1. Introduction
During cell motility, cells must integrate multiple physical inputs from the microenvironment as the extracellular matrix architecture, fibers and cell packing limit the available space. Indeed, migratory cells must constantly pass through narrow spaces as they migrate or grow within densely packed microenvironments [1, 2]. Therefore, they frequently experience cellular and nuclear deformations. Since the nucleus is the largest and stiffest cellular organelle, it will often constitute a limiting factor for confined cell migration. The nucleus is a mechanosensory organelle that senses compressive forces and rigidity of the extracellular environment and modulates migratory strategies [3, 4]. Importantly, in physiological contexts, when growing in densely packed environments or migrating across landscapes of varied porosities, geometries and densities, the nucleus is subjected to a wide range of deformations that will in turn modulate cell behavior, function and shape.
2. Contexts of nuclear deformations: the example of cell migration
2.1. Single cell migration
Migration in complex 3D environments brings its own set of constraints and presents many challenges to the cells, especially to their nuclei. They often face the challenge of squeezing through narrow gaps and constrictions in the surrounding tissue because of dense matrix meshworks (interstitial migration). The crossing of narrow pores results in a local compression of the cell and generates large intracellular forces that are transmitted across the cytoskeleton to the nucleus, leading to its deformation. Cells and cell collectives possess different migratory modes and change their migratory behavior in a tailored manner to adapt to complex 3D architectures of in vivo microenvironments. As a consequence of these highly complex and dense scenarios, motile cells often exhibit features associated with high nuclear deformability. For instance, leukocyte transendothelial migration (TEM) involves the opening of micron-wide gaps by the cells through paracellular junctions, followed by rapid squeezing of their bulky nuclei through relatively tight endothelial barriers (transmigration) [5, 6, 7, 8] (Figure 1). Interestingly, a study has demonstrated an active role for the nucleus in the process of TEM. Leukocyte nuclear lobes physically push and generate gaps and pores in between the endothelial cells for successful TEM [9]. Alternatively, several cell types change their protrusion mode when migrating in constricted spaces, alternating between lamellipodia and contractility-driven plasma membrane blebs [10, 11, 12, 13]. Some subtypes of aggressive invasive cancer cells solve the problem by secreting extracellular proteases that digest the matrix, therefore relieving the restrictive role of the environment. However, many other types of motile cancer cells and a large number of professionally migrating immune cell types do not ubiquitously employ the matrix proteolysis strategy; rather, they adapt their dimensions and migration choices to the available space around them.
Schematic representation of the different contexts of nuclear deformation and mechanosensitive signaling pathways discussed in this review.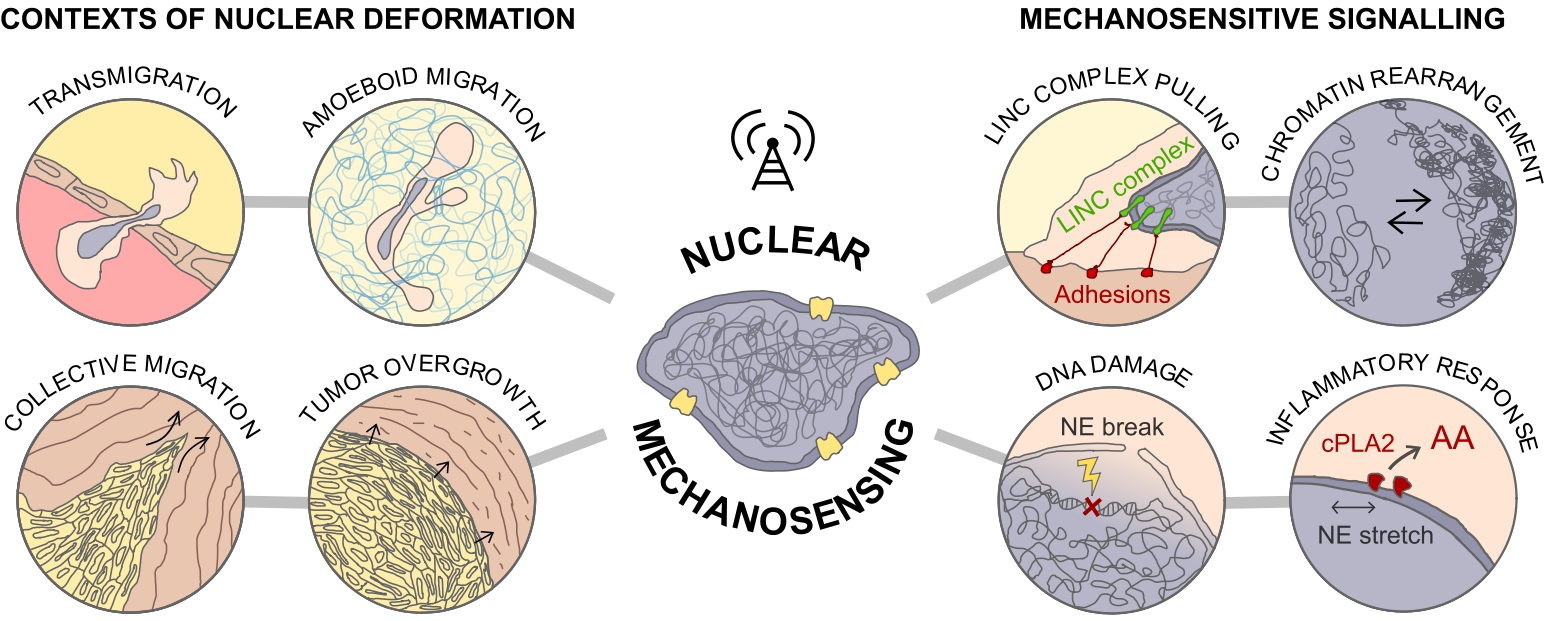
2.2. Collective cell migration
