1 Introduction
In recent years, global concern has arisen concerning the risks of decline in the health of marine ecosystems and the associated functions and services they provide. This has brought about many innovative studies and much path-breaking research, and has led to the development of guidance and policies worldwide. Most of this information underlines the need for observing and managing the state of marine ecosystems as a whole (Tett et al., 2013, Grand Challenge 4 in Borja, 2014). Moreover, scientific literature about marine ecosystems has increased exponentially during the last 15 years (Borja, 2014). Coastal ecosystems are likely to have been altered substantially by human activities and probably also by climate drivers of change (climate change related key drivers) (Parry et al., 2007). As stated by Halpern et al. (2008), no marine system remains unaffected by human influence and a large fraction (41%) is strongly affected by multiple drivers.
Marine ecosystems are highly diverse given their biogeographical characteristics on a global scale. Their morphology controls internal hydrodynamics and exchanges with the adjacent sea, regulating in this way the level of anthropogenic pressures and sensitivity to eutrophication (Kennish, 2002). Semi-enclosed coastal systems (SECS), which usually include open, leaky, and choked lagoons and transitional waters (EU, 2000), are sentinel systems and hotspots of coastal vulnerability at a global scale (Newton and Weichselgartner, 2014; Newton et al., 2014 and references therein). Because of the physical connection between the benthic layer and the whole water column, the geochemical and biological dynamics extensively depend on benthic–pelagic interactions.
The Términos Lagoon in the southern Gulf of Mexico is one of the largest lagoons situated in the intertropical zone. Considering the extension and shallowness of the Términos Lagoon as well as its potential sensitivity to environmental change, the objective of this paper is to review and combine existing information on what has been identified as a model tropical coastal lagoon in the Gulf of Mexico (García-Ríos et al., 2013), where seasonal signals are distinct from those in temperate regions. The following sections present an overview of the general features of the Términos Lagoon, as well as its hydrodynamic characteristics and its benthic habitats, with special attention paid to trophic status and benthic communities, as they strongly contribute, together with external loadings, to the biogeochemistry of the water column. In the context of the expected Global Change in the Central American region, the present review also presents many specific drivers of change and pressures, including land use, watershed management, river inputs, coastal erosion processes, and contaminant levels, to assess what major changes can be expected and what effect they might have on the current environmental status, finally issuing recommendations for a long-term environmental survey strategy.
2 General features
The Términos Lagoon borders the southern Gulf of Mexico in Campeche and is, by area and volume, Mexico's second largest estuarine system. The lagoon borders two geologic provinces: to the east the Yucatan Peninsula (low rainfall, calcareous soils, and very low surface drainage) and to the west and south the lowlands of Tabasco and the highlands of Chiapas and Guatemala, an area of high rainfall and fluvial soils (Fig. 1). Three main rivers discharge directly into the lagoon: the Candelaria, the Chumpan, and the Palizada (a tributary of the Grijalva-Usumacinta), with a catchment area totaling 49,700 km2.
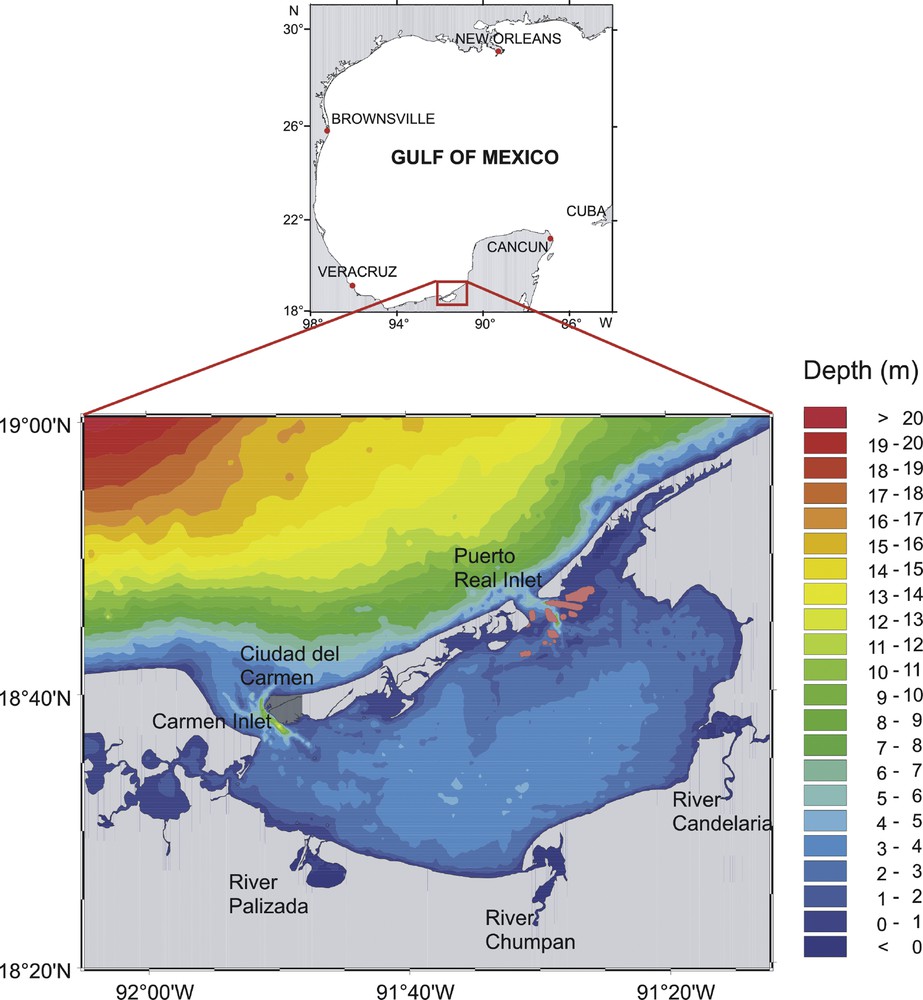
Bathymetric map of the study area with major rivers and connections to the Gulf of Mexico.
