1. Introduction
Iron is widely recognized for its crucial role in the functioning of terrestrial and marine ecosystems (Raiswell and Canfield, 2012; Y. S. Lin et al., 2025). However, in the marine environment, iron can only influence productivity and the functioning of the food chain if it can participate in (bio-)chemical reactions. This is what can be called reactive iron. Iron is not reactive if it is present, that is, locked, within the crystal lattices of minerals, depending on the mineral solubility (e.g., Raiswell and Canfield, 1998). Located inside the crystallographic network of a lowly soluble mineral such as pyrite, iron cannot participate in the (bio-)chemical reaction. Iron, which is « trapped » within the structure of minerals, can still be released and become reactive if the crystal lattices that contain it are disrupted during diagenesis or alteration. However, this transformation can be quite time-consuming, ranging from a few days to a much longer period, even a million years (e.g., Canfield and Raiswell, 1999; Raiswell and Canfield, 1998; Raiswell and Canfield, 2012). In marine environments, particularly continental shelf environments, reactive iron inputs originate from the emerged landmasses. These inputs are carried by rivers and winds (e.g., Chester and Jickells, 2012, and references therein). Fluvial (river-borne) iron is in the form of organometallic and clay-humic complexes from which it can easily be released. Aeolian (airborne) iron is primarily adsorbed onto the surface of clay minerals and is carried away by the winds, especially from desert regions worldwide (Chester and Jickells, 2012; Vosteen et al., 2024). In proximal environments (shoreface and upper offshore), aeolian inputs are often masked by fluvial inputs, the fluxes of which are generally much higher. Aeolian inputs should not, however, be forgotten or neglected. Once introduced into the marine environment, iron can present two redox states, Fe2+ or Fe3+, and its solubility depends on the oxidation state. Several factors contribute to immobilizing the iron present in the water column and bringing it to the sediments: it can form poorly soluble oxides and oxyhydroxides (and among them, magnetite); it can react with sulfide ions, and form pyrite, directly or with intermediate phases; it can also be incorporated into more complex authigenic minerals such as phyllosilicates, and, in particular, glauconite. Our recent work (Tribovillard, Averbuch, et al., 2025) examined both glauconite ((K,Na)2(Fe3+,Fe2+,Al,Mg)4[Si6(Si,Al)2O20](OH)4) and magnetite (Fe3O4). Both minerals contain Fe(II) and Fe(III). Our result have shown that glauconite and magnetite can form early and very rapidly in shallow environments where the abundance of reactive iron was not a limiting factor and where redox conditions allowed iron to be present in both its valences (Tribovillard, Bout-Roumazeilles, Abraham, et al., 2023; Tribovillard, Bout-Roumazeilles, Guillot, et al., 2023; Tribovillard, 2024). We will describe as weakly reducing such conditions where iron is not entirely present in its reduced form. Reciprocally, environments in which reactive iron is exclusively present in its reduced form will be designated as strongly reducing. Our work has shown that authigenic, iron bearing minerals formed in shoreface environments (such as beach sands) can disappear during subsequent stages of diagenesis if nothing protects them. Nevertheless, early carbonate cementation can typically protect glauconite and magnetite from the expected usual diagenetic evolution, namely, dissolution or transformation into stable structures such as pyrite (Tribovillard, Averbuch, et al., 2025; Tribovillard and Bout-Roumazeilles, 2025). In the present study, we explore the authigenic trapping of iron in the form of magnetite, still in continental shelf deposits, but in deeper environments (lower offshore). We have identified Jurassic sedimentary deposits from the Boulonnais (northernmost France) combining the presence of reactive iron and weakly reducing redox conditions at the time of deposition. We isolated magnetite from these rocks and observed it using electron microscopy and Raman spectroscopy. Our goal was to provide answers to the following questions: why is authigenic magnetite not more frequently observed in sedimentary rocks, whereas glauconite is relatively common? What is the role or place of authigenic magnetite in the sedimentary iron cycle? What does the presence of magnetite mean with regard to paleoenvironmental reconstruction?
2. Material and methods
2.1. Material
To study the distribution of authigenic or terrigenic (inherited) magnetite, the geological formation called the Assise de Croï was chosen. This formation is visible on the cliff on both sides of the town of Wimereux, wich t is to say to the north and to the south, and the finest outcrop, from a visual point of view, is to the south of Wimereux, near the small promontory called Pointe de la Crèche (Figure 1). This formation, also called Argiles et Calcaires de la Tour de Croï (Deconinck, Geyssant, et al., 1996; Mansy et al., 2007; Deconinck and Baudin, 2008) is of Tithonian age (albani and glaucolithus ammonite zones) and appears in the form of alternating limestone beds and marly interbeds (Figures 1 and 2). Its total thickness is about 8 m; the limestone beds are generally less than 20 cm thick and result from the coalescence of limestone nodules. The marly interbeds are a bit thicker than the beds (Figure 2). The Assise de Croï is made up of sediments deposited under upper offshore conditions (Mansy et al., 2007). The Assise de Croï overlies the Argiles de Wimereux Formation, a marly sequence also reflecting lower offshore depositional conditions (ibid.). It is overlain by the Grès de la Pointe aux Oies Formation, made up with sandstones beds and corresponding to shoreface depositional conditions (Deconinck, Baudin and Tribovillard, 2000).
