1 Introduction
Multicomponent reactions (MCRs) provide unmatched opportunities for the expeditious increase of complexity and diversity in synthetic outcomes. The strategy offers significant advantages over classical stepwise approaches, allowing the formation of several bonds and the construction of complex molecular architectures from simple precursors in a single synthetic operation without the need for the isolation of intermediates [1]. MCRs, particularly those performed in aqueous media, have become increasingly useful tools for the synthesis of chemically and biologically important compounds because of their environmentally friendly, atom economy and green characteristics [2,3].
Important pharmaceuticals often possess heterocyclic moieties as their building blocks. Since pyrazoles and its derivatives possess various biological activities, such as anti-inflammatory, antipyretic, gastric secretion stimulatory, antidepressant, antibacterial, and antifilarial agents [4–7], the development of new methods for the synthesis of pyrazole derivatives, which will yield subsets of heterocycles having the potentiality to serve as templates for new biologically active molecules, is of great importance.
2 Results and discussion
In continuation of our interest in the synthesis of heterocyclic compounds based on in MCRs [8], we developed herein the synthesis of spiro indeno[1,2-b]quinoxalines 8 via a three-component condensation reaction of 11H-indeno[1,2-b]quinoxalin-11-one 6, pyrazolone 3 and malononitrile 7 in ethanol by using Na2CO3 at 70 °C in high yield (Scheme 1). It should be noted that pyrazolone 3 was synthesized by the condensation between β-keto esters 1 and hydrazines 2 in ethanol after 5 min [9]. Also, 11H-indeno[1,2-b]quinoxalin-11-ones 6 was synthesized according to a previous work [10] by means of a reaction between 1,2-phenylenediamines 4 and ninhydrine 5 (Scheme 1).
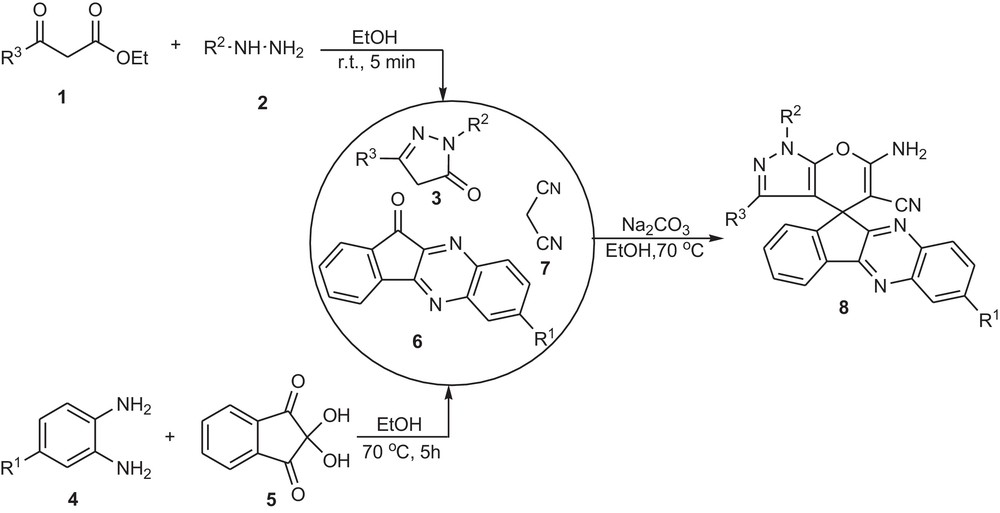
Synthesis of spiro indeno[1,2-b]quinoxalines.
As a model reaction, we first investigated the condensation of 11H-indeno[1,2-b]quinoxalin-11-one 6, 3-methyl-1H-pyrazol-5-ol 3 and malononitrile 7 under various conditions (Table 1). We first investigated the model reaction rate in different solvents by measuring the isolated yield using identical amounts of reactants in the presence of 10% mol of Na2CO3 for a fixed reaction time of 12 h at 70 °C (Table 1, entries 1–7). The desired product was obtained in polar solvents, such as water, ethanol, and methanol, ethyl acetate, and acetonitrile, but ethanol can afford the product in good yield even better than other solvents (Table 1, entry 2). The desired product was not obtained in non-polar solvents, such as dichloromethane, toluene (Table 1, entries 6–7). This effect can be explained by a simple acid-catalysis mechanism facilitated by the strong hydrogen bond interaction at the organic–ethanol interface, which stabilizes the reaction intermediate. Also, it was found that the addition of water to the ethanol solution cannot improve the reaction outcome (entry 2 and entries 8–10) and that, interestingly, the corresponding product was obtained in high yield when the reaction was performed in pure ethanol (entry 2).
