1 Introduction
Social wasps, which very often have arboreal nesting habits, are regularly confronted with patrolling ants, so that, in the Neotropics, the predatory pressure exerted by ants has been considered the main driving force behind the evolution of wasp nest architecture [1–4]. Thus, to protect their colonies, non-swarming Polistinae build a nest with a pedicel onto which the workers deposit substances repellent to ants, while most swarming species build an envelope to protect the combs, facilitating the defence of the nest [5]. Furthermore, social wasps rely on active vigilance and physical removal to prevent ant intrusion [6,7] and select sites where attacks by ants are unlikely or more difficult (e.g., man-made structures, attaching their nests to the long thorns of palm trees that serve as easily-defended ‘pedicels’ [1,8,9]).
Whereas these defences seem effective against many ant species, they are futile against columns of army ants that force wasps to abscond and build a new nest elsewhere [10,11]. As a suitable defence mechanism against army ants, some wasp species install their nests in the vicinity of certain arboreal ant nests, the workers of which tolerate them, whereas they are very aggressive towards intruders. For instance, Polybia rejecta generally nests in close vicinity to large arboreal nests of Azteca chartifex [9,12] whose workers can defend the access to their host tree by attacking army ants at its base, causing the columns to deviate [13,14]. Such nest associations, well known throughout the Neotropics, concern a relatively limited number of social wasps species [9,13,15–18]. Although they benefit from protection by their associated ants, these wasps must nevertheless avoid attacks by the latter.
In French Guiana, the nests of Protopolybia emortualis de Saussure are systematically installed on trees sheltering colonies of the arboreal ant Dolichoderus bidens (L.) [9]. We therefore aimed to assess the level of specificity of this relationship by revisiting a corpus of field data collected during studies spreading over 20 years where all cases of associations between these wasps and arboreal ants were noted. To better understand the characteristics of the links between P. emortualis and D. bidens, we evaluated the opportunities for contact between the two species by comparing their respective rhythms of activity, studied the characteristics of the P. emortualis nest architecture and verified if a chemical mimicry exists between the wasps and the ants (see [16,19]) by comparing the cuticular hydrocarbons of P. emortualis and D. bidens individuals sharing the same trees.
2 Materials and methods
2.1 Study sites
These studies took place in the Sinnamary district, French Guiana, in a 30 km radius around the Petit-Saut dam (5°03’39“N, 53°02’36”W), mostly along dirt road corridors. The climate of the area studied is moist tropical with 3400 mm of yearly precipitation typically recorded at Petit-Saut. The maximum and minimum monthly temperatures average 33.5 °C and 20.5 °C, respectively. The dry season occurs between July and November and the rainy season between December and June with a marked decrease in precipitation during March.
2.2 Ant and wasp species
The arboreal ant D. bidens is abundant in pioneer formations, secondary forests and plantations [20]. The colonies, of up to a few thousand workers, are polydomous (i.e. multiple nests) with each carton nest built under a host tree leaf. The external part of the nests cover part or all of the adaxial surface of a single leaf or envelop two to a few contiguous leaves [11]. This is an aggressive, territorially dominant arboreal species active 24/7 that hunts insect prey in tree crowns [21]; consequently workers defend the access to their host tree by leaf-cutter ants and army ants at its base (AD, pers. obs.; see [13,14] for Az. chartifex).
Social wasps of the recently revised genus Protopolybia [22] are characterized by very different nest architectures, including transparent envelope made predominantly of chitin that is secreted orally in some species [23–25]. Diversification in female aggressiveness is seen in this genus, from very docile females in P. chartergoides, to extremely aggressive ones and a hemolytic venom in P. exigua [26,27]. Protopolybia emortualis is a swarming polistine species known to nest in close contact with D. bidens societies [28,29]; the workers of both species are approximately the same size.
2.3 Nesting association occurrences
In a previous field survey [9], wasp nests were looked for along forest edges and in secondary formations, noting, each time we encountered a wasp nest, whether or not it shared its host tree with arboreal ants. Because P. emortualis nests are difficult to detect, a complementary survey was conducted where we inspected 750 arboreal ant nests to verify whether their supporting trees also sheltered nests of this social wasp species (survey occurring from 2006 to 2016). Voucher specimens of P. emortualis and D. bidens were deposited in the American Museum of Natural History (New York) and the Laboratório de Mirmecologia (CEPEC-CEPLAC, Itabuna, Bahia, Brazil), respectively.