Location of the outcrop shown on Figure 2, with a schematic stratigraphic log of the topmost part of the Jurassic deposits of the Boulonnais region. Maps from the Géoportail website of the French Institut national de l’information géographique et forestière (IGN).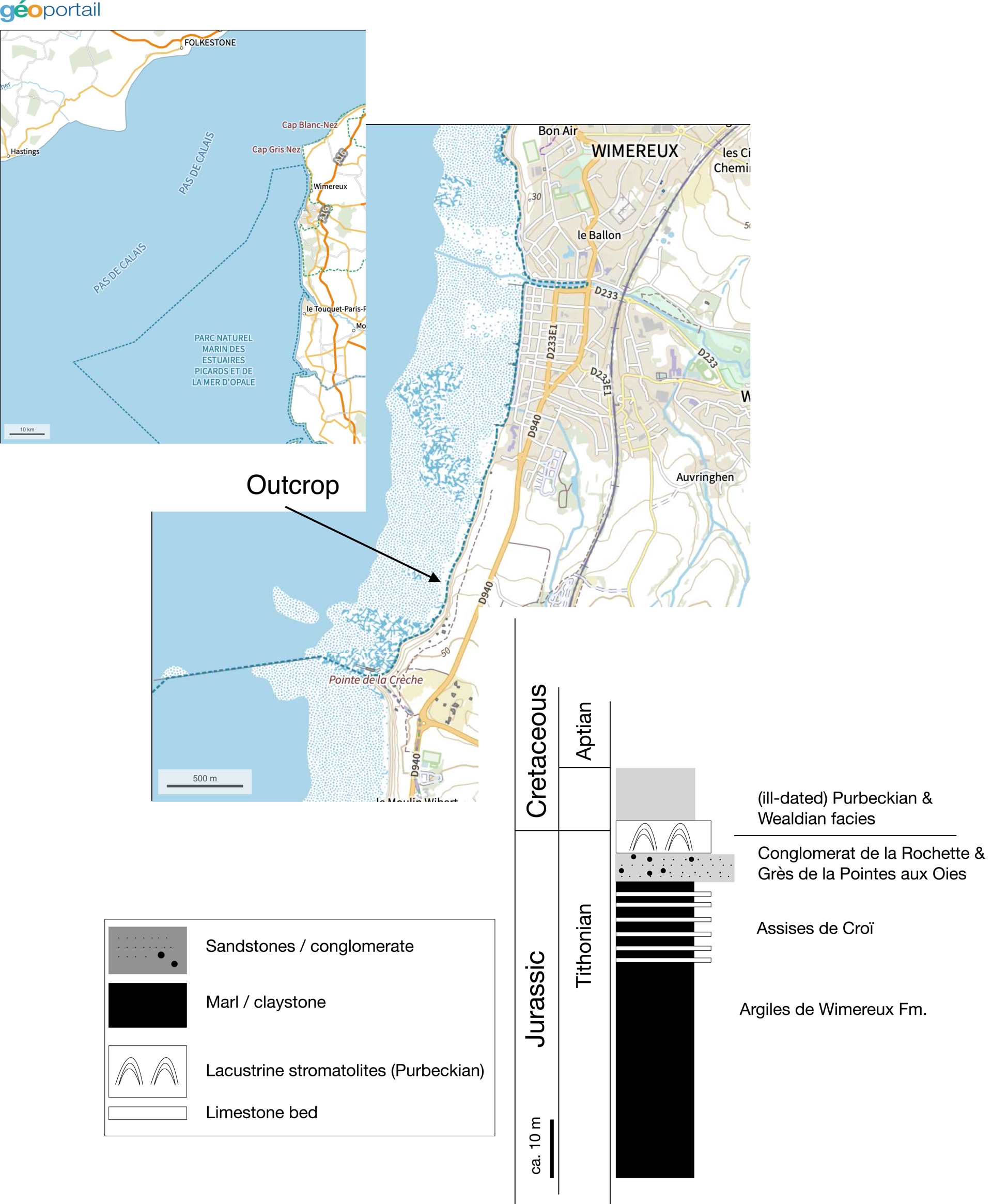
(A) The topmost part of the Jurassic deposits exposed along the cliff between the Cap de la Crèche and Wimereux city (Figure 1). (B) Close up view of the alternation of carbonate beds and marly interbeds of the Assise de Croï Formation.
Whether calcareous or marly, the Assise de Croï deposits are bioturbated and rich in bivalve bioclasts, most often Nanogyra and Trigonia. They are also relatively rich in glauconite. The work of Tribovillard, Bout-Roumazeilles, Guillot, et al. (2023) showed that the nodular beds were of early diagenetic origin and that the glauconite they contained was authigenic and even earlier than the calcareous nodules. The geochemical study by Tribovillard and Bout-Roumazeilles (2025) showed that the enrichment of trace elements markers of redox conditions was modest and that the depositional conditions were weakly reducing. The presence of authigenic glauconite is consistent with these results, since this mineral, which contains iron in its two oxidation states, can only form under weakly reducing conditions. Moreover, the presence of this mineral shows that reactive iron was not a limiting factor at the time of deposition (Tribovillard, 2024; Tribovillard, Averbuch, et al., 2025). All these reasons explain why the Assise de Croï was chosen to study the magnetite content, whose authigenic formation conditions are similar to those of glauconite.
