1 Introduction
Polychaetes of the family Sabellariidae build structures, qualified as reefs, which can extend over several hectares. These reefs are known worldwide for their biodiversity. Their associated fauna has been studied on the coasts of India [1], South America [2], Spain and Portugal [3], and western America [4]. With respect to French coasts, Gruet [5–7] and more recently Dubois [8] have provided in-depth analysis of the associated fauna of Sabellaria alveolata reefs. This species is common along European coasts, occurring from Cornwall (Great Britain) [9,10] to the south of Morocco [11]. It builds structures of two major types: small sheet-like reefs adhering to rocks on the upper level of the intertidal zone and extensive formations of several hectares located on sand flats lower down in the intertidal zone. The latter, which are much less common, have developed most in two areas along the French coast: south of Noirmoutier Island (47°00N, 02°10W [6]) and in Mont-Saint-Michel Bay (48°40N, 01°40W [11]).
Concerning the ecological role of Sabellaria reefs, their function as a habitat has been most often studied (see the references cited above for associated fauna). The influence of these reefs on sedimentary processes has also been considered [12]. However, their trophic role within the ecosystem has never been quantified. Mont-Saint-Michel Bay possesses the largest S. alveolata reef in Europe [7], covering a surface area of ca 100 hectares, with polychaete densities of up to 60 000 worms m−2 [8]. No data are currently available concerning the ecophysiological responses of this species, which are of primary importance for assessment of food uptake in Mont-Saint-Michel Bay, where trophic capacity is threatened by increasing shellfish farming (oysters and mussels).
Studies of polychaete feeding are rare compared to those performed for molluscs, especially with regard to quantitative aspects of filter-feeding. Subsequent to the work of Jorgensen [13], Dales [14] provided some quantitative data on the filtering process in Sabellid and Serpulid worms. Since then, several studies have been carried out in polychaete families with suspension feeding activity, such as Sabellidae [15,16], Serpulidae [17,18] and Terebellidae [19–21]. To our knowledge, the only study performed for Sabellariidae is that of Wells [22] concerning Sabellaria kaiparaensis, a species restricted to the New Zealand coast. Thus, basic data about feeding processes are still lacking, despite the fact that species of this family such as S. vulgaris on American coasts or S. alveolata on European coasts are very common.
The purpose of this study was to obtain clearance rate and retention efficiency values for S. alveolata in order to provide basic data concerning the trophic role of this gregarious species. The methodology to record ecophysiological responses was derived from flow-through systems used for suspension-feeding bivalves. This study is part of a larger project (‘Programme national d'environnement côtier’) designed to investigate the respective roles of the main primary consumers (cultivated species and wild biota) on phytoplankton and resuspended microphytobenthos in Mont-Saint-Michel Bay.
2 Materials and methods
2.1 Sampling and conditioning of Sabellaria alveolata
Samples were obtained from the Champeaux reef located in the eastern part of Mont-Saint-Michel Bay (Figs. 1, 2A and B). Blocks were extracted using a 225-cm2 metal corer 15×15-cm wide (Fig. 2C). The core depth chosen represents the thickness of the living part of the reef. Gruet [6] has shown that this part is about 15-cm wide, equivalent to the distance between the aperture of the tube occupied by the worm when filtering or building and the layer to which it can withdraw during low tide or periods of stress. Thus, the reef is dead below the first 15 first centimetres, consisting of mud-filled tubes used by the worms as a support. If core depth had been less than 15 cm, too many individuals would have been injured. Five samples were extracted in the same area to maintain a similar hydrodynamic environment, and from the same reef stages (platforms), according to the definitions provided by Gruet [5,7].