Besides the physical challenges experienced by single cells, mechanical constraints are also present during migration of cell collectives, potentially on a greater scale. Collective invasion of multiple cells occurs when moving cells maintain functional cell–cell adhesion, leading to a “supracellular” organization of the actin cytoskeleton as well as multicellular polarity and traction force generation [14, 15]. In tissues, cells do not exist in isolation, and the ability of cells to migrate collectively is crucial in shaping organisms during the complex morphogenetic events of development, and for several physiological and pathological events such as wound healing and cancer metastasis. For instance, leader cells of collective melanoma invasion developed adaptive invasion patterns along preformed tracks of complex topography, combining single-cell and collective migration modes, without immediate anatomic tissue remodeling or destruction [16]. These events led to both cell body and nuclear deformation. Likewise, the complex mechanical behavior of biological tissues and their active nature constantly generate laminar flows, swirling motions of cell clusters and vortices [15, 17, 18]. In particular, cell extrusion events were shown to be preceded by a coordinated, long-range flow of cells towards the eventual location of the extrusion. This causes considerable bending of cells, leading to high compressive stresses that are sufficient to trigger the extrusion of a nearby cell [17]. Thus, such dynamic regime of tissues is likely to impose intermittent physical strain to the cells and their nuclei, ultimately affecting cell behavior, fate and tissue homeostasis. Likewise, the movement of the collective strand of cells, including invasion through tissue barriers, can inflict severe physical challenges to the nucleus. When tissue gaps are smaller than the cell width, the proteolytic degradation of boundaries or non-proteolytic track widening (by passive outward pushing and space expansion) impose strong deformations to the cells and their nuclei. This is illustrated when breast cancer cells digest and breach the myoepithelial layer of the confining mammary duct to invade into the stroma. During this metastatic process, the collective motion of the strand of cells breaching the barrier encounters a “collective constriction” (the surrounding myoepithelial layer) that imposes high nuclear deformation and DNA damage to the invasive strand [19, 20] (Figure 1).
In summary, cells can squeeze themselves not only when individually navigating or growing in complex and dense microenvironments. They can also squeeze each other in motile invasive strands or during aberrant tissue overgrowth, such as in solid tumors [19] (Figure 1). Therefore, cells are constantly receiving mechanical input from extracellular environments of different topographical and physical features, and the nucleus has been a central player in modulating the cellular responses. Critically, nuclear plasticity and integrity will affect cell behavior and function, ultimately impinging on physiological and pathological processes. This demands the existence of specific mechanosensing mechanisms through which cells can “sense” the nuclear deformation and the tension state of the nuclear envelope (NE) to appropriately respond to them, thus maintaining proper cellular and tissue functions.