The samples examined for the present work are the same as those studied in Tribovillard, Bout-Roumazeilles, Guillot, et al. (2023) and Tribovillard and Bout-Roumazeilles (2025). The sample set was collected from eight bed-interbed pairs in the lower part of the Assise de Croï, north of Wimereux, where the outcrop conditions are propitious to sampling.
2.2. Methods
The limestone or marl samples were crushed using a jaw crusher to ensure that the maximum fragment size was only a few millimeters. The analytical protocol is intended to concentrate iron-bearing minerals, as well as other non-carbonate fractions (quartz, heavy minerals, clay minerals, …). Poulton and Canfield (2005) investigated the sequential extractions that could be employed to analyze the distribution of iron among various mineral phases. Notably, the authors emphasize that the dissolution of carbonate while preserving magnetite necessitates the use of acetic acid instead of hydrochloric acid. Here, we used dilute acetic acid (10%) to remove the carbonate fraction without damaging the magnetite grains (as evidenced through microscope observation. The samples were then rinsed and dried, and the magnetite grains were isolated by magnetic separation. We used a Franz Isodynamic magnetic separator applying the protocol described in Tribovillard, Averbuch, et al. (2025). The particles were imaged using a scanning electron microscope (SEM) equipped with an EDS-type analytical probe (FEI Quanta 200 SEM equipped with a Quantax QX2 Roentec energy-dispersive X-ray spectroscopy system). To determine or confirm the mineralogical composition of magnetite and other accessory minerals, Raman microspectroscopy was performed with a JY Horiba, Labram HR800UV, equipped with an electronically cooled charge-coupled device (CCD) detector, using 532 nm laser excitation. Spectra was obtained using 100× magnification objective.
3. Results
Magnetite grains are present in both facies of the Assise de Croï, i.e., nodular limestone beds and marly interbeds. However, the abundance of these grains is very low (a few grains out of 50–100 g of rock) and could not be quantified. It should be reminded that these two facies are also rich in glauconite (Tribovillard, Bout-Roumazeilles, Abraham, et al., 2023; Tribovillard, Bout-Roumazeilles, Guillot, et al., 2023), it will be discussed below.
3.1. Authigenic carbonate beds
The nodular carbonate beds contain two types of magnetite grains: (1) relatively small grains with euhedral morphology (Figure 3) and, (2) to a lesser extent, larger, rounded grains showing signs of wear (Figure 4). The euhedral grains are made of nickel- and chromium-rich magnetite and contain apatite inclusions (Figure 3). As regards the rounded grains, they do not contain apatite inclusions, nor do they show enrichment in nickel or chromium, but they are very clearly enriched in titanium. These are titanomagnetite grains.
Small dimensioned, euhedral grains of authigenic magnetite. (A) Picture after photonic microscope observation, (B, C) scanning electron microscope (SEM) images.
Large dimensioned, rounded grains of terrigenic magnetite. (A) Picture after photonic microscope observation; (B–D) SEM images of magnetic grains, commonly yielding the typical lattice structures observed with magnetite from igneous rocks (D).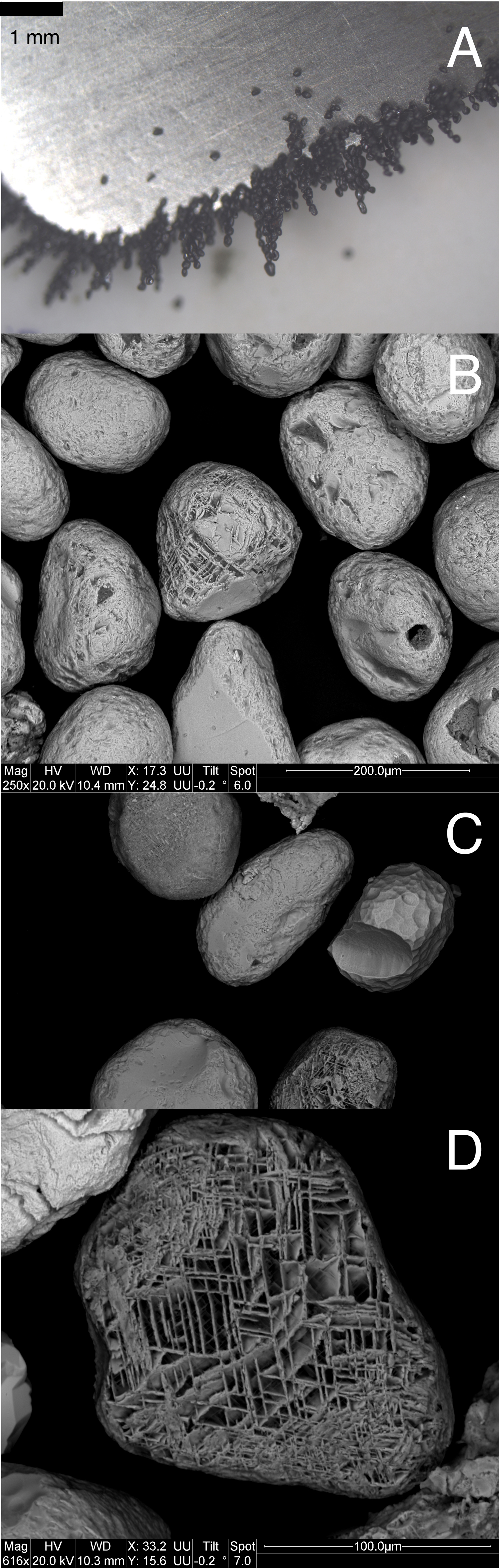
3.2. Marlstone interbeds
The marly interbeds contain only titanomagnetite grains, which also show signs of wear. No euhedral magnetite was observed in the interbeds.