The average freshwater flow rate is approximately 12.5 × 109 m3·yr−1, with the Palizada River catchment area on the western coast of the lagoon accounting for most of the fresh water inputs. The tidal regime is mixed, mostly diurnal with a mean range of 0.3 m (Contreras Ruiz Esparza et al., 2014).
The continental shelf (Campeche Sound) is one of the most important fishery areas in the western central Atlantic region. Campeche Sound (Tabasco/Campeche) contributes 34% of the total Mexican fishery yield in the Gulf and Caribbean coasts, including penaeid shrimps, mollusks, demersal, and pelagic fishes (CONAPESCA, 2008).
Parallel to fisheries, crude oil extraction in the Gulf of Mexico represents a large economic activity, with Petroleros Mexicanos (PEMEX), a major non-OPEC oil producer (top 10) operating here since 1938. PEMEX is a state-owned company extracting around 2 million barrels per day in Bahia de Campeche (Cantarell oil field). More significantly, revenues from the oil industry (including taxes and direct payments from PEMEX) accounted for about 32% of total Mexican government revenues in 2013. This activity has generated the urban development of Ciudad del Carmen, the largest town (located on the western tip of the island) and, in 1994, it contributed to a great extent to the declaration of the Términos Lagoon as a Protected Area of Flora and Fauna (APFFLT). Ten years later, the lagoon was designated as a wetland of international importance (RAMSAR), and today it represents the largest such zone in Mexico, with 705,016 ha (www.ramsar.org). Unfortunately, PEMEX's activity continues to grow within the protected area, potentially threatening the whole ecosystem through accidental oil spills and pipeline leaks (e.g., Ixtoc-1 in 1979–1980).
The vast wetlands surrounding the Términos Lagoon provide ideal conditions for the migration and breeding of numerous species such as sea bass, shellfish, shrimp, and manatees, amongst others. This area also harbors the highest concentration of dolphins in the Gulf. This protected area is also one of the most important bird wintering areas in the Gulf of Mexico, and its level of marine and terrestrial flora and fauna diversity is considered as very high (Yáñez-Arancibia and Day, 1988). The coastal fringe is occupied by vast areas of mangroves, and the lagoon comprises various habitats, including important oyster and sea grass beds (Moore and Wetzel, 1988).
The area presents two climatic seasons (Yáñez-Arancibia et al., 1983): the rainy season (June–September) and the dry period, which splits into a period with dominant north winds and winter storms (November–March), and a second with lower wind forcing (February–May). The average evaporation is 1512 mm·a−1 and the average rainfall is 1805 mm·a−1 (Yáñez-Arancibia and Day, 1988). Nevertheless, a strong variability in rainfall and subsequent freshwater discharge to the lagoon has recently been linked to climate variability and to the ENSO regime (Fichez et al., 2017).
The Términos Lagoon stretches over more than 2000 km2 (Fig. 1). The lagoon is connected to the sea through two inlets: the ‘Carmen Inlet’ on the western side (4 km in width) and the ‘Puerto Real Inlet’ on the eastern side (3.3 km in width). These are separated by a carbonate-rich barrier (Carmen Island, 30 km long and 2.5 km wide). The Términos Lagoon is shallow, with an average depth of 2.4 m, apart from the tidal flats and of the deep channel on the eastern part of each inlet. The eastern entrance is influenced by transparent marine waters that form an interior delta. In the western inlet, the suspended terrigenous materials of the Palizada River estuary generate high turbidity and contribute to the formation of an exterior delta oriented towards the west in the Gulf of Mexico.