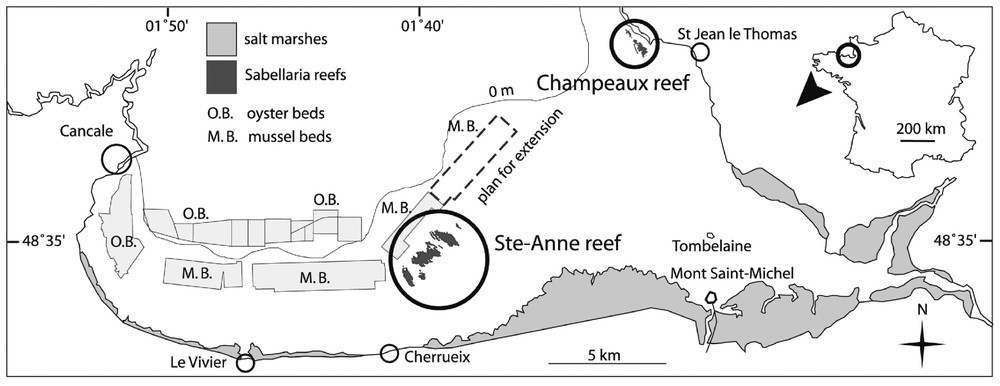
Mont-Saint-Michel Bay and location of S. alveolata reefs.

(A) General view of the reef formation at Champeaux (Normandy, France). (B) Part of the reef showing coalescent ball-shaped structures and platform stages. (C) Corer in the reef. (D) Troughs containing reef blocks. (E) Ventral view of the cephalic part of Sabellaria alveolata, with tentacular filaments in extension.
Samples were carefully washed in seawater and placed in an aquarium containing natural seawater with a flow-through system at a constant temperature (15 °C). Individuals were acclimated for two weeks and fed live algal cultures of Skeletonema costatum and Isochrysis galbana three days before the start of the experiment. These microalgae were chosen to cover a broad size range (2 to 16 μm, Equivalent Spherical Diameter, ESD) as determined by a particle counter. During the acclimation period, dead individuals were easily detected and carefully removed. In the course of experiment, no mortality had been noticed.
Platform stages generally have a very low abundance of other species associated with S. alveolata [8]. However, to avoid disturbances, some rare juveniles of the mollusc Venerupis saxatilis within micro-crevices and individuals of nemertean Lineus sp. or polychaetes such as Perinereis cultrifera and Eulalia ornata were extracted from the reef blocks.
2.2 Experimental equipment
Ten-litre troughs based on those used in studies of Crassostrea gigas [23] and Lanice conchilega [21] were constructed and adapted to S. alveolata. Their size needed to be large enough to receive a perforated removable box (Figs. 2D, 3) containing the 15×15×15-cm3 reef core. Once the removable box was inserted into the trough, a 2-l volume flow chamber existed along the upper surface of the reef, with two baffles ensuring laminar flow. The volume of this flow chamber was adjusted to detect significant filtration. Below this flow chamber, the rest of the cored reef remained immersed in seawater and without connection with the upper circulating water (Fig. 3).
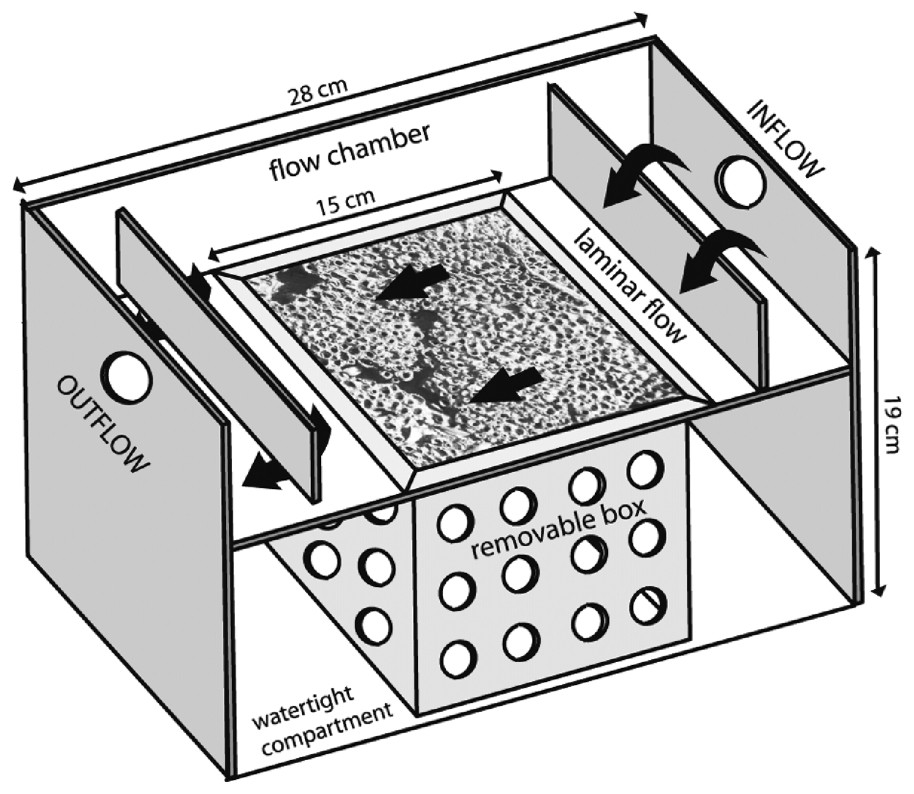
Details of the experimental troughs containing Sabellaria reef blocks. Laminar water flow is shown by black arrows.