Optimization of the reaction.
| Entry | Solvent | Temperature (°C) | Yield (%) |
| 1 | Water | 70 | 85 |
| 2 | Ethanol | 70 | 93 |
| 3 | Methanol | 25 | 90 |
| 4 | Ethyl acetate | 70 | 75 |
| 5 | Acetonitrile | 70 | 92 |
| 6 | Toluene | 70 | Trace |
| 7 | Dichloromethane | 25 | Trace |
| 8 | Water/ethanol (25%) | 70 | 83 |
| 9 | Water/ethanol (50%) | 70 | 80 |
| 10 | Water/ethanol (75%) | 70 | 75 |
| 11 | Ethanol | 25 | 80 |
| 12 | Ethanol | 50 | 85 |
| 13 | Ethanol | 100 | 93 |
Next, we studied the model reaction in ethanol at different temperatures (entry 2 and entries 11–13). The reaction rate increased as the temperature was raised. At 70 °C, the maximum yield (93%) was obtained in a reaction time of 12 h (entry 2).
Various basic catalysts, such as Na2CO3, K2CO3, Et3N, and NaOH, were examined for this reaction; in all cases, the reaction yields were good, with very low differences. The results indicated that, although all these catalysts were found to be effective for this reaction system, the reaction yield was a little better when Na2CO3 was used. Also, Na2CO3 is a cheap, common and available catalyst and acts as a heterogeneous one in this reaction. Therefore, we decided to use Na2CO3 as the base catalyst in this reaction. Further work indicated that the best results were obtained when the reaction was carried out at 70 °C for 12 h in ethanol in the presence of 10% mol of Na2CO3.
With optimized conditions established, we next attempted to extend the process to two different 11H-indeno[1,2-b]quinoxalin-11-ones, such as 11H-indeno[1,2-b]quinoxalin-11-one and 7-methyl-11H-indeno[1,2-b]quinoxalin-11-one, and various pyrazole as 3-methyl-1H-pyrazol-5-ol, 3-propyl-1H-pyrazol-5-ol and 3-methyl-1-phenyl-1H-pyrazol-5-ol. The results are summarized in Table 2. The structures of the products were established by spectroscopic methods. In all cases, good yields were obtained.
Substituted spiro indeno[1,2-b]quinoxalines.
| Entry | R1 | R2 | R3 | Product | Yield(%) |
| 1 | H | H | Me | 8a | 93 |
| 2 | Me | H | Me | 8b | 90 |
| 3 | H | H | Pr | 8c | 89 |
| 4 | Me | H | Pr | 8d | 87 |
| 5 | H | Ph | Me | 8e | 85 |
| 6 | Me | Ph | Me | 8f | 80 |
Mechanistically, it is conceivable that the reaction involves the initial formation of the Michael acceptor 9 by the Knoevenagel condensation of the 11H-indeno[1,2-b]quinoxalin-11-one 6 with malononitrile 7. The active methylene of pyrazolone 3 is converted into the enolate form in the presence of Na2CO3; it was then reacted with 9 in a Michael addition reaction to give the intermediate 10, which then undergoes a Thorpe–Ziegler intramolecular cyclization and subsequently, a 1,3-sigmatropic shift into spiro indeno[1,2-b]quinoxalines 8. To clarify the proposed mechanism, first, the Michael acceptor 9 was synthesized by Knoevenagel condensation of the 11H-indeno[1,2-b]quinoxalin-11-one 6 with malononitrile 4. Subsequently, the reaction of 9 with pyrazolone 3 afforded the corresponding spiro indeno[1,2-b]quinoxalines 8 (Scheme 2).

Proposed mechanism.
3 Conclusion
In conclusion, we have developed a new and efficient approach for the synthesis of a wide range of spiro indeno[1,2-b]quinoxaline derivatives from 11H-indeno[1,2-b]quinoxalin-11-one, pyrazolone and malononitrile in ethanol by using Na2CO3 at 70 °C in high yield. The reaction has been shown to display good functional group tolerance, is high yielding and product isolation is very straightforward.
4 Experimental
4.1 Materials and techniques
Melting points were taken with an Electrothermal 9100 apparatus and left uncorrected. IR spectra were obtained with a Shimadzu IR-470 spectrometer. 1H and 13C NMR spectra were recorded with a Bruker DRX-250 Avance spectrometer at 250 and 62 MHz. NMR spectra were obtained with solutions in DMSO using tetramethylsilane as the internal standard. All the chemicals were purchased from Fluka, Merck, and Aldrich, and used without purification.