Waters are mostly oligo- to mesohaline, with a tendency to being euhaline in some parts of the system; the east inlet is characterized by marine waters, whereas the west inlet is mesohaline to oligohaline near the river mouths (Bach et al., 2005). Predominant sediments are silt and clay, with calcareous sands (shells and shell fragments) present in the eastern part and close to the barrier island only. The sediments of the western part are silty-clay (Yáñez-Arancibia et al., 1983).
A study by Yáñez-Correa (1963) compared the sediment structures of both inlets, and showed that sediment accumulation outside the Carmen Inlet in the west contributed to the formation of a small alluvial delta outside the lagoon, due to the predominant seaward direction of the currents. Conversely, in the east, at the Puerto Real Inlet, the existence inside the lagoon of a submarine alluvial delta composed of a coarse sediment fraction of marine origin reveals an inward component of the currents; one of the main sources of sediments is thus offshore derived waters entering the lagoon.
The few published studies dealing with hydrodynamic modeling in Términos Lagoon (Contreras Ruiz Esparza et al., 2014; Jensen et al., 1989; Kjerfve et al., 1988) converge to describe a general circulation pattern from east to west, with a net westward transport of the water masses entering the lagoon through the Puerto Real Inlet and exiting through the Carmen Inlet during the dry season, and a dominant net export of water through both inlets during the wet period. Tidal effects combined with freshwater inputs tend to bend the southwestward current into a cyclonic circulation inside the lagoon — with a vortex core located in the northeastern part — while predominant winds from the east (67% from 45° to 135°) tend to drive the river plumes toward the Carmen Inlet (Contreras Ruiz Esparza et al., 2014). Based on tidal current measurements in and around the inlets, David and Kjerfve (1998) estimated that fifty percent of the lagoon water volume was renewed in nine days. However, long-term trends of residence times of the water masses for the whole lagoon remain difficult to estimate, considering that water replacement in the Términos Lagoon does not behave linearly but decays exponentially (Yáñez-Arancibia and Day, 2005). The recent development of a 3-D hydrodynamic modeling approach of the Términos Lagoon accounted for tidal forcing only (Contreras Ruiz Esparza et al., 2014) and thus prevented the computing of water mixing and renewal indicators (Umgiesser et al., 2014). However, a relatively simple water budget could be established using the net freshwater inputs (river + groundwater + net precipitation) previously estimated at 12.5 × 109 m3·yr−1 (Fichez et al., 2017) and the water volume of 4.65 × 109 m3 (Contreras Ruiz Esparza et al., 2014), yielding an average flushing time (Tf, Monsen et al., 2002) of 135 days, a value within the 1 to 5 months range previously reported by Robadue et al. (2004) for different seasonal conditions.
3 Water column biogeochemistry
The Términos Lagoon is a “choked” lagoon subject to significant fresh water inputs, so salinity is strongly variable at the spatial as well as at the temporal scale. Recent results from a survey (currently October 2008 to September 2010, 10 samplings over a network of 34 stations) yielded an average salinity of 25.7 at a depth of 1 m (standard deviation = 8.1) with values ranging from 3 to 37 (Fichez et al., 2017). Such high variability may be related to the high variability in precipitation and subsequent river discharge, sea level, and wind patterns, as documented in Fuentes-Yaco et al. (2001).
Nevertheless, the distribution of salinity in the Términos Lagoon for January 2010 (Fig. 2) may be considered as a good example of the general pattern of spatial variability.

Spatial variability in salinity measured in the Términos Lagoon in 2010 during the dry (left) and rainy (right) seasons (modified from Fichez et al., 2017).
Lower salinities are generally measured near Palizada River, with Candelaria and Chumpan Rivers contributing to a lesser extent to freshwater inputs to the lagoon. The highest salinities in the range of 33 to 36, similar to those of adjacent marine waters, are measured close to Puerto Real Inlet and, according to the general hydrodynamic pattern, the more marine water bodies tend to travel to the southwest along the coast of Carmen Island. Salinity varies considerably within the lagoon as a function of the imbalance between those two sources of input.
Quite surprisingly, the trophic status (dissolved inorganic and organic material and particulate organic material) of the Términos Lagoon has been poorly documented. Silicate concentrations generally correlate to salinity as freshwater transports huge quantities of silica originating from soil leaching. In the Gulf of Mexico, marine offshore waters have silicate concentration of roughly 2 μM, whereas coastal waters are generally in the range of 20–40 μM. Concentrations in the lagoon are around 70 μM on average with strong temporal and spatial variability, as for salinity.