This experimental set-up functioned like the flow-through system described by Palmer and Williams [24] for suspension-feeding bivalves. It consisted of five troughs connected in parallel and containing living individuals plus a control consisting of a reef block without polychaetes or associated fauna. The control was obtained by drying a reef core at 60 °C for 3 days and then leaving it at ambient temperature for a week, with regular washing with seawater. The six troughs received seawater filtered onto 0.45-μm Millipore R.A. filters and mixed in a 200-l tank with an experimental diet composed of I. galbana and S. costatum. These species, maintained in the culture collection of the Marine Biology Laboratory of the College of Sciences in Nantes, were grown in a culture room (temperature: 14 °C; photoperiod: L14/D10; light intensity: 100 με m−2 s−1 with με=microEinstein). The diet in the 200-l tank was adjusted to 23 700 cells ml−1 (s.d.=2600; n=6) for I. galbana and 3800 cells·ml−1 (s.d.=130; n=6) for S. costatum in order to obtain a distinct bimodal size distribution over the 2–16 μm range (ESD). Mean particulate volume per litre of seawater in the 200-l tank was 2.8×106 mm3 l−1 (s.d.=1×106; n=6). Flow speed was adjusted with Gilmont© flow-meters to 2.5 L h−1 (according to an abacus under 1 atm and 20 °C) to maintain inflow particle concentration 30% higher than at outflow, thereby avoiding recirculation artefacts [25] and abnormal feeding conditions [26].
2.3 Measurements
Polychaetes were placed in troughs one hour prior to any measurements. Five measurements, with an interval of one or two hours, were made during the experiment. Each measurement (at 10 h, 12 h, 13 h, 15 h and 16 h) consisted in water samples collected simultaneously from the six troughs (5+1 control). Duration of sampling was about 30 s. Mean values have been calculated using all five troughs. Particle-size distributions were determined with a Multisizer particle counter fitted with a 100-μm aperture. Particle counts were made with coefficients of coincidence < 5%. All particle sizes are expressed as equivalent spherical diameter (ESD). Particle concentrations were estimated for a size range of 2 to 16 μm. Retention efficiencies were computed for this entire size range as: Er(%)=100×[(Inflow−Outflow)/Inflow], where ‘Inflow’ represents particle concentration in the control and ‘Outflow’ particle concentration in the troughs containing the animals. Details about retention efficiency calculations are given in Barillé et al. [23]. Retention efficiency data were then used to avoid underestimation of clearance rate calculations due to small particles cleared from the suspension by polychaetes, but not retained on tentacular filaments (Fig. 2E), and therefore released into troughs. To determine this potential underestimation, three retention ratios (Inflow – Outflow/Inflow) were calculated from particle counter data as the sum of particle concentrations in 2–16 μm, 3–16 μm, 6–16 μm size ranges. The clearance rate was subsequently calculated only for particles above a threshold ensuring that all were cleared with 100% efficiency. Given the mean flow in an individual trough, the clearance rate (l h−1) was calculated as: [Inflow−Outflow/Inflow]×flow rate.
After experiments, all individuals were killed in 4.5% formaldehyde solution. Tubes were then disintegrated in seawater, and all S. alveolata individuals were collected and counted, and their dry meat weight was estimated after 48 h at 65 °C.
2.4 Statistical analysis
One-way ANOVA was used to test for differences in clearance rates. Data were checked for normality and homogeneity of variance. A significant level of p<0.05 was used in all tests.