To summarize, worn titanomagnetite grains are present both in the beds and interbeds, but in addition, the beds also contain small euhedral magnetite grains showing apatite inclusions and often enriched in nickel and chromium.
4. Interpretations
4.1. Two types of magnetite grains
Our observations allow two types of grains to be identified. The largest ones are made up of titanomagnetite and show traces of wear. Titanomagnetite is common in igneous (and metamorphic) rocks (strictly, it is a homogeneous cubic solid solution of ilmenite and magnetite; e.g., Moskowitz et al., 2015). Consequently, we will consider that, in the present case, the titanomagnetite grains are of terrigenous origin, consistent with their round-shaped morphologies.
Reciprocally, the small euhedral grains will be considered to be authigenic. They are too large to be interpreted as deriving directly from bacterial activity (magneto-tactic bacteria): bacterially secreted biogenic grains are reported to be sub-micrometric in scale (e.g., Z. Lin, Sun et al., 2020; Yamazaki, 2020; Mathon et al., 2024). However, bio-induced magnetite resulting from the activity of dissimilatory iron-reducing bacteria may be micrometric or pluri-micrometer large (Z. Lin, H. Lin, et al., 2024). This size range is compatible with what is observed in the present work. Therefore, terrigenic magnetite is present everywhere in the Assise de Croï, whereas authigenic magnetite is present only in the carbonate nodular beds.
4.2. Reconstruction of the authi-diagenetic sequence
The deposits of the Assise de Croï, whatever their facies—either diagenetic carbonate beds or marly interbeds, contain small amounts of terrigenic magnetite, closer to titanomagnetite than to magnetite stricto sensu. This terrigenic (titano)magnetite accompanies quartz grains that are also terrigenic (Tribovillard, 2024), and it was not completely dissolved during later stages of diagenesis. What does this tell us about paleoenvironmental conditions?
Magnetite and titanomagnetite are minerals relatively sensitive to reductive dissolution (Nowaczyk, 2011). This is to be ascribed to the redox variations of the iron oxidation state. According to the classical reaction succession of early diagenesis associated with organic matter remineralization (Figure 5), iron reduction (from Fe3+ to Fe2+) starts after the sequential progression from aerobic respiration, to nitrate reduction (a.k.a. denitrification) and to manganese reduction (B. B. Jørgensen, 2006; B. B. Jørgensen, 2021; Burdige, 2006; Adhikari et al., 2017). Below the iron redox boundary, where iron reduction begins, magnetic mineral dissolution may occur (Canfield and Berner, 1987; Yamazaki, 2020), as the following equation may illustrate it:
| \begin {equation}\label {eq1} \mathrm {Fe}_{3}\mathrm {O}_{4} + \mathrm {H}_{2} + 3 \mathrm {H}_{2}\mathrm {S} \rightarrow 3 \mathrm {FeS} + 4 \mathrm {H}_{2}\mathrm {O} \end {equation} | (1) |
| \begin {equation}\label {eq2} 2 \mathrm {Fe}_{3}\mathrm {O}_{4} + \mathrm {CH}_{2}\mathrm {O}+ 11 \mathrm {H}^{+} \rightarrow 6 \mathrm {Fe}^{2+} + \mathrm {HCO}_{3}^{-} + 6 \mathrm {H}_{2}\mathrm {O} \end {equation} | (2) |