Inorganic nitrogen concentrations generally come within the 0 to 4 μM range with some very occasional peaks close to or above 10 μM near the Palizada River. Concentrations in the middle of Términos Lagoon are — in most cases — significantly below 1 μM. Nitrate + nitrite concentrations are higher during the wet season than during the dry season.
Phosphate (orthophosphate) concentrations come within the 0 to 1.00 μM range with an average value of roughly 0.13 μM. It is clear from the recent study by Conan et al. (2017) that phosphate distribution in the lagoon is for the most part (spatially and temporally) disconnected from nitrogen. During their study, the authors identified the Palizada River and Puerto Real Inlet as the two main sources of NO3 and NH4, whereas PO4 originated from Candelaria and Chumpan R. inputs, and primarily from the mineralization of organic phosphorus (PP and DOP) by bacterial activity. This impacted the stoichiometry of particulate organic matter (N:P ratio) throughout the lagoon.
When averaged over the whole lagoon, the dissolved inorganic N:P ratio ranges from 3:1 to 18:1 with an average value of 9:1, thus suggesting that nitrogen is the most probable element to limit pelagic primary production. However, ratios well over the 16:1 Redfield Ratio and as high as 353:1 were occasionally measured (mostly close to the Palizada River mouth), demonstrating that such an assumption must not be applied uniformly to the whole lagoon system.
On average, concentrations in dissolved organic nitrogen and phosphorus are around 15 and 0.6 μM, respectively. Some strong spatial variation can be observed with occasional high concentrations in the northeastern part of the lagoon, possibly due to the export of organic compounds from the mangrove. On average, concentrations in particulate organic carbon, nitrogen, and phosphorus are around 70, 10, and 0.5 μM, respectively.
Chlorophyll a as an indicator of phytoplankton biomass and potential eutrophication process has been more extensively documented. Average concentration in the lagoon, as reported by various authors, ranged broadly between 1 and 10 mg·m−3, with the lower values being measured as a whole in Puerto Real and the highest close to the Palizada estuary. Chlorophyll a versus total chloropigment ratio was recorded as being recurrently close to 0.75, demonstrating a relatively healthy population of pelagic primary producers. There is virtually no assessment of primary production barring two publications, which report a gross primary production rate of 478 g C·m−2·a−1 at one station of the lagoon (Rivera-Monroy et al., 1998) and variations from 20 to 300 g C·m−2·a−1 (Day et al., 1982), suggesting a potential shift from oligotrophic to eutrophic conditions.
A recent study on biogeochemical cycling in the Términos Lagoon has shown that the water column behaves globally as a sink, and in particular as a “nitrogen assimilator” due to the elevated production of particulate and dissolved organic matter and the weak exportation of autochthonous matter to the Gulf of Mexico (Conan et al., 2017). The same study has revealed that “bottom-up” control largely accounts for the observed variability in phytoplankton productivity. Phosphates delivered by Palizada River inputs and strong on site bacterial mineralization of organic matter result in maximal phytoplankton production in the western part of the lagoon.
A nutrient and carbon budget calculated by Reyes et al. (1997) and David (1999) showed that the Términos Lagoon is slightly autotrophic (carbon sink) on a yearly basis with a net production rate of +0.2 mol C·m−2·yr−1, even though the authors themselves underlined the fact that such results should be taken with great caution due to the evident gaps in the available datasets.
4 Seagrass
Seagrass meadows are known to play many important ecological roles in estuarine and shallow-water coastal ecosystems: they enhance primary production and nutrient cycling, stabilize sediments, elevate biodiversity, and provide nursery and feeding grounds for a range of invertebrates and fish. (Cullen-Unsworth and Unsworth, 2013; Orth et al., 2010). The highest productivity of seagrasses is during the dry season because of higher water clarity, according to Moore and Wetzel (1988). In the Términos Lagoon, the distribution of the seagrass communities clearly reflects the conditions of water movement, clarity, and water salinity. Seagrasses develop denser meadows along the lagoon shoreline of Carmen Island and in particular in the delta of Puerto Real, where high water transparency and salinities, and high percentage of calcium carbonate in the sediments, have been measured (Calva and Torres, 2011).
The communities are dominated by the turtle grass Thalassia testudinum and in lower density by Halodule wrigtii (Raz-Guzman and Barba Macias, 2000). H. wrigtii grows in the shallowest areas, while T. testudinum extends down to depths of 3 m (Ortega, 1995). Total primary production of T. testudinum in the Lagoon is estimated at 260 t·a−1 (Moore and Wetzel, 1988). The epifauna associated with the seagrass meadows is abundant and includes epibenthic amphipods and their predators, i.e. crustacean species of Hippolyte and Pink shrimp (Corona et al., 2000; Negreiros-Fransozo et al., 1996).