3 Results
Size spectra obtained with the particle counter and expressed in ESD (Fig. 4A) showed two modes at the inflow of the experimental troughs. The first, at around 4 μm ESD, was due to I. galbana (linear length of cells=5–6 μm), while the second, more dispersed and centred around 10 μm ESD, was due to S. costatum (chain length of five cells=50 μm). In some cases, a third peak at 2.5–3 μm ESD appeared for size distributions at the outflow of the experimental troughs (Fig. 4B). Microscopy studies revealed that these particles corresponded to spermatozoa. However, these size distributions could no be used to compute retention efficiencies. A similar phenomenon of even smaller particles at the outflow was recorded in several troughs. Observations of worm activity inside the troughs showed that some individuals expelled particles from the tubes. Size analysis of these particles directly pipetted near the tubes revealed that most were under 4 μm ESD (Fig. 4B), while microscopy studies indicated that they were not a gamete emission. Despite the two-week acclimation period in filtered seawater, it seems likely that worms collected in a turbid area retained small inorganic particles released during the experiment. Each size distribution at the outflow of the troughs was therefore carefully examined prior to retention efficiency calculations.
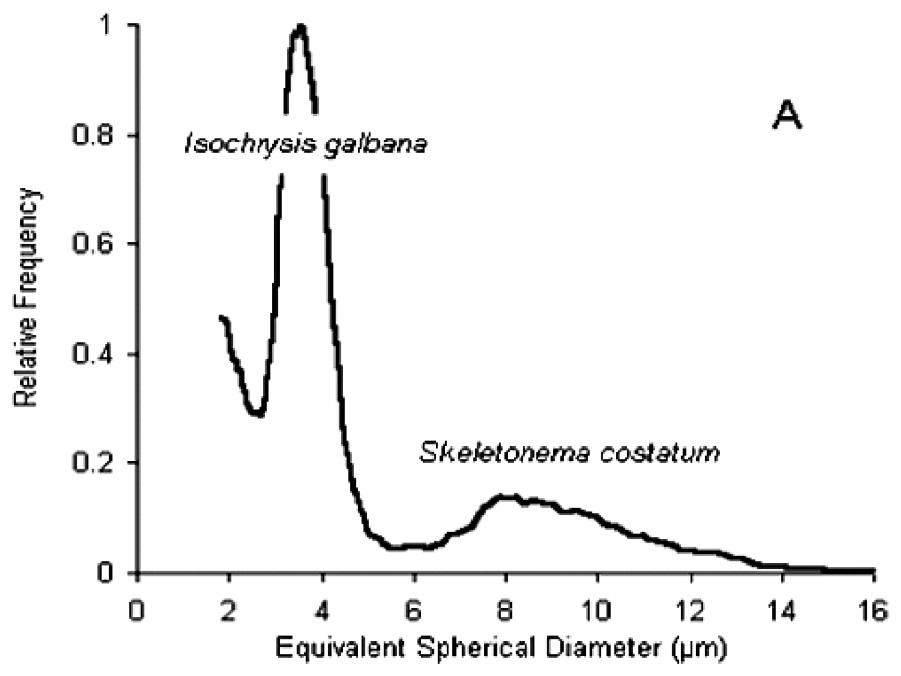
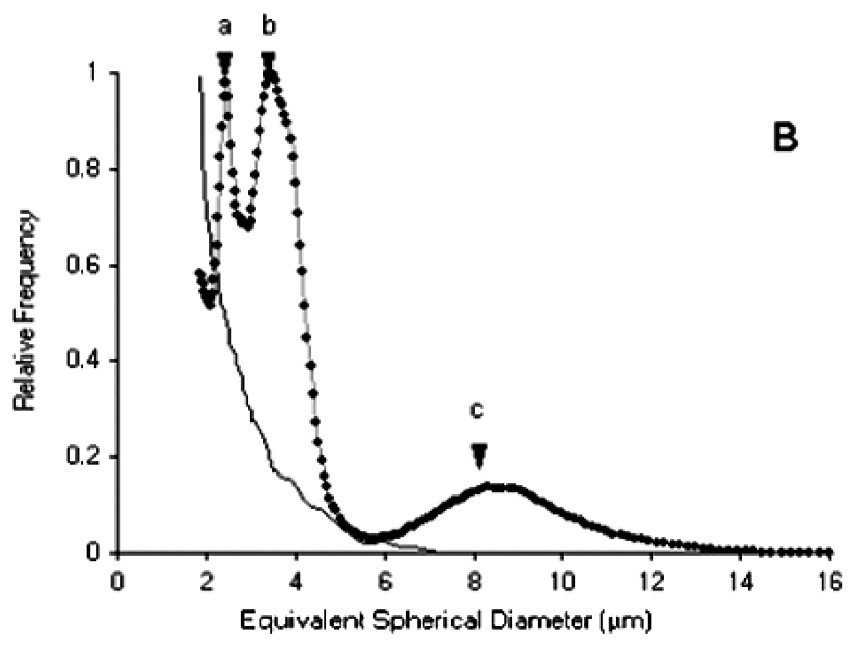