4.2 General procedure for the synthesis of pyrazolone derivatives (3)
A solution containing 1,3-ketoester (1.0 equiv) and hydrazines (1.1 equiv) in ethanol (10 mL) was stirred at room temperature for 5 min. After the reaction was completed, the solution was diluted with ethanol (10 mL) and stirred in an ice bath for 30 min. The resultant solid was filtrated, washed with cold ethanol (10 mL), and recrystallized from ethanol to give pure pyrazolones.
4.3 General procedure for the synthesis of 11H-indeno[1,2-b]quinoxalin-11-one derivatives (6)
A solution containing 1,2-phenylenediamines (1.0 equiv) and ninhydrine (1 equiv) in ethanol (10 mL) was stirred at 70 °C for 5 h. After the reaction was completed, the solution was diluted with ethanol (10 mL) and stirred in an ice bath for 30 min. The resultant solid was filtrated, washed with cold ethanol (10 mL), and recrystallized from ethanol to give pure 11H-indeno[1,2-b]quinoxalin-11-one.
4.4 General procedure for the synthesis of spiro indeno[1,2-b]quinoxalines (8a–f)
To a neat solution containing 11H-indeno[1,2-b]quinoxalin-11-one (1 mmol), pyrazolone (1 mmol), and malononitrile (1 mmol), 10% mol of Na2CO3 in ethanol (10 mL) was added. The reaction mixture was heated at 70 °C for 12 h. After completion of the reaction as indicated by TLC, the precipitate was filtrated and washed with cold ethanol to afford the pure product 8.
4.5 6′-Amino-3′-methyl-1′H-spiro [indeno[1,2-b] quinoxaline-11, 4′-pyrano[2,3-c]pyrazole]-5′-carbonitrile (8a)
Light green powder (0.36 g, yield 93%). Mp 263–266 °C. IR (KBr) (vmax/cm−1): 3450, 3274, 3250, 3112, 2976, 2191, 1637, 1607, 1465, 1401. 1H NMR (250 MHz, DMSO-d6): δH (ppm) 1.12 (3H, s, CH3), 7.45–8.13 (10H, m, H–Ar and NH2), 12.51 (1H, s, NH).13C NMR (62 MHz, DMSO-d6): δC (ppm), 9.5 (CH3), 56.5 (C–Spiro), 96.8 (C–CN), 119.3 (CN), 121.4, 122.1, 126.2, 129.4, 129.6, 130.1, 130.8, 133.5, 135.5, 136.0, 141.8,142.5, 151.4, 153.3, 155.9, 162.9 (C–Ar and C–Pyrazole), 164.8 (C–NH2). Anal. calcd for C22H14N6O: C, 69.83; H, 3.73; N, 22.21. Found: C, 69.88; H, 3.72; N, 22.22.
4.6 6′-amino-3′, 7-dimethyl-1′H-spiro [indeno[1,2-b]quinoxaline-11′,4-pyrano[2,3-c] pyrazole] carbonitrile (8b)
Light brown powder (0.35 g, yield 90%). Mp 258–262 °C. IR (KBr) 3445, 3250, 3224, 2966, 3110, 2193, 1638, 1597, 1447, 1398. 1H NMR (250 MHz, DMSO-d6): δH (ppm) 1.13 (3H, s, CH3), 2.58 (3H, s, CH3–Ar), 7.32-8.14 (9H, m, H–Ar and NH2), 12.6 (1H, s, NH). 13C NMR (62 MHz, DMSO-d6): δC (ppm) 8.9 (CH3), 21.1 (CH3–Ar), 56.5 (C–Spiro), 96.3 (C–CN), 118.7 (CN), 121.4, 125.6, 127.8, 128.6, 129.4, 131.5, 132.7, 135.6, 139.7, 140.4, 142.0, 150.8, 152.6, 155.0, 162.4 (C–Ar and C–Pyrazole), 163.3 (C–NH2). Anal. calcd for C23H16N6O: C, 70.40; H, 4.11; N, 21.42. Found: C, 70.44; H, 4.14; N, 21.39.
4.7 6′-amino-3′-propyl-1′H-spiro[indeno[1,2-b]quinoxaline-11,4′-pyrano[2,3-c]pyrazole]-5′-carbonitrile (8c)
Light orange powder (0.34 g, yield 89%). Mp 248–252 °C. IR (KBr) (vmax/cm−1): 3307, 3240, 3225, 3156, 2956, 2199, 1654, 1605, 1462, 1402. 1H NMR (250 MHz, DMSO-d6): δH (ppm) 0.16 (3H, m, CH2CH2CH3), 0.58 (2H, m, CH2CH2CH3), 1.48 (2H, m, CH2CH2CH3), 7.36- 8.21 (10H, m, H–Ar and NH2), 12.43 (1H, s, NH). 13C NMR (62 MHz, DMSO-d6): δC (ppm) 12.8 (CH2CH2CH3), 20.6 (CH2CH2CH3), 23.1 (CH2CH2CH3), 56.5(C–Spiro), 95.9 (C–CN), 118.8 (CN), 121.5, 125.8, 128.8, 129.1, 129.6, 130.2, 132.9, 135.6, 138.9, 141.2, 141.9, 151.3, 152.8, 155.2, 162.4 (C–Ar and C–Pyrazole), 164.6 (C–NH2). Anal. calcd for C24H18N6O: C, 70.92; H, 4.46; N, 20.68. Found: C, 70.95; H, 4.48; N, 20.69.