5 Mangroves
Mangrove forests play an important role in the functioning of tropical coastal systems. A classification of mangrove forest based on the importance of hydrodynamic exchange describes different categories that are present in the Términos Lagoon: (1) riverine tidal forests that receive high inputs of freshwater, (2) fringe forests that are flushed regularly by tide, (3) basin forests that are less regularly flushed, and (4) scrub forests that have very slow flushing and low nutrient levels (Lugo and Snedaker, 1974). The Términos Lagoon is bordered almost completely by extensive mangrove swamps composed by three dominant species: Rhizophora mangle L. (red mangrove), Avicenia germinans L. (black mangrove) and Laguncularia racemosa Gaertn.f. (white mangrove) (Day et al., 1987). These authors measured higher productivity near the river mouths, probably caused by many factors that include lower salinity, higher nutrient inputs, and a lower hydrogen sulfide level due to lower sulfate concentrations in freshwater. Since stresses are lower, productivity is higher, resulting in more available energy for building biomass. The productivity and the extent of mangrove forests in the area are directly related to high fisheries productivity (Yáñez-Arancibia and Day, 1982).
6 Benthic fauna
Besides the importance of benthic diversity that has been underlined in numerous studies (Yáñez-Arancibia and Day, 1988), the benthos in the Términos Lagoon represents a major trophic resource, and plays an important role in the biogeochemical budget of such a shallow system.
Ayala-Castañares (1963) and Phleger and Ayala-Castanares (1971) described the taxonomy and the distribution of foraminifers in the Términos Lagoon. They reported densities in the range of 250–900 ind·10 cm−2, which suggests relatively high rates of organic matter production, probably consecutive to high river runoffs. Their distribution reflects the circulation in the lagoon, with open-gulf foraminifers close to Puerto Real Inlet and along the southeastern side of Carmen Island, while lagoon foraminifers occur close to the Carmen Inlet. A mixed fluvial assemblage was also observed in and near the river mouths.
Marrón-Aguilar (1975) described the systematic and abundances of polychaetes throughout Términos Lagoon, while Reveles (1984), focused on polychaete species associated with seagrass beds of Thallasia testudinum, and Hernandez-Alcantara and Solis-Weiss (1991) examined those found in the mangrove Rhizophora mangle. In the seagrass beds and mangroves, polychaetes dominated in both abundance and species diversity. Their distribution is closely linked to the salinity gradient, turbidity, and sediment types, and three assemblages may be identified: a first group, localized in the eastern part of the lagoon, is mainly characterized by families Spionidae and Cirratulidae. The second group is located in the central part and the South of the lagoon, with families Cirratulidae and Lumbrinereidae, while the third group, located close to Carmen Island, is characterized by families Capitellidae and Nereidae.
Mollusks have received extensive attention due to the exploitation of two bivalve species. García-Cubas (1988) published a review of all the molluscs encountered in this lagoon, and several studies have been carried out on the exploited bivalves Rangia cuneata and Crassostrea virginica, mainly concerning their distribution (Ruiz, 1975), the physiology of their reproduction (Rogers-Nieto and Garcia-Cubas, 1981) as well as the behavior of larval stages (Chávez, 1979).
Based on previous works, García-Cubas (1988) identified four typical subsystems in the Términos Lagoon: (a) interior lagoon (western part of the area, but with limited connections to Términos Lagoon–salinity lower than 10 in summer periods) associated with rivers and characterized by three bivalve species of commercial interest (Rangia flexuosa, R. cuneata and Polymesoda caroliniana); (b) lagoons joined to the southwestern part of the Términos Lagoon where naturally occurring reefs of Crassostrea virginica and their typically associated community are found (salinity in the range 0–15); (c) the central basin of the main lagoon, where eight gastropod species and nine bivalve species constitute the mollusk community and are submitted to a wide range of salinity throughout the year (10–36); (d) an area of strong marine influence located close to Carmen Island (salinity ranging from 28 to 38), where a broad community of eight gastropods and nine bivalves dominates, mainly in association with Thalassia testudinum beds.
Additional extensive studies have been conducted on fish distributions in the lagoon. Most fish have a direct trophic link with benthic macrofauna (Yáñez-Arancibia and Day, 1988), even if only 10% of the species are permanent residents in the lagoon (45% use the lagoon as a nursery, and 45% are occasional visitors). While half of the fishes are primarily carnivorous, a quarter are higher carnivorous and the last quarter is constituted of herbivorous, detritivorous or omnivorous fishes. A maximal juvenile flow into the lagoon was recorded in September–November, mainly via the Puerto Real inlet.