(A) Particle size spectra of the diet. (B) Dotted line: size spectra at the outflow of the experimental troughs, showing peaks for I. galbana and S. costatum (respectively b and c) and a third peak (a) corresponding to an emission of spermatozoa. Full line: size spectra for inorganic particles expelled by individuals during the experiment.
The retention efficiency curve obtained for the experimental conditions used shows an inflection at around 4 μm ESD (Fig. 5). Below this size, retention efficiencies decrease rapidly, and worms are unable to retain particles smaller than 2 μm ESD. Eighty percent of 4-μm-ESD particles were retained by S. alveolata, and all particles larger than 6 μm ESD were cleared with 100% efficiency. The clearance rate calculation was therefore based on particles above 6 μm ESD, as determined by the particle counter, which corresponded to the microalga S. costatum. This choice was also supported by calculations of the retention ratios for the three size ranges at the five time intervals (Table 1), which showed that most determinations for the 2–16-μm and 3–16-μm ranges provided negative values related to generation of the particles identified above. In all cases, mean retention ratios increased from the widest to the narrowest range (6 to 16 μm), while variability decreased.
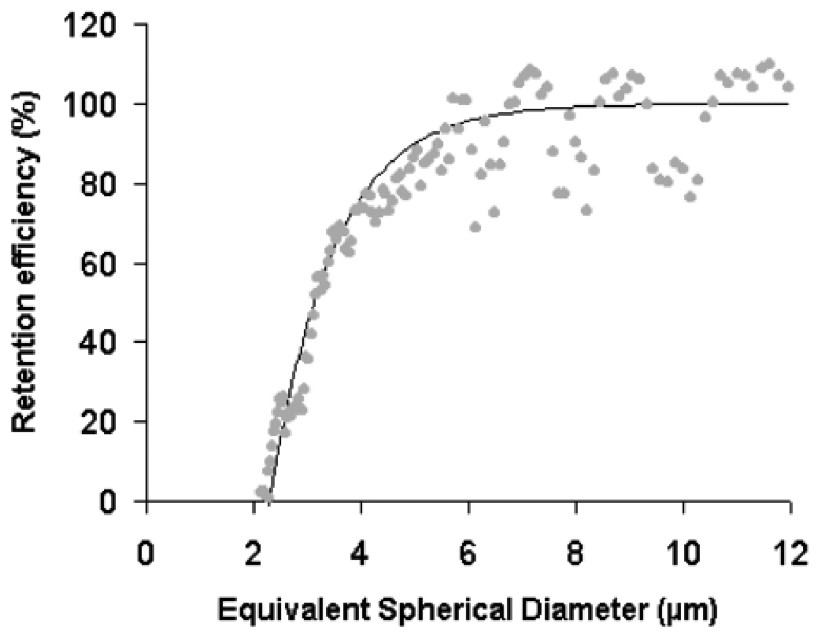
Retention efficiency of S. alveolata for the tested diet.