This figure summarizes the main stages of remineralization of organic matter in marine sediments. This process involves aerobic and anaerobic respiration, fermentation, and finally, methanogenesis (left-hand side of the figure). In particular, under anaerobic conditions, the reduction of sulfate, iron oxyhydroxides, and manganese oxides can be replenished with electron acceptors by the reactions illustrated on the right-hand side of the figure. The former process consumes organic matter, but the latter does not, although these reaction (not involving organic-matter consumption) do prolong this anaerobic respiration stage by regenerating electron acceptors. These repeated cycles of iron reduction and oxidation allow magnetite to form and disappear in successive loops, a phenomenon referred to here as the cryptic iron cycle. This cycle also allows the formation of glauconite, which does not disappear as easily because this mineral is less labile than magnetite under reducing conditions. This figure is developed and enriched from that of Schulz and Zabel (2000).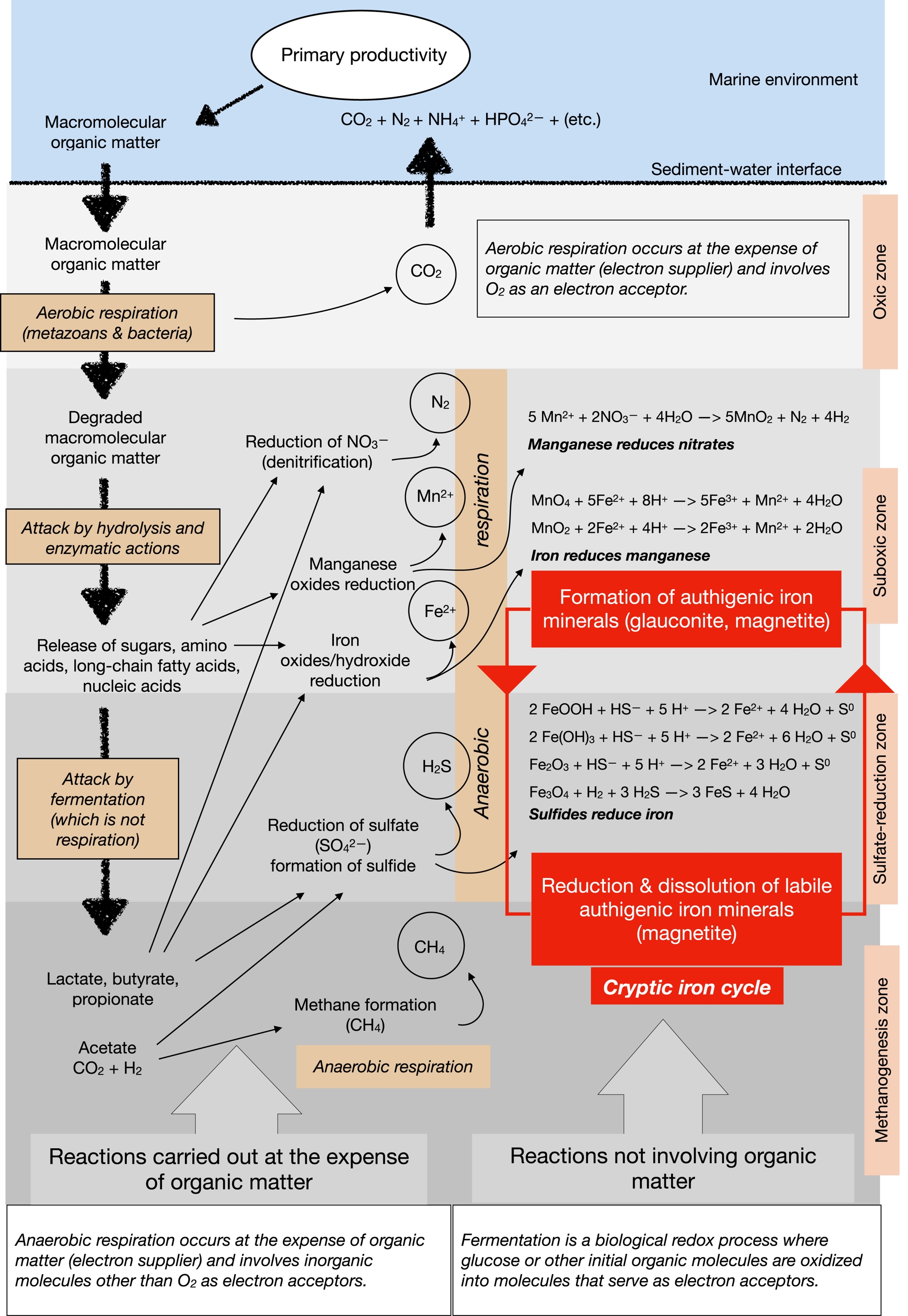
In the situation examined in this paper, the presence of terrigenic magnetites indicates that the sediments of the Assise de Croï did not undergo sustained or intense sulfidic diagenesis, as also evidenced by the low enrichment of the rocks in trace metal elements that are sensitive to reductive conditions (Tribovillard, Bout-Roumazeilles, Abraham, et al., 2023; Tribovillard, Bout-Roumazeilles, Guillot, et al., 2023; Tribovillard and Bout-Roumazeilles, 2025). The sediment was therefore not subjected to strongly reducing conditions during the early diagenesis; otherwise any form of magnetite would have disappeared.
In addition, the carbonate beds have preserved intact authigenic magnetite minerals, which are even more fragile than the inherited magnetite. However, these minerals are absent from the marly interbeds. This suggests that the conditions for preserving labile magnetite were not gathered in the marly interbeds, or that magnetite could not have formed there during early diagenesis. Since authigenic glauconite, another iron-bearing mineral, is present in the interbeds, it can be deduced that reactive iron was present in the pore waters, and that the conditions were favorable for the formation of authigenic minerals mixing Fe2+ and Fe3+. Therefore, it may be deduced that authigenic magnetite could also have formed within the marly interbeds, but that it did not survive the diagenetical conditions. If labile magnetite is still present in the nodular beds, it is because the very early diagenetic growth and cementation of the nodules sealed micro-environments hosting both authigenic and inherited magnetite. The partitioning or closure of the internal environment of the nodules no longer allowing the circulation of fluids, the magnetite was protected against any further evolution, that is to say, from any degradation or dissolution. The early cementation of the limestone nodules effectively « fossilized » the geological conditions at the time of deposition, or shortly thereafter.
5. Discussion
Our interpretations are that the Assise de Croï sediments hosted authigenic magnetite growth during the early stages of diagenesis. This magnetite then disappeared during later stages of diagenesis, likely through reductive dissolution during the sulfidic phase, induced by the decomposition of organic matter (organoclastic sulfate reduction; B. B. Jørgensen et al., 2019). However, this magnetite was preserved in certain horizons where the initial marly deposits were cemented early into carbonate beds made up with coalescent nodules. This indicates that the geochemical conditions were right for the growth of iron-rich minerals to take place. These interpretations are based on the usual conditions leading to the dissolution of magnetite, as well as factors that prevented such destruction, contrary to all expectations. However, it can be instructive to question the conditions of magnetite formation.