As described above, the diversity of benthic communities is strongly structured by the balance between marine and riverine inputs and this structuration strongly influences the benthic metabolism and its coupling with the biogeochemistry of the water column.
7 Benthic metabolism
Dissolved and particulate exchanges between sediments and the water column remain poorly documented, and most budgets are based on the assumption that the benthic boundary layer is in balance (David, 1999; Gómez-Reyes et al., 1997). Studies of sediment-water exchanges of nutrients remain poorly documented, except in the mangrove forest (Day et al., 1996; Rivera-Monroy et al., 1995) or seagrass beds of Thalassia testudinum.
In a fringe mangrove, sediment and nitrogen exchanges at the sediment–water interface have been estimated using a 12-m-long flume extended through a fringe forest from a tidal creek to a basin forest. Rivera-Monroy et al. (1995) demonstrated that the tidal creek was the main source of ammonium (0.53 g·m−2·a−1) and nitrate and nitrite (0.08 g·m−2·a−1), while the basin forest was the principal source of total suspended sediments (210 g·m−2·a−1). On the contrary, a net export of particulate nitrogen occurred from the fringe forest to the tidal creek (0.52 g·m−2·a−1), while less particulate nitrogen was exported to the basin forest (0.06 g·m−2·a−1). They also demonstrated that the exchanges were highly variable with seasonal weather forcing, as salinity (hence nutrient concentrations) in the creek was influenced by inputs from rainfall and river discharge to the lagoon. Moreover, as the carbon to nitrogen ratio of particulate matter exported during ebb tides was generally higher than particulate matter imported into the forest during flooding, they suggested that there is a greater nitrogen loss during ebb tide caused by the export of nitrogen-deficient detritus from fringe and basin mangroves.
Several works describing nutrient dynamics have been published on Thalassia testudinum seagrass beds located off the inner littoral of Carmen Island (Hopkinson et al., 1988; Kemp et al., 1988; Stevenson et al., 1988). Rates of NH4 regeneration in sediments of T. testudinum beds were ten times higher in surficial sediments (0 to 2 cm) than at depth (18 to 20 cm). Turnover-time for ammonium pools in surface sediments were about 1 day. Both anaerobic decomposition and denitrification are important biogeochemical processes in Términos Lagoon seagrass beds and rates of ammonium regeneration were sufficient to supply > 70% of the nitrogen required for seagrass growth in this system (Kemp et al., 1988). Measurements of nitrogen uptake in intact cores showed low rates ranging from 0.8 μmol N·m−2·d−1 in February (‘Nortes’) to 50 μmol N·m−2·d−1 in August (rainy season). Separate nitrogen uptake by leaf, root, rhizome, and sediment components measured in small serum bottles suggest that N fixation provides 10 to 40% of the nitrogen demand of the seagrasses (Stevenson et al., 1988). The highest uptake rates occurred just prior to the Thalassia testudinum production peak in February (Rojas-Galaviz et al., 1992). Measurements of stocks of organic and inorganic nitrogen in sediment, water and the biota indicates that biotic stocks of 13,220 μmol·m−2 dominated abiotic stocks of 19 μmol m−2 of nitrogen in the Thalassia system, with less than 0.2% of the nitrogen being in the inorganic form (Hopkinson et al., 1988). A large percentage of the total organic nitrogen pool (94%) is contained in dead material (746 μmol·m−2 versus 12,610 μmol·m−2, living and dead material, respectively) and 97% of the organic nitrogen is located in the sediments, as opposed to the water column. Approximately 75% of the inorganic nitrogen is in the sediments, as opposed to the water column (Hopkinson et al., 1988), while inorganic nitrogen uptake requirements are 7.5, 2.5, and 4.0 mmol N·m−2·d−1 for phytoplankton, epiphytes, and T. testudinum, respectively. The nitrogen turnover times ranged from less than 1 day for inorganic nitrogen in the water column to over 3000 days for sedimentary organic nitrogen (Hopkinson et al., 1988).
8 Drivers of environmental change
The Términos Lagoon and its surrounding wetlands are oppositely impacted by their conservation status and by human development pressure. Urbanization, wastewater discharge, industrialization, alteration of the hydrologic regime, agricultural and cattle production, petroleum extraction and fishing have been exhaustively addressed as the most important deleterious issues (Robadue et al., 2004). Land cover is one of the most systemic indicators of environmental change in the protected area surrounding the Términos Lagoon. Between 1974 and 2001, 31% of the area was detrimentally affected, which results mainly from the replacement of tropical forest and mangroves by grassland and urban areas (Soto-Galera et al., 2010).