Mean retention ratios of Sabellaria alveolata reef blocks based on the sum of all particles counted for three size ranges at five time intervals
| 2–16 μm | 3–16 μm | 6–16 μm | |
| 10 h | 0.15 (0.06) | 0.25 (0.07) | 0.34 (0.07) |
| 12 h | −0.10 (0.18) | −0.03 (0.17) | 0.30 (0.06) |
| 13 h | −0.04 (0.08) | 0.04 (0.07) | 0.26 (0.05) |
| 15 h | −0.31 (0.18) | −0.18 (0.15) | 0.26 (0.04) |
| 16 h | −0.35 (0.17) | −0.27 (0.18) | 0.24 (0.02) |
Comparison of the mean clearance rate (CR) calculated for the five experiments did not reveal any significant statistical differences (Fig. 6, ANOVA, P<0.05), although a regular decrease of variability was observed from 10 to 16 h. A mean clearance rate of 0.7 l h−1 (with a 95% confidence interval of 0.41 to 0.72 l h−1) was calculated for all data. Since the 225 cm2 reef blocks used for the experiment contained a mean number of 940±102 (S.E.) individuals (2.74±0.45 g dmw−1), the mean clearance rate of an individual was estimated at 0.00075 l h−1 (assuming that all the worms were equally filtering).
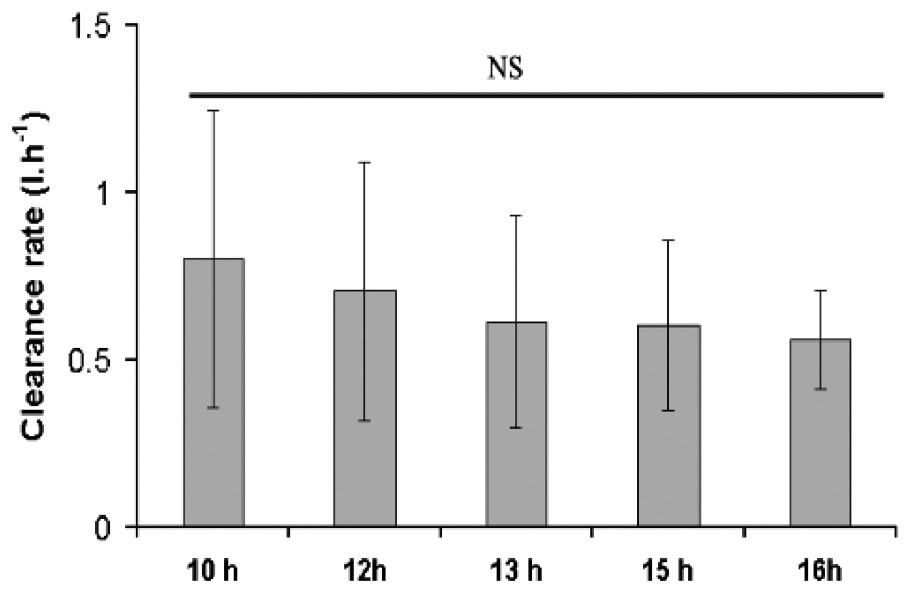
Mean clearance rates of S. alveolata calculated at five time intervals (n=5; 95% confidence intervals are shown).
4 Discussion
This work reports the first retention efficiency and clearance rate data for the polychaete Sabellaria alveolata, a gregarious species widely distributed along European coasts. In some shellfish ecosystems, the reef formations built up by this polychaete reach several hectares (e.g., Bourgneuf Bay or Mont-Saint-Michel Bay), but its role in the trophic web has been largely disregarded. Clearance rate data are needed for S. alveolata in order to assess the role of this species as a potential trophic competitor with farmed bivalves, especially in Mont-Saint-Michel Bay, where the largest reef formations in Europe are found.