4.8 6′-amino-3′-propyl-7-methyl-1′H-spiro[indeno[1,2-b]quinoxaline-11,4′-pyrano[2,3-c]pyrazol]-5′-carbonitrile (8d)
White powder (0.34 g, yield 87%). Mp 249–252 °C. IR (KBr) (vmax/cm−1): 3444, 3288, 3275, 3050, 2965, 2197, 1635, 1605, 1459, 1437.1H NMR (250 MHz, DMSO-d6): δH (ppm) 0.16 (3H, m, CH2CH2CH3), 0.56 (2H, m, CH2CH2CH3), 1.48 (2H, m, CH2CH2CH3), 2.53 (3H, s, CH3–Ar), 7.44-8.13 (9H–Ar and NH2), 12.48 (1H, s, NH). 13C NMR (62 MHz, DMSO-d6): δC (ppm) 12.8 (CH2CH2CH3), 19.9 (CH3–Ar), 20.6 (CH2CH2CH3), 21.1 (CH2CH2CH3), 56.5 (C–Spiro), 96.1 (C–CN), 118.8 (CN), 121.3, 125.7, 128.0, 128.4, 129.4, 132.2, 132.6, 135.7, 138.8, 139.8, 140.3, 141.3, 151.0, 155.2, 162.3 (C–Ar and C–Pyrazole), 164.5 (C–NH2). Anal. calcd for C25H20N6O: C, 71.41; H, 4.79; N, 19.99. Found: C, 71.42; H, 4.75; N, 19.93.
4.9 6′-amino-3′methyl-1′H-spiro[indeno[1,2-b]quinoxaline-11,4′-pyrano[2,3-c]-1′-phenylpyrazole]-5′-carbonitrile (8e)
Light orange powder (0.33 g, yield 85%). Mp 241–244 °C. IR (KBr) (vmax/cm−1): 3320, 3293, 3063, 2930, 2204, 1665, 1632, 1492, 1396. 1H NMR (250 MHz, DMSO-d6): δH (ppm) 1.11 (3H, s, CH3), 7.38–8.37 (15H–Ar and NH2). 13C NMR (62 MHz, DMSO-d6): δC (ppm) 12.5 (CH3), 56.2 (C–Spiro), 98.7 (C–CN), 118.1 (CN), 120, 121.3, 123.7, 125.6, 127.5, 128.2, 129, 129.4, 131.2, 136.2, 134.3, 138.3, 138.7, 141.2, 142.5, 143.7, 151.9, 154.4, 160.6 (C–Ar and C–Pyrazole), 164.4 (C–NH2). Anal. calcd for C28H18N6O: C, 74.00; H, 3.99; N, 18.49. Found: C, 74.04; H, 3.97; N, 18.46.
4.10 6’-amino-3’,7-dimethyl-1’H-spiro[indeno[1,2-b]quinoxaline-11,4’-pyrano[2,3-c]-1’phenylpyrazole]-5’-carbonitrile (8f)
Light brown powder (0.31 g, yield 80%). Mp 206–210 °C. IR (KBr) (vmax/cm−1): 3322, 3287, 3080, 2919, 2205, 1654, 1627, 1455, 1394. 1H NMR (250 MHz, DMSO-d6): δH (ppm) 1.10 (3H, s, CH3), 2.57 (3H, s, CH3–Ar), 7.41–8.48 (14H–Ar and NH2). 13C NMR (62 MHz, DMSO-d6): δC (ppm) 11.72 (CH3), 15.1 (CH3–Ar), 56.1(C–Spiro), 96.87 (C–CN), 118.12 (CN), 120.0, 121.5, 125.9, 126.5, 128.1, 128.6, 129.4, 130.6, 131.9, 132.8,135.1, 136.9, 140.6, 141.3, 142.5, 143.7, 151.2, 153.1, 161.1 (C–Ar and C–Pyrazole), 165.1 (C–NH2). Anal. calcd for C29H20N6O: C, 74.34; H, 4.30; N, 17.94. Found: C, 74.35; H, 4.31; N, 17.92.
Acknowledgements
We gratefully acknowledge financial support from the Research Council of Razi University.