5.1. What are the conditions for the authigenic growth of magnetite?
As mentioned above, the growth of iron-bearing minerals such as magnetite and glauconite requires weakly reducing conditions, as well as the presence of reactive iron. The literature provides several scenarios for magnetite (Hu et al., 2025): (1) Iron may be released by the dissolution of pre-existing pyrite or titanopyrite, whose alteration during deposition could provide the required iron (Katz et al., 1998; Nowaczyk, 2011; Badesab et al., 2023). (2) Z. Lin, H. Lin, et al. (2024) observed micrometer-sized euhedral magnetites in sediments impacted by cold methane seeps, with iron released by dissimilatory iron-reducing bacteria. (3) Karlin et al. (1987) identified magnetite as a byproduct of metabolic reactions conducted by microorganisms using iron redox transitions as an energy source. All these scenarios rule out a simpler hypothesis regarding the origin of iron: that reactive iron could be brought to the marine environment in the form of metal-organic or metal-clay complexes, without necessarily resorting to a pre-existing source of iron in the marine environment that needs to be remobilized. In addition, Halevy et al. (2017) and Koeksoy et al. (2019) reported that green rust, a mixed-valence hydroxide that contains carbonate ions in the interlayer, could be a precursor to magnetite. According to these authors, carbonate green rust can form near the iron redoxcline and commonly decomposes into magnetite and/or siderite.
We have no satisfactory means of rejecting or retaining this or that hypothesis, and in this case, it does not matter much. What matters is that authigenic magnetite could have formed. It formed before being embedded in carbonate nodules that have been proven to be very early, because very rapid confinement would have prevented any circulation of mineralizing fluids essential for authigenesis. Therefore, magnetite, like glauconite, already existed when the calcareous nodules formed. This means that at the time of sediment deposition, a certain amount of reactive iron was present in the sedimentary deposits and could have fueled the authigenic growth of magnetite and glauconite. The work of Tribovillard, Averbuch, et al. (2025) reached the same conclusion, but for much more proximal sandy deposits (shoreface sands of the Grès de Châtillon Formation). This study showed that early-cemented sand injectites contained magnetite grains that had formed during the earliest diagenesis. In the present study, the deposits took place on a significantly deeper platform, with of course drastically contrasting depositional conditions. It is concluded that the phenomenon of iron diagenesis goes beyond the coastline alone and can be observed more widely on the continental shelf. This discovery broadens the spectrum of all phenomena involving iron during very early diagenesis in the continental shelf domain.
5.2. The diagenetical transition zone where magnetite can form
When iron is trapped in the form of pyrite, or more broadly iron sulfides, it is relatively tightly retained in a chemically stable medium, which has a low probability of being oxidized later during diagenesis. Indeed, the classic diagenetic sequence, as described in the works of Jørgensen and co-workers (B. B. Jørgensen, 2006; B. B. Jørgensen, 2021; C. J. Jørgensen et al., 2009; B. B. Jørgensen et al., 2019) or that of Burdige (2006), Canfield (2006), leads inexorably from an environment where aerobic respiration dominates to one where reducing conditions prevail and remain unchanged. Re-oxidation of the sedimentary pile can only be caused by phenomena affecting the entire thickness of the water column, simultaneously with the first few decimeters or meters of sediment. These phenomena can be sudden, such as large-scale turbidites, or large-scale, but slow, phenomena such as those associated with sea-level variations. Consequently, the classic sequence of diagenetic phenomena leads to the sequestration of iron in sulfides and authigenic phyllosilicates. What is stressed on here is that this virtually inexorable phenomenon can be significantly slowed by redox loops involving labile minerals, such as magnetite. The same way water can turn into ice and ice melts at 0 °C, magnetite can form or be dissolved at the redox boundary where iron has its tipping point between Fe2+ and Fe3+. In the diagenetical zone above the horizon where sulfate reduction takes place, iron is involved in bacterially mediated reactions of iron oxyhydroxides reduction with organic matter being an electron supplier, as illustrated with Equations (3) and (4).