Ecophysiological responses were obtained by applying a methodology derived from a flow-through system used to study suspension feeding in bivalves. The choice of a reef block instead of a single individual was related to technical and biological considerations. Although some adjustments were necessary for the troughs used, the equipment (pump, flow-meter) designed to measure bivalve physiological responses [27] was appropriate to detect response in a reef block. Moreover, responses obtained with a mean number of 940 individuals per block integrated inter-individual behaviours such as proximity or constraint effects (see for example [28,29]). Behavioural responses in gregarious species generally differ for isolated individuals and groups of individuals of the same species, e.g. for mussel beds [30,31] or bryozoans [32]. Thus, it was considered more realistic to obtain clearance rate data for a reef block rather than a single individual, as these data could be used in the context of an ecological study, i.e. in an ecosystem. If it is considered that 0.0225 m2 of reef (the surface area sampled in this study) has a mean clearance of 0.7 l h−1, this value can be extrapolated to filtration for the entire Sainte-Anne reef (Fig. 1). This reef, regarded as the largest S. alveolata formation in Europe, is gradually being surrounded by mussel farms. Aerial photographs indicate that this reef area covers 98 ha (Dubois, unpublished data), so that the reef clearance rate should be 30 500 m3 h−1. Due to its position on the shore, the mean daily immersion period is about 13 h. During this period, the Sainte-Anne reef filters 396 500 m3 of seawater. Given a value of around 0.4 m3 water per square metre per day. The Sainte-Anne reef population clearance rate appears to be well below the rates of ca 100 m3 m−2 day−1 found in dense mussel beds [33]. In the project designed to evaluate the trophic capacity of Mont-Saint-Michel Bay, this simple calculation should be used as an order of magnitude. To assess the impact of Sabellarian reefs, comparisons need to be made with the filtration pressure exerted by cultivated species (mussels and oysters), whose stock estimations are currently in progress. And vice versa, it is also possible that the reefs could be endangered by a lack of food for the worms. Obviously, this scale-up calculation must rely on complementary experimental work, particularly studies of the influence of the main environmental factors (temperature, turbidity, food concentration, hydrodynamic) on the ecophysiological responses of these worms.
The mean individual clearance rate of S. alveolata determined in this study (0.00075 L h−1) can be compared with the only results reported for the genus Sabellaria, i.e. a rate of 0.018 L h−1 (25 times higher) found by Wells [22] for the species S. kaiparaensis. This author assessed the clearance rate by means of variations in optical density in a closed circulation system using graphite suspension, according to the method proposed by Dales [14]. However, no details are given concerning particle size, temperature or cell concentration. A comparison with Sabellids and Serpulids [14] indicates that the S. alveolata clearance rate is much lower than that of larger species such as Sabella pavonina (0.073 l h−1) or Myxicola infundibulum (0.28 l h−1) as well as smaller species such as Pomatoceros lamarcki (0.027 l h−1) or Hydroides norvegica (0.011 l h−1), but close to that of Spirorbis borealis (0.00023 l h−1) or Salmacina dysteri (0.00029 l h−1). Nevertheless, the value determined here for S. alveolata is probably underestimated, because of the assumption that all worms were equally active. A recent study of the filtering activity of Ditrupa arietina [17] indicates that this Serpulid could spend less than 25% of its time feeding. Therefore, further investigations are needed to assess the number of filtering worms, together with determinations of particle size and concentration analysis.
Ropert and Goulletquer [21], who studied the trophic competition between Lanice conchilega (Polychaeta: Terebellidae) and cultivated oysters, obtained worm clearance rates ranging from 0.073 to 0.108 l h−1 ind−1 and retention efficiencies starting above 4 μm. Jørgensen et al. [34] found an optimum particle size retention of 3 μm in Sabella penicillus, a species characterised by a downstream collecting process [35]. The results obtained in the present study suggest that S. alveolata is less efficient than the above-mentioned Sabellid polychaete in retaining small particles. Examination of the structure and ciliation of the feeding organs of S. alveolata should help in interpreting these differences (Dubois et al., in preparation). However, S. alveolata has a retention efficiency comparable to that of the cultivated oyster C. gigas [21,23], which indicates that both species exploit a similar particle-size range. Wild oysters have recently colonised the degraded part of the Sainte-Anne reef, where S. alveolata is no longer abundant. In addition to spatial competition, the trophic impact of bivalves on the re-colonisation of the reef by S. alveolata is a critical development.
Further experiments are needed to elucidate the role of the sabellarian reef in the functioning of Mont-St-Michel Bay in the context of divergent views regarding the worms as competitors for cultivated species or as artisans of historical bioconstructions that are part of national or world heritage.
Acknowledgements
The authors are grateful to P. Rosa for algal cultures and P. Garen for lending flow-meters. This work was supported by the PNEC (‘Programme national d'environnement côtier’) project for Mont-Saint-Michel Bay.