2.4 Rhythm of activity
We recorded the rhythms of activity of both the wasps and the ants living on nests installed on a Vismia sessilifolia (Clusiaceae) tree. The tree sheltered 42 nests of a D. bidens colony and one of P. emortualis. Due to frequent rainfall, observations were spread over one week in order to complete a 24-hour-day cycle. For each hour of the contiguous 24 h period, during 10 minutes we recorded:
- • the number of wasps leaving or returning to their nest;
- • those resting on the wasp nest;
- • the number of D. bidens workers patrolling on the wasp nest (i.e. on its “external envelope” or “shelter”) and on the three closest ant nests;
- • the number of ants on the main foraging trail running along the trunk of the tree;
- • the number of ants in two distinct foraging areas where they attended Hemiptera.
2.5 Gas-chromatography analysis
For the analysis of cuticular substances, we used three P. emortualis nests associated with two D. bidens colonies (i.e. two wasp nests associated with the same ant colony). Each ant and wasp sample (i.e. workers, larvae, and pupae) was prepared using the combined cuticular substances from five individuals killed by freezing and immersed in 1 mL of hexane for 5 min. The extracts were then evaporated under nitrogen and redissolved in 200 μL of hexane for D. bidens and P. emortualis workers and 50 μL for P. emortualis larvae and pupae and D. bidens larvae, pupae and queens (see Table 1). A sample of 2 μL of these solutions was analyzed with a Hewlett-Packard 5890 Series II gas chromatograph equipped with a split-splitless injector, a flame ionization detector, and a nonpolar fused-silica capillary column (HT-5, 25 m × 0.22 mm ID × 0.1 μm film thickness). Sample injections were performed in splitless mode using helium as the carrier gas with injector and detector temperatures at 300 °C and 320 °C, respectively. The oven temperature program was as follows:
- • 100 °C to 180 °C at a rate of 15 °C/min;
- • increased to 250 °C at 5 °C/min;
- • increased to 320 °C at 3 °C/min;
- • isothermal (320 °C) for 15 min.
Average Ochiai's resemblance coefficient (mean ± SE) as a measure of similarity in the cuticular profiles between D. bidens and P. emortualis larvae, pupae and adults (workers and queens for D. bidens). Ochiai's resemblance coefficient was calculated from binary values (i.e. presence/absence) in each recorded peak and vary between 0 (totally different) and 1 (identical).
| D. bidens larvae |
D. bidens pupae |
D. bidens workers |
D. bidens Queens |
P. emortualis larvae |
P. emortualis pupae |
P. emortualis adults |
|
| D. bidens larvae | 0.90 ± 0.05 | 0.92 ± 0.05 | 0.90 ± 0.02 | 0.80 ± 0.05 | 0.68 ± 0.07 | 0.69 ± 0.07 | 0.68 ± 0.08 |
| D. bidens pupae | – | 0.96 ± 0.03 | 0.87 ± 0.02 | 0.83 ± 0.02 | 0.70 ± 0.05 | 0.70 ± 0.05 | 0.70 ± 0.07 |
| D. bidens workers | – | – | 0.97 ± 0.01 | 0.78 ± 0.02 | 0.66 ± 0.05 | 0.69 ± 0.05 | 0.64 ± 0.07 |
| D. bidens queens | – | – | – | 0.95 | 0.65 ± 0.01 | 0.69 ± 0.05 | 0.66 ± 0.06 |
| P. emortualis larvae | – | – | – | – | 0.89 ± 0.10 | 0.78 ± 0.06 | 0.75 ± 0.08 |
| P. emortualis pupae | – | – | – | – | – | 0.86 ± 0.11 | 0.80 ± 0.11 |
| P. emortualis adults | – | – | – | – | – | – | 0.83 ± 0.13 |
Integrations were performed with Millennium 2.15 software (Waters). To estimate the similarities of the profiles, we converted the data into binary values (i.e. presence/absence) and used Ochiai's resemblance coefficient, which takes the same value as Nei's distance when binary data are used [30].