| \begin {eqnarray} 2 \mathrm {Fe}_{2}\mathrm {O}_{3} + \mathrm {CH}_{2}\mathrm {O} + 3 \mathrm {H}_{2}\mathrm {O} \rightarrow \mathrm {HCO}_{3}^- + 4 \mathrm {Fe}^{2+} + 7 \mathrm {OH}^- \label {eq3} \end {eqnarray} | (3) |
| \begin {eqnarray} 4 \mathrm {FeOOH} + \mathrm {CH}_{2}\mathrm {O} + \mathrm {H}_{2} \mathrm {O} \rightarrow \mathrm {HCO}_{3}^- + 4 \mathrm {Fe}^{2+} + 7 \mathrm {OH}^- \label {eq4} \end {eqnarray} | (4) |
However, iron thus reduced by iron-reducing bacteria (also termed ferri-reducing bacteria) may fuel oxidation reactions mediated by iron-oxidizing bacteria (so-called ferro-oxidizing bacteria) and get back to the Fe(III) state (e.g., Burdige, 2006; Miot and Etique, 2016; Laufer et al., 2016). In addition, H2S released by sulfate reducers may also reduce iron as illustrated by the following equations, because iron is also involved in chemical reactions operating without organic matter acting as an electron supplier:
| \begin {eqnarray} 2 \mathrm {FeOOH} + \mathrm {HS}^- + 5 \mathrm {H}^{+} \rightarrow 2 \mathrm {Fe}^{2+} + 4 \mathrm {H}_{2}\mathrm {O} + \mathrm {S}^{0}\label {eq5} \end {eqnarray} | (5) |
| \begin {eqnarray} 2 \mathrm {Fe(OH)}_{3} + \mathrm {HS}^- + 5 \mathrm {H}^{+} \rightarrow 2 \mathrm {Fe}^{2+} + 6 \mathrm {H}_{2}\mathrm {O} + \mathrm {S}^{0} \label {eq6} \end {eqnarray} | (6) |
| \begin {eqnarray} \mathrm {Fe}_{2}\mathrm {O}_{3} + \mathrm {HS}^- + 5 \mathrm {H}^{+} \rightarrow 2 \mathrm {Fe}^{2+} + 3 \mathrm {H}_{2}\mathrm {O} + \mathrm {S}^{0} \label {eq7} \end {eqnarray} | (7) |
Reciprocally, iron is also oxidized in the presence of nitrate ions:
| \begin {equation}\label {eq8} 10 \mathrm {Fe}^{2+} + 12 \mathrm {H}^{+} + 2 \mathrm {NO}_{3}^- \rightarrow 10 \mathrm {Fe}^{3+} + \mathrm {N}_{2} + 6 \mathrm {H}_{2}\mathrm {O} \end {equation} | (8) |
Therefore, this diagenetic zone, where reactive species such as nitrate, manganese, or iron are present and interact the ones with the others, is the place where magnetite may form because this mineral may result from iron reduction reactions as well as iron oxidation ones (Karlin et al., 1987; Brown et al., 1995; Gibbs-Eggar et al., 1999; Chaudhuri et al., 2001). Lastly, Usman et al. (2012) showed that when ferric minerals, such as ferrihydrite, are aged within a Fe2+ containing media (such as ferruginous pore waters), they readily transform into green rust, which can be a precursor to magnetite, as mentioned above. However, when the sediment gradually passes into the sulfate-reducing zone (organoclastic sulfate reduction or sulfate-dependent anoxic oxidation of methane; B. B. Jørgensen et al., 2019; B. B. Jørgensen, 2021) as a consequence of continuous particle settling, magnetite will definitively disappear because of iron reduction (Canfield and Berner, 1987; Canfield, Stewart, et al., 2010).
5.3. Magnetite at the expense of sedimentary organic matter
Magnetite can form or disappear in the diagenetic zone where conditions easily switch between the “world” of oxidation and that of reduction. Consequently, iron can be trapped or released numerous times before the situation becomes permanently locked in highly reducing conditions. In other words, within this zone, iron is reversibly trapped. It remains reactive, even when transiently trapped in mineral phases because it can be released. Thus, in the sediments of shallow marine environments, Fe remains usable by living organisms and accessible to the chain of reactions that determines the remineralization or conversely, preservation of organic matter as shown in Figure 5. Since these loops keep iron in a reactive state for a relatively long time, it can be remobilized by the action of bottom dwellers, and it can be involved, on the one hand, in chemical reactions mobilizing organic matter as an electron supplier in redox reactions, or on the other hand, in oxidation/reduction reactions not involving organic molecules (Adhikari et al., 2017; Arellano et al., 2019, Figure 5).
5.4. Iron, an agent playing a role in the increased degradation of organic matter
First, iron is known to play a negative, indirect but effective, role in the preservation of organic matter. In a few words, organic matter may be sulfurized (vulcanized) in the presence of sulfide ions, as has been known for decades now (Vandenbroucke and Largeau, 2007; Amrani, 2014; Quijada et al., 2016; Raven et al., 2016; Cohen-Sadon et al., 2025; Ma et al., 2025, to mention a few recent papers). Nevertheless, sulfide ions have a high chemical affinity for reduced iron (Rickard, 2012; Rickard, 2021; Rickard, 2024; Cohen-Sadon et al., 2025; Ma et al., 2025), and when Fe2+ is available in pore waters or even at the sea bottom, sulfide ions are monopolized by reactive iron. If iron is present in abundance, the entire pool of sulfide ions may be caught by iron sulfide formation ; therefore, no sulfurization of organic matter can occur, which facilitates the remineralization of organic products (Tribovillard, Hatem, et al., 2015). Second, before reaching the point where iron is entirely transformed into iron sulfide, Fe can participate in the redox loops described above and fuel reactions that destroy organic matter through iron-mediated anaerobic respiration. Consequently, environments where reactive iron is abundant are unfavorable for the preservation of fragile organic matter. Such milieus are typically shallow environments located near the mouths of large rivers, where iron may be supplied in large amounts (Burdige, 2011; Bianchi, 2011; Bianchi et al., 2016). There, organic matter accumulated in the sediments is mainly (if not exclusively) present in the form of refractory organic products (land-derived woody fragments), because all the labile organic matter would have got lost, as the result of the iron-mediated remineralization reactions, among other processes remineralizing organic matter.
