of the subculture process, and the genetic characteristics specific to each cultivar.
In fact, and in accordance with previous studies, remarkable differences were well established in terms of the time required and percentages obtained for embryogenic callus formation with mature and immature inflorescences [23,27]. Similar differences were found when comparing the response of these mature inflorescences with juvenile leaves already studied [23,25]. As far as callus induction is concerned, the process was previously reported to require a longer initiation time in monocots [34]. Several previous studies have also reported that callus induction was difficult to achieve and that the proliferation of initiated calli was very slow and difficult to maintain for a number of alliums species [35,36].
The findings presented in the current study revealed that the first culture phase of the mature female flowers must be performed in the dark so as to increase their morphogenetic potential and decrease polyphenol secretion since exposure to light would promote the synthesis of chlorophyll and impede any possibility for proliferation, which corroborates previously reported results [8,37] using vegetative and floral tissues of date palm, respectively.
Furthermore, all of the date palm varieties examined in this work were noted to show reactivity to 2,4-D when used at an average concentration of 1 mg/l, except for the Mkr variety, which was characterized by a high recalcitrance requiring a high 2,4-D concentration of up to 10 mg/l. These results are in line with those previously reported in an earlier study [25] where juvenile leaves from the Mkr variety were used as initiation explants.
It is worth noting in this context that the literature presents ample evidence that the control of the propagation process using very low 2,4-D concentrations (0.1 mg/l) has become feasible for young flowers or leaves [38] of some varieties, thus limiting the risk of somaclonal variation. Several studies have also reported that the differentiation degree of the flowers, such as those of the sugarcane, is an important factor defining their ability to develop [39].
As far as callus development issued from the flower primordia, the findings indicated that it started since the fourth week of culture while the mature flowers required at least 8 weeks to start this process, which confirmed that mature flowers needed longer time and higher 2,4-D concentrations to acquire the meristematic characteristics than young flowers.
The percentages of tissues producing shoots and somatic embryos generated from mature, as well as young, female flowers were relatively high. Accordingly, this technique can be considered promising for it represents an optimal alternative to overcome the inadequacies associated with current micropropagation technique, and possibly, a unique solution for the propagation of rare and offshoot-lacking date palm varieties. However, the question that remained to be determined was where this high reactivity of female date palm flowers resided, especially for those taken at the final stage of the floral differentiation.
Overall, the results from the experimental assays carried out on mature flowers taken from various date palm varieties presented in this work revealed that:
- • all the varieties studied, even the highly recalcitrant ones, react to the 2,4-D effect as long as the conditions of favorable proliferation, as defined here, are applied;
- • this reactivity is strongly dependant on the genotype since wide variations in terms of appropriate hormonal dose and tissue reaction time are tightly connected to this effect;
- • for all the varieties studied, a reactive zone showed subsistence regardless of the floral differentiation degree.
In fact, those zones, which were abound at the base of the flower, corresponded to the sepal and petal armpits and covering all the basal area of the flower.
If the female flowers, at both young and adult stages, are characterized by zones maintained at a meristematic state, their tissues are noted to have different reactive capacities, given that they are not subjected to the same morphogenetic influences accounting for their very distinct differentiation states. However, the major difference between the two structural types resided in the ability of the female flower primordia, all through their meristem functioning, to assure the reversion of the whole floral structure, straight from the reproductive state to the vegetative one, to reconstitute a unique plant whose meristem is the subject of a simple deviation in functioning. In the case of the mature adult flowers, however, only the deviation of the floral tissues’ potentialities towards a vegetative type of functioning remained possible, with the floral meristem having already undergone the differentiation of all its constitutive territory, excluding all alternative possibility of direct reversion.
Concerning the deviation of the floral tissue potentialities to a vegetative type of functioning, it is worth noting that the tissues of the immature young flowers are endowed with the best capacities of proliferation and plant regeneration, without that those of the adult flowers being reduced to nothing. In fact, the initiation of plant issued via organogenesis and somatic embryogenesis methods was possible from adult flower organs, so these completely mature flowers conserve some zones which preserve all their morphogenetic potential as well as those of very juvenile flowers insofar as they are able to evolve into typical stamens at the latest mature stages.
Overall, the high ability of some cultivars to reverse, after few months of culture, from a flower propagation stage to a vegetative one through a high rate of flower-producing shoots is a strong evidence for the promising potential that mature flowers hold for existing propagation programs. This process, however, requires a high control of various physico-chemical and hormonal factors. Moreover, the ability of flower tissues to produce great numbers of nodules, despite their low percentage of development to organogenic growths through embryogenic callus, is another evidence for their ability to be used in rapid propagation programs. The zones located at the sepal and petal armpits might be more important than the other flower organs since they are meristematic and represent a store of dormant tissues that can be activated.In addition to their originality and important fundamental and practical attributes, as compared to the other plant organs used in regeneration processes, mature female inflorescences represent abundantly available plant material for use as explants and offer several highly valued advantages, namely the possibility of reversion from the reproductive to the vegetative state. This readily available material is accessible in large quantities every year, easy to collect without harming or sacrificing the mother plant, and carry no undesirable contaminative effects due to their protective spathe. Last but not least, the diverse neoformations initiated from the development of the floral tissue and the ease with which the induction and conversion processes occur as compared to those resulting from the vegetative tissue developments conferred to the floral tissues, represent an important morphogenetic plasticity that stands in contrast with the morphogenetic rigidity characterizing vegetative tissues [27].
Overall, the model presented in the current work is demonstrated to hold an number of promising and valuable attributes that make it a potential strong candidate for future application in a wide range of dioecious plants, particularly those having the ability to maintain some zones in a juvenile state, as is the case of date palm, and whose vegetative organs, used as collection source, were not abundant or sufficiently reactive. More succinctly, the extraordinary capacity of totally mature date palm female flower to proliferate to the regeneration of whole and viable plants from all the varieties studied resides in the preservation of minuscule zones at a meristematic state, capable of reacting irrespective of the differentiation state of the other floral organs.
Disclosure of interest
The authors declare that they have no conflicts of interest concerning this article.
Acknowledgements
This work was supported by the Tunisian Ministry of Higher Education, Scientific Research and Technology. The authors would like to express their gratitude to Mr Anouar Smaoui from the English department at the Sfax Faculty of Science for the valuable proofreading and language polishing services he made to the text of the current paper.