Between 1986 and 2001 however, the protected status of Términos Lagoon prompted some positive action in terms of environmental restoration and protection, allowing one to reduce losses in natural habitats by half and demonstrating the potential of efficient remediation strategies. A mangrove restoration experiment headed by the Gulf of Mexico Large Marine Ecosystem Based Assessment and Management Project (GEF LME-GoM) identified increasing salinity in the porewater of mangrove soils as responsible for the most long-term deleterious effects and defined remediation based on hydrological restoration and local community involvement, which yielded an inversion in the recessing mangrove dynamics (Zaldivar-Jimenez et al., 2017).
Currently, the fluvial-lagoon systems of the Términos Lagoon are disturbed. According to Herrera et al. (2002) and EPOMEX (2002), the zones having registered the most change are the river-mouths of the three main rivers (the Candelaria, the Chumpan, and the Palizada), due to excessive inputs of terrigenous materials and growth of oyster reefs. According to Márquez-García (2010), about 30% of the lagoon is in the process of sediment deposition, which means that the lagoon is facing a major sediment accumulation problem. The formation of an internal delta at the mouth of Puerto Real involves the retention of sandy sediments in the Términos Lagoon due to the decrease in current velocity within the system. Conversely, the processes of erosion–deposition in the lagoon generate changes in the depth of the system and erosive morphology of some parts of the coastal area. This can lead to various problems ranging from the siltation or death of seagrasses to constraints in small boat navigation. Approximately 42 m of beach have been lost in the eastern side of the Puerto Real Inlet, a process aggravated by poorly engineered breakwaters that partially disrupt littoral transport (Màrquez-Garcìa et al., 2013). These changes primarily in the east part of the lagoon have likely transformed the biogeochemical characteristics of the sediments, thus impacting the distribution of benthic species and lagoon biodiversity.
Significant changes in fish population have been reported since 1980, including combined alterations in both fish taxonomic and fish functional diversity, with decreases of 41% and 58% in fish abundance and biomass, respectively (Abascal-Monroy et al., 2016; Ramos-Miranda et al., 2005a, 2005b; Sirot et al., 2015, 2017; Villéger et al., 2010). Such changes in biota have been tentatively linked to a loss of favorable habitats and a sustained increase in salinity due to decreasing river inputs to the lagoon (Sosa-Lopez et al., 2007). However, such a sustained trend in salinity increase is supported neither by long-term surveys of rainfall and river discharge nor by modeling outputs that foresee stability in rainfall during the first half of the 21st century. The observed differences have been related to strong interannual variability in runoff, potentially related to ENSO conditions (Fichez et al., 2017). It is true, however, that the very same climate change modeling approaches foresee reductions of rainfall by 5 to 30% in the region during the second half of the 21st century (Biasutti et al., 2012; Hidalgo et al., 2013), corresponding to an overall reduction in runoff of up to 80 percent in the central Yucatan Peninsula and Guatemalan highlands, and of 20 percent in the Veracruz, Tabascan and Campeche lowlands (Imbach et al., 2012; Kemp et al., 2016). Such alteration in hydrologic regime will dovetail with sea level rise, leading to flooding of the most coastal lowlands including the Términos Lagoon, whose surrounding wetlands have been identified as some of the most critical areas in the region in terms of submersion risk and flooding (Ramos-Reyes et al., 2016).
Considering pollutants, the highest dissolved total Polycyclic Aliphatic Hydrocarbons (PAH) concentrations were reported from the Carmen Inlet, whereas water masses close to the Palizada River estuary supported an abundant bacterial community of PAH degraders (Conan et al., 2017). PAH concentrations in oyster tissues were reported to range from 2.5 to 42.5 μg·g−1 (Norena-Barroso et al., 1999). The predominance of low and medium molecular weight alkylated compounds over their parent compounds indicated the petrogenic source of these PAHs, hence pointing to offshore oil activities as a major source of PAHs inputs. A study in the western part of Términos Lagoon showed that PAH concentrations in fish flesh exceeded the maximum values recommended by international regulations (greater than 40.0 μg·g−1 for the epibenthic Petenia splendida) (Orozco-Barajas et al., 2014).