3 Results
3.1 Occurrences
During this survey, we recorded 45 active P. emortualis nests; one was associated with a Dolichoderus omacanthus colony and the 44 others with 40 D. bidens colonies out of the 147 noted (27.2%) among the ca. one thousand arboreal ant colonies surveyed. Moreover, three D. bidens colonies were associated with two P. emortualis nests, another with three P. emortualis nests (see [9] for other ant species).
3.2 Nest architecture
The P. emortualis nests were generally composed of one comb, rarely two (six cases out of 45) or three (two cases); each comb was protected by an envelope typical of swarm-founding wasps. This envelope started at the margins of a pedicellate comb as has generally been reported for Protopolybia [3]. For P. emortualis, the combs were hidden by an external carton envelope, or ‘shelter’, perfectly duplicating the envelope covering the ant nests; as for the latter, their envelopes sometimes enveloped two to four contiguous leaves. In most cases, the shelter margins were attached to the margins of the protecting leaf that acted as an umbrella. This carton shelter was connected to the comb envelope by several pillars that look like the pedicels attaching the combs to the leaf, which, as reported by Carpenter and Wenzel [31], seem to be made using salivary secretions. The nest entrances, located between the shelter and the supporting leaf margins, are often very narrow (ca. 2 mm in diameter), hence easy to defend by the resident wasps. So, access to these nests is difficult or impossible for D. bidens workers (Figs. 1, S1). In five cases, the P. emortualis nests were devoid of any shelter, which indicates that the latter are built after the comb and its envelope are constructed, although we never observed nest building.
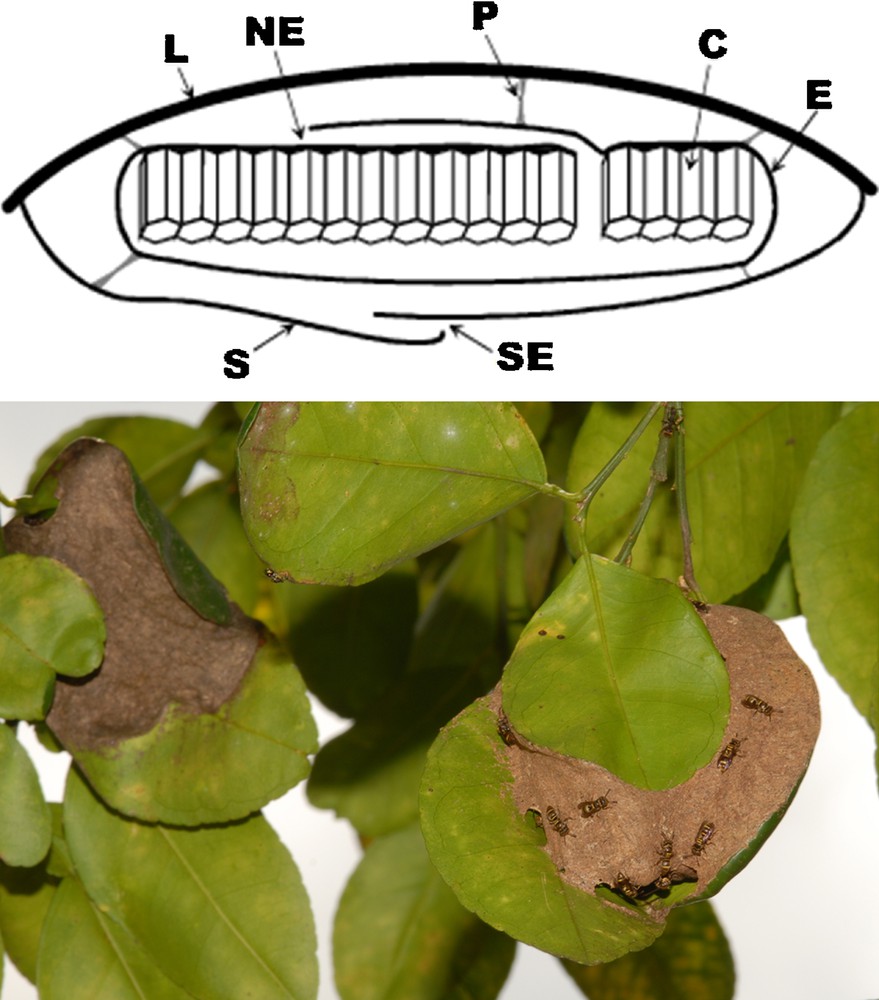
A. Diagram of a transverse section of one single-combed Protopolybia emortualis nest (C: comb; E: wasp-nest envelope; L: leaf; NE: P. emortualis nest entrance; P: pedicel; S: shelter; SE: shelter entrance). B. P. emortualis nest (on the right) near a D. bidens nest (on the left). The slightly opened shelter of the wasp nest allows to see the underlying envelope.