5.5. Magnetite and redox/iron-sensitive trace metals
Following the events occurring with reactive iron, all these diagenetic transformations of iron-bearing phases discussed here can affect trace metal elements, whose behavior is related to that of iron, whether in the form of sulfides or oxides and hydroxides (molybdenum, arsenic, copper, nickel, chromium, antimony, etc.; e.g., Banerjee et al., 2016; Baldermann et al., 2022; Chanda et al., 2025). To explore this aspect, sufficient quantities of authigenic magnetite are required to allow for chemical analysis. So far, we have not been able to achieve this, but it represents a challenge for our ongoing work. The point is to examine possible enrichments in redox-sensitive trace metals within authigenic magnetite grains that offer a double opportunity to capture them: the presence of iron and of (mildly) reducing conditions.
6. The cryptic iron cycle
The same way a cryptic sulfur cycle has been conceptualized by Canfield, Stewart, et al. (2010), a cryptic iron cycle may be put forward here. As explained by Callbeck et al. (2018) and Grijalva-Rodriguez et al. (2025), the cryptic S cycle refers to the simultaneous activity of bacterially mediated, sulfate-reduction and sulfide-oxidation pathways in, for instance, a marine particle aggregate or any other closely defined space. Sulfide produced locally by sulfate-reducing bacteria is immediately reoxidized to elemental sulfur or sulfate ions by sulfide-oxidizing bacteria, and dissolved sulfide remains at submicromolar concentrations. Here, iron is captured in magnetite but the loose capture is most of the time only transient, even if it may be repeated through cycles of capture/release of iron in precipitating/dissolving magnetite. Magnetite can form and dissolve, and form again and dissolve again as long as the hosting sediment is maintained in mildly reducing conditions, that is, in the “oscillatory zone” of diagenesis where iron is cyclically oxidized/reduced. This cyclic, possibly protracted, redox transition can be conducted by bacterially mediated reactions or abiotic ones. Nevertheless, once diagenetic conditions turn to reducing, beyond all return, then magnetite may be quasi-permanently transformed into pyrite (see Brothers et al., 1996, for an exception to the rule). Consequently, the magnetite-driven cryptic iron cycle leaves no trace in the sediments, unless an early-diagenetic cementation occurs (as is the case in the present work), allowing the authigenic magnetite to be preserved and recorded. A consequence of the cryptic iron cycle is to stimulate iron-depending bacterial activity at the expense of labile organic matter in marine sediments. The longer the cryptic cycle, the larger the amount of remineralized, labile, organic matter, and thus, the higher the proportion of recalcitrant (also termed refractory) organic matter in the organic content of the sediments.
7. Conclusions
The examination of magnetite distribution in the Croï Formation, revealing two contrasting facies, provides new information on early diagenesis. The sediments of this geological formation, regardless of their facies, contain detrital magnetite, indicating that the redox conditions throughout early diagenesis were slightly reducing and did not allow for the complete dissolution of detrital magnetite (which is more resistant to reducing dissolution than authigenic magnetite). Authigenic magnetite is found only in the early diagenetic limestone beds that protected the tiny euhedral magnetite grains from reducing dissolution. The early precipitation of a kind of carbonate matrix thus encapsulated and fixed the results of the initial stages of diagenesis, during which magnetite could precipitate and dissolve repeatedly, following a dynamic and cyclic redistribution of iron. The subsequent stages of diagenesis, through sulfate reduction reactions, resulted in the long-term capture of iron as iron sulfide, erasing all traces of the iron’s “versatile life” during its early diagenesis. This turbulent early life, which we refer to here as the cryptic iron cycle, allowed for the remineralization of reactive organic matter by bacteria whose metabolism relies on the reduction or oxidation of iron, thus providing energy to the microorganisms.
The cryptic iron cycle is predictably enhanced when large quantities of reactive iron are introduced into the marine environment: typically off the mouths of major rivers, on the continental shelf. In these environments, the sediment grain size is generally relatively coarse (clay-poor sediments), and the (relatively) shallow depth promotes sediment oxygenation through atmospheric O2 diffusion on the one hand, and through seawater turbulence on the other hand. Consequently, shallow marine environments are not conducive to the development of reducing conditions at the base of the water column. Thus, the significant iron input, combined with slightly reducing conditions, stimulates the cryptic iron cycle, resulting in substantial remineralization of labile organic matter. As a result, the sediments accumulated in these environments primarily reflect the preservation of recalcitrant or refractory organic products.
Disclosure of interests. The author does not work for, consult, own shares in, or receive funding from any company or organization that would benefit from this article, and has disclosed no relevant affiliations beyond his academic appointment.
Acknowledgments
This work has been performed using the Plateform CARMIN – Ulille infrastructure and technical support. Our thanks go to the Programme Tellus Syster of the French Institut des Sciences de l’Univers (INSU) for funding our work. We thank Monique Gentric for the financial/administrative management, as well as Sandra Ventalon and Marion Delattre for the technical support of this project, and lastly, the LOG laboratory and the Department of Earth Sciences of the University of Lille for their support. Lastly, I would like to express my sincere gratitude to the anonymous reviewers of the manuscript for their assistance.
Declaration of interests
The author does not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and has declared no affiliations other than their research organization.






 CC-BY 4.0
CC-BY 4.0