3. Signaling through nuclear mechanosensing
As cells sense the complexity of the extracellular microenvironments, mechanical signals are transmitted to the nucleus via the cytoskeleton and converted into signaling pathways to regulate cell behavior and function. The first study to show mechanical continuity between cell surface receptors and the nucleus was performed by the group of Donald Ingber. Upon micromanipulation of ligand-coated microbeads, they observed that cytoskeletal filaments reoriente, nuclei distorte, and nucleoli redistributs along the axis of the applied tension field [21]. Moreover, forces applied to integrins also induced displacements of nuclei up to 20 μm away from the site of force application, whereas generalized deformation of the surface bilayer only produced local effects at the cell surface [22]. Now, we know that such phenomenon of mechanosensation and force transmission through the cytoplasm to the nucleus occurs via the LINC complex [23] (Figure 1). Additionally, nucleoskeleton and transmembrane NE proteins bind chromatin, generating stress at the level of the NE, leading to nuclear deformation and changes in chromatin organization [24, 25, 26] (Figure 1). Therefore, these mechanical signals ultimately influence gene expression and control multiple aspects of cell behavior, including growth and differentiation, and even cancer progression. Additionally, during nuclear mechanosensing, nuclear pores physically expand and contract, thus altering their transport rates. Indeed, more recently, the direct effect of nuclear deformation on the import of transcription factors has brought back into the picture the deformation of the nucleus, and more precisely the NE as a sensor of mechanical constraints. Importantly, this study contributed with a potential general applicability for mechanosensing pathways in transcriptional regulation [27]. Altogether, these findings demonstrate that mechanical forces that are applied at the cell surface do more than activate membranesignaling events. The subsequent mechanosensing pathways promote structural rearrangements deep in the nucleus. The magnitude of the signal will shape both short- and long-term responses, differently impacting gene regulation, cell behavior and cell fate. Broadly, modulation of the NE state can affect more than an individual cell at a given time. The consequences can be far-reaching, be it spatially or temporally, all the way to impacting tissue homeostasis and developmental fate. Critically, extreme perturbations on cells and their nuclei might also have detrimental effects. Therefore, how cells adapt their responses or avoid harmful perturbations when growing and migrating within confined 3D microenvironments becomes critical. Indeed, in more extreme cases, nuclear blebs have been shown to systematically form and rupture at sites of high local nuclear curvature [28, 29, 30, 31]. Critically, these events might bring harmful consequences such as repeated loss of nucleo-cytoplasmic compartmentalization and chronic DNA damage [1, 2, 19, 32]. In contrast, severe nuclear deformations might also trigger mechanisms that help cells navigate the complexity of dense matrices. In an elegant work, Lomakin and colleagues showed that cells measure the degree of spatial confinement using their nucleus. When deformed below a specific height, the NE unfolds to its maximum and it becomes tensed. The augmented NE tension leads to recruitment of cytosolic phospholipase A2 (cPLA2) to the inner NE surface, causing an increase in cortical actomyosin contractility, producing pushing forces to resist physical compression [33]. NE stretching has also been shown to play a critical role in modulating inflammatory response via cPLA2 [34] (Figure 1).
Another major perturbation experienced by cells under mechanical strain lies at the level of the chromatin. Chromosome dynamics and motion are locally transmitted to discrete sites of the NE via lamina-associated domains (LADs). LADs and heterochromatinization can be responsible for forming large NE folds that will pull on the NE and generate topological tension [35]. Moreover, there is evidence of cross-talk between the NE and chromatin state during cell migration. For instance, epigenetic reprogramming was shown to start shortly after initiation of migration. Likewise, treatment of cells with drugs to abolish epigenetic modifications led to decreased motility [36, 37]. It is also plausible that the fluctuations on NE tension experienced by cells migrating on stiff matrices or in confined 3D microenvironments might change their transcription programs [26, 38].
4. Conclusions
The crosstalk between microenvironmental physical strains and the cell nucleus triggers nuclear mechanosensing pathways that regulate cellular homeostasis and cell behavior. Critically, in order to maintain cellular homeostasis, a fine balance must exist between what cells sense as a perturbation (potentially threatening their integrity), and the signaling pathways that orchestrate their behavior, function, and at a greater level, tissue homeostasis. In this context, the nucleus has emerged as a key mechanosensory organelle engaged in tuning these responses. In the light of evolution, frequent and unavoidable sources of damages, such as extreme nuclear deformations and NE ruptures, are likely to be integrated into signaling pathways to modulate cellular behaviors, fate and tissue physiopathology.
Declaration of interests
The authors do not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and have declared no affiliations other than their research organizations.
Acknowledgements
We thank Dr. Matthieu Piel (Institut Curie, Paris, France) for all the scientific input, invaluable mentoring and for providing the funding for the postdoctoral research of GPFN, which made this article possible.




 CC-BY 4.0
CC-BY 4.0