3.3 Rhythm of activity and alarm behavior
The foraging activities of P. emortualis workers were strictly diurnal (not represented here), while one to six individuals patrolled their nest shelter day and night (Fig. 2A). Dolichoderus bidens workers were active day and night, with nest patrolling mainly nocturnal (Fig. 2B, C). Despite the proximity of the respective nests (as close as 4 cm as the nests were located under adjacent leaves), the ants rarely climbed onto the wasp nests (only seven cases observed in 24 h). The D. bidens workers that approached the wasp nest entrance were immediately confronted with wasp guards with their mandibles open. This was enough to cause these ants to flee. Very rarely, P. emortualis workers landed on the D. bidens territory at close proximity to their own nests. They were not attacked by D. bidens workers as they avoided contact and immediately returned to their own nest (N = 14 observations). Short agonistic interactions with ants occurred only when the latter stepped onto the wasp nest and were reduced to a wasp approaching the ant intruder with the mandibles wide open; this was sufficient as the ant workers fled in all cases.
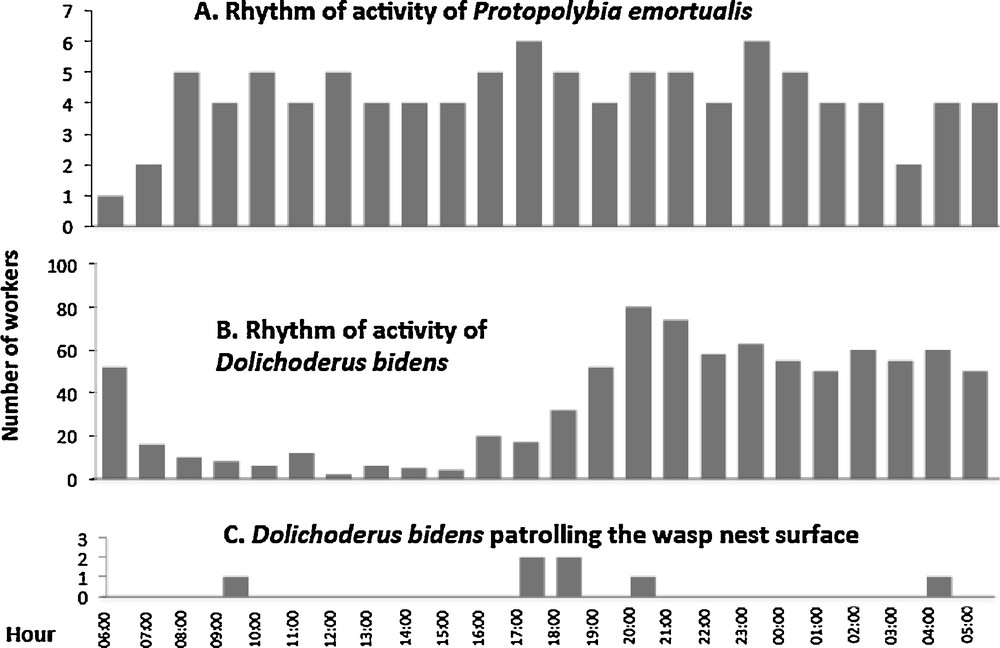
A. Rhythm of activity of Protopolybia emortualis workers on their nest's surface. B. Rhythm of activity of Dolichoderus bidens on three nests of their colony. C. Number of D. bidens workers patrolling the wasp nest surface.