Chlorpyrifos was detected in the water at concentrations up to 72 pg·l−1, and amongst organochlorine compounds, PolyChloroBiphenyl (PCB) averaged 1177 pg·l−1 and DDT 279 pg·l−1, respectively (Carvalho et al., 2009a). Residues of chlorinated compounds were present in both the sediments and the biota, with DDT averaging 190 pg·g−1 and 5876 pg·g−1 in sediments and oysters, respectively. Concentrations of residues did not reach alarming levels and were in fact lower than those reported for other coastal lagoons of the region (Carvalho et al., 2009b). The Palizada River is by far the major contributor (85% to 99% depending on the considered metal) of riverborne metal inputs to the lagoon (Páez-Osuna et al., 1987). Reported concentrations of total metals in lagoon water were higher than in pristine environments (Vazquez et al., 1999), but not very different than in other coastal waters. The authors related the highest metal concentrations in the lagoon to anthropogenic inputs from rivers as well as to more diffuse atmospheric input from the nearby petroleum industry, but no direct evidence was provided.
Comparisons between metal concentrations in the oyster Crassostrea virginica with previous data (Vazquez et al., 1993) showed that, whereas zinc concentrations remained virtually unchanged since the mid-1970s, the levels of cadmium decreased and concentrations of copper and lead increased significantly. In a recent paper dealing with the study of the effect of various contaminants on the oyster C. virginica (Gold-Bouchot et al., 1995), the authors concluded that the pollution level in the Términos Lagoon could be considered as moderate.
Long-term effects of pesticides and heavy metals were studied in aquatic mammals inhabiting the Términos Lagoon. Heavy metals and pesticides in bottlenose dolphins (Tursiops truncatus) were present in skin and blubber biopsy samples, albeit in lower concentrations than those recorded in other studies (Delgado-Estrella et al., 2014).
9 Conclusions and recommendations
The great variability and heterogeneity in the numerous variables that control and characterize transitional water systems greatly hinder the potential for inter-comparison between very diverse sites, because tropical coastal lagoon systems are driven, for example, by very specific factors when compared to temperate ones (Camacho-Ibar and Rivera-Monroy, 2014; Ortiz-Lozano et al., 2005). While most of the coastal lagoons in the world are more or less exposed to the same set of detrimental factors arising from human development and sea level rise, global climate change impacts can, on the other hand, be strongly geographically dependent, even at the regional scale. This variability is particularly strong in the Gulf of Mexico and the Central American region, and joint simulations foresee an increase in the mean annual discharge of the Mississippi–Atchafalaya River by 11 to 63%, compared to a severe decrease by up to 80% for the Usumacinta-Grijalva River (Kemp et al., 2016). Such a contrasting forecast regarding the two major watersheds of the Gulf of Mexico is of major consequence to their respective downstream coastal systems and prefigures a drastic environmental shift in the Términos Lagoon.
Considering the sensitivity of the biota to the balance between riverine and marine influences, salinity is obviously an essential indicator of the main alteration process that will impact the Términos Lagoon system on a long term and global scale. In the absence of a structured environmental monitoring plan focusing on the hydrology of the Términos Lagoon, in particular, urgent action should be taken to gather information on the ongoing trends in environmental change. Indeed, the previously exposed arguments demonstrate that salinity is the most obvious basic indicator of environmental change to be continuously measured in various representative parts of the lagoon.
Larger scale studies are required to more precisely capture the dimensions and nature of the changes produced and their effects on the structure and function of the landscape. Additionally, deeper examination of the history of land use should be conducted to better interpret elements concerning the causes of changes and modifications occurring in the area. Regional trends over the past 30 years have replaced forests with pastures, and there is a call today for more efficient and proactive conservation policies through paid environmental services.
Regarding pollutants, environmental threats seem to be primarily localized around urban areas and river mouths, or restricted to specific ecosystem components, especially in the highest trophic levels. Furthermore, studies on emerging pollutants are entirely absent in the Términos Lagoon and are urgently needed.
Finally, even if several compartments of the benthic ecosystem have been studied, some topics remain to be assessed, such as sediment–water exchanges of nutrients and benthic carbon mineralization in the lagoon, as well as the processes related to microphytobenthic productivity. Such information may be of prime importance in understanding the functioning of the Términos Lagoon and for calculating accurate budgets to preserve this protected but threatened ecosystem.
Acknowledgements
This study was financially supported by the “Institut de recherche pour le développement” (IRD), the “Centre national de la recherche scientifique” (CNRS), the “Université de Lille”, the “Universidad Autónoma Metropolitana–Iztapalapa” (UAM–I) and the French National Program “Écosphère continentale et côtière–Dynamique et réactivité des interfaces littorales” (EC2CO-DRIL). Acknowledgements are also due to the “Instituto de Ciencias del Mar y Limnologia, Universidad Nacional Autónoma de México” (ICMyL-UNAM) for providing access to their field station in Ciudad del Carmen. French Bi National collaboration was federated by ECOS-Nord (ECOS/ANUIES/SEP/CONACyT contract M12-U01) and Danone-Bonafont S.A. de C.V.