When we simulated a disturbance by tapping the twig supporting the nests with a stick (N = 10), the D. bidens workers reacted with a typical alarm behavior consisting of drumming the abdomen on the branch, leaf or their nest surface (producing a sound similar to raindrops). This caused numerous nestmates to rush out of their nests ready to attack any intruder. On the contrary and simultaneously, P. emortualis wasps flew inside their nests, avoiding any contact with ants, while their nest entrance was blocked by the head of a wasp individual.
3.4 Gas-chromatography analysis
The analysis of the cuticular substances of D. bidens and P. emortualis workers and brood did not bring to light any congruence in the patterns (Fig. 3). Qualitative comparisons of the cuticular profiles using Ochiai's resemblance coefficient demonstrated that individuals were closer to their conspecifics (i.e. larvae, pupae or workers) than to the associated species (Table 1). Furthermore, the lowest qualitative similarity in the cuticular profiles corresponded to D. bidens and P. emortualis workers (i.e. those individuals that are likely to encounter each other).
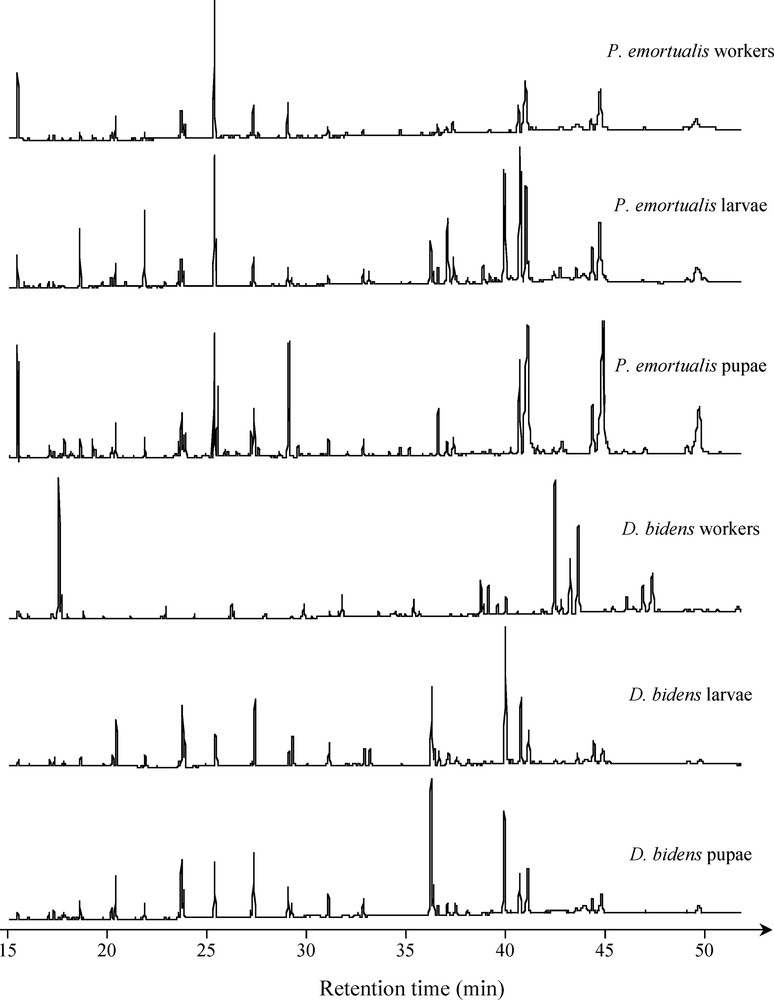
Comparisons of the cuticular profiles between developmental stages of Protopolybia emortualis and Dolichoderus bidens (i.e. workers, pupae, and larvae).
4 Discussion
Despite a high level of sampling effort during this study, P. emortualis was found only once out of 45 cases in the absence of D. bidens and this was with a congeneric species, whereas 27.2% of the inspected D. bidens colonies shared their host tree with one to three P. emortualis nest(s). Thus, P. emortualis swarms likely selects trees hosting D. bidens colonies for their installation (see also [9]).
Although P. emortualis and D. bidens workers do not tolerate contact with one another, the wasp nests are generally installed very close to an ant nest. Tolerance of the wasps by D. bidens workers and vice versa is not reflected by a convergence in the cuticular profiles of workers from the two species (chemical mimicry or camouflage) as is known for Parachartergus apicalis wasps nesting on myrmecophytic Vachellia spp. sheltering Pseudomyrmex ant colonies [16] (see also the case for several myrmecophiles [19,32–34]). Rather, such a scenario is reminiscent of the parabiotic associations between two ant species sharing the same nest in ant gardens, each species having its own colony odor. Thus, individuals are able to learn the chemical profile of both the parabiotic species and their colonymates [20].
Consequently, whereas their foraging activities are diurnal (as for most other wasp species), P. emortualis workers patrol the outer surface of their nest day and night, preventing D. bidens worker access. By very rarely venturing beyond their nest surface, P. emortualis wasps avoid physical contact with D. bidens workers, so that such contacts were rare. When an alarm was triggered in the ant colony, the wasps immediately took shelter in their nest, only protecting its small entrance (see also [28]). More generally, the wariness of P. emortualis enables them to mostly avoid D. bidens workers and thus contributes to the peaceful cohabitation. This is comparable to the way territorially-dominant arboreal ant species (D. bidens belongs to this group) tolerate non-dominant ant species (which have small colonies) on their territory that they would aggressively defend against other dominant ants [35,36].
The peculiarity of building an external supplementary envelope we called the ‘shelter’ in their nests constitutes the main novelty of P. emortualis. First, the shelters resembles the D. bidens nests (see also [37]: p 144) confusing predators such as birds. As far as we know, this is the only case of ant nest-mimicking construction in a social wasp species. This is reminiscent of the quite perfect camouflage of the greenish envelope of Leipomeles dorsata also displayed under a leaf [38]. Second, the presence of the ‘shelter’, hence a double envelope, renders access to the nest's interior more difficult for any intruder as it furnishes additional protection for the brood. Thus, the P. emortualis colonies benefit from two lines of defence where, in both cases, intruders need to gain access through a small entrance hole easy to defend by a single wasp (one of the guards). This situation is a strong protection from ant raids whose strategy is based on the Square Law of the Lanchester theory of combat predicting that when combatants can mix freely, numerical superiority is the deciding factor. Here, the wasp guards need to defend a narrow entrance, a situation corresponding to the Linear Law where fighting ability contributes more toward victory than the number of combatants. This is known for several ant species defending their nest entrances from army ant raid attacks (see [14] and papers cited therein). Thus, this double envelope can be useful when the D. bidens workers are excited by a strong external disturbance. However, it is very improbable that this strategy allows the tiny P. emortualis to resist any army ant raid.
Therefore, the main potential enemies of P. emortualis are birds, even tiny birds, detecting wasp nests by sight and striking them, making the wasps abscond (and to swarm to create a new nest), so that these birds can freely feed on the wasp larvae from the striken nest [39]. Yet, thanks to their mimetism with D. bidens nests, P. emortualis nests are undetectable by birds. Furthermore, because D. bidens workers are particularly aggressive and bite en masse all animals that venture on their host tree (AD, BC pers. com. [21]), a bird landing on such a tree is immediately attacked by hundreds of workers and instantly flies away. Note that the situation differs from that of Az. chartifex that are preyed upon by birds, but defended by associated aggressive social wasps [18].
In conclusion, the spatial nesting association between P. emortualis and D. bidens is asymmetrical with a minimum level of tolerance by the ants towards the presence of wasps. The wasp nesting behavior is based on:
- • an appropriate nest site selection (i.e. branches sheltering a D. bidens colony) permitting them to benefit from protection against army ant raids;
- • a nest architecture including a supernumerary envelope, the ‘shelter’, making the wasp nest indistinguishable from neighboring ant nests, hence the wasps benefit from a double line of defense from the D. bidens;
- • behavioral wariness which facilitates the cohabitation with D. bidens workers.
Disclosure of interest
The authors declare that they have no competing interest.
Acknowledgements
We are grateful to the Laboratoire Environnement de Petit Saut (HYDRECO) for logistical help and to Andrea Yockey-Dejean for proof-reading an early version of manuscript and Jacques H.C Delabie for the identification of the ants. Financial support for this study was provided by the project PEGGUY of the French PEPS-INEE programme (CNRS) and by an “Investissement d’Avenir” grant managed by the French “Agence Nationale de la Recherche” (CEBA, ref. ANR-10-LABX-25-01).


