1 Introduction
New approaches to improve the clinical outcome of vascular structures, including vascular stents and grafts, have been extensively investigated over the past 50 years. The ultimate goal is to increase the success rate of these implants by improving their biocompatibility. The most effective way of improving the success rate of small diameter conduits is to reduce thrombogenicity and intimal hyperplasia. Surface endothelialization will achieve this desired outcome, as we know that the luminal surface within a blood vessel naturally prevents thrombotic lesions due to specific properties of endothelial cells [1]. Endothelialization of implants is limited by the inability of cells to retain adhesion after exposure to flow. Extracellular matrix proteins, including fibronectin and collagen, have been shown to enhance cell adherence and retention on materials [2–4]. These proteins provide a better support structure for many cell types [4,5]. Both collagen [2,4,5] and fibronectin [6,7] are suitable support structures especially for endothelial cells.
The interactions between endothelial cells and their underlying extracellular matrix are crucial for basic cell functions including migration, attachment and proliferation. The extracellular matrix is a complex and dynamic structure composed of proteins, polysaccharides, proteoglycans and sometimes minerals [8]. These proteins are responsible for initiating cell behaviour by binding to specific integrins located on the surface of the cell. The interaction between these proteins and the cell stimulates an intracellular signalling cascade, which enables the cell to carry out many functions. The ability of extracellular matrix molecules to interact and bind with each other is crucial for the functions and integrity of the matrix. Extracellular matrix components stimulate different cellular responses, however, it is often the interaction and binding of two or more proteins in the extracellular matrix that will elicit a response [9]. Therefore, it is surprising that in most, if not all, studies investigating the endothelialization of an extracellular matrix modified surface, only one protein is used in the coating. Most studies have focused on the interaction and functioning of extracellular matrix proteins together [10–17] but not on their effect on endothelialization. It needs to be determined if extracellular matrix proteins function better in vitro when coated together compared to separately. The two extracellular matrix proteins of particular interest are collagen and fibronectin as they co-exist in the extracellular matrix and their interaction with cells is essential for many extracellular matrix-dependent cell functions [9]. These two proteins are also known to have important roles in stimulating cell migration and proliferation. Moreover, collagen I is known to be a major matrix component of the endothelial wall of the blood vessels [18,19].
As a result, this present study not only investigated the effect of a single extracellular matrix coating on endothelial cell growth but also a double coating to mimic more closely the complexity of this extracellular matrix. The single coatings were collagen and fibronectin alone, and the double coating was a combined collagen and fibronectin (collagen + fibronectin). The main objective was to study the proliferation and growth of Human Umbilical Vein Endothelial Cells (HUVEC) on these three extracellular matrix coatings. The mechanisms involved in the enhancement of cell growth were investigated both at molecular and cellular levels. Polystyrene was coated with and without the three coatings to measure any variations in protein adsorption and cell binding domains. The behaviour of HUVEC on extracellular matrix coated polystyrene were analysed with respect to cell attachment, proliferation and morphology.
2 Materials and methods
2.1 In vitro culture of HUVECs
HUVEC were obtained from PromoCell (ref. C-12200) and cultured in M199 supplemented with 20% foetal bovine serum FBS (Gibco), 1% antibiotics (penicillin/streptomycin, Gibco), 1% L-glutamine (2 mM Gibco), 1% heparin (500umh/mL, Gibco) at 37 °C in a humidified incubator with 5% CO2. The medium was replaced every 2–3 days and HUVECs were grown to near confluence and passaged when required. For cell passage, the media was aspirated and replaced with 4 mL of trypsin. After a 10-minute incubation, the cell suspension was neutralised with 2 mL FBS and centrifuged at 200 g for 5 minutes. The pellet was resuspended in 1 mL of media and transferred into 75 cm2 culture flasks at a resulting density of 10,000 cells/cm2.
2.2 Extracellular matrix proteins (ECM)
Collagen (Coll, purified bovine type I collagen, ref CBPE2, Symatese) and fibronectin (Fn, human plasma fibronectin, Ref. 11051407, Roche Diagnostics) were used in this study as single- and double-layered ECM coatings. Each protein was diluted according to the manufacturer's guidelines. Fn was used at a concentration of 2 μg/mL in phosphate buffer saline (PBS) (120 ng/cm2), Coll was used at 1 mg/mL in distilled water (60 μg/cm2). This coating protocol has been described by Nagel et al. [20] and a Coll coating at this concentration has been shown to enable fibril formation in vitro [21].
2.3 ECM protein coatings
ECM protein coated polystyrene culture plates were used for studies with and without an ECM coating. The Fn solution was allowed to adsorb on polystyrene for 45 minutes, at 37 °C and washed with PBS 3 times, for 10 minutes. The Coll was left to adsorb for 10 minutes at room temperature and then washed twice in PBS for 10 minutes. The protocol for the Coll + Fn coating was as described for the single coating, with the Coll coated first, followed by the Fn. Petri dishes were freshly coated prior to each application.
2.4 HUVEC proliferation on ECM coated polystyrene
Cells were seeded at a density of 10,000 cells/cm2 on freshly coated polystyrene culture plates. After 24 and 48 hours in culture, the samples were rinsed in PBS to remove non-adherent cells. The cells were then trypsinized, and counted with trypan blue (Sigma-Aldrich). The proliferation rate was calculated by dividing the number of living cells by the number of cells seeded. The experiment was conducted in triplicate and repeated three times (n = 9).
2.5 HUVEC migration on different ECM coating
The Oris™ cell migration assay (Platypus Technologies) was used to assess the cell migration. Micro-titration plates (96-well) with a black border were coated with Coll, Fn and Coll + Fn and migration stoppers were inserted into each well. HUVEC were seeded around the perimeter of the stoppers at a density of 20,000 cells/cm2. After a 24-hour incubation, the stoppers were removed and cells were cultured for a further 24 hours to enable migration into the detection zone, then the cells were fixed and labelled with Fluorescein Isothiocyanate (FITC, 2 mg/mL for 2 minutes, Sigma-Aldrich). Each well was thoroughly washed with PBS to remove any FITC excess. Migration was observed by quantifying FITC emission in the detection zone by using a BioTek Synergy™ HT Multi-Mode Microplate Reader at 525 nm. Data represent the mean ± SE from 9 wells for each condition.
2.6 Fibronectin and collagen labeling
FITC labeled Fn and Rhodamine Isothiocyanate (Rhod) labeled Coll, were prepared as followed, 1 mg of Fn or 1 mg of Coll, was dissolved in 1 mL of NaHCO3 0.1 M pH 9, and incubated for 1 hour at RT with 10 μL FITC (F351, Sigma-Aldrich) or 10 μL of Rhod (R1755, Sigma-Aldrich©) both 10 mg/mL in dimethyl sulfoxide (DMSO). The unbound fluorochromes were separated on PD-10 desalting columns (GE Healthcare). Fn-FITC and Coll-Rhod were coated on polystyrene culture plates as the non-labeled ECM proteins. Cells were seeded at 20,000 cells/cm2 and incubated for 2 hours at 37 °C. After the incubation period, the culture medium was eliminated and the polystyrene culture plates were observed under fluorescence microscope (DMI600, Leica System).
2.7 HUVEC morphological analysis
The HUVEC morphology was examined by different microscopic methods. Cells were seeded at 10,000 cells/cm2 and cultured on ECM coated surfaces. Fluorescence microscopy was used to determine the cytoskeleton arrangements of HUVEC. Twenty-four hours post-seeding, HUVEC were fixed with 4% formaldehyde overnight, permeabilized in 0.1% Triton X-10 and incubated in 2% bovine serum albumin (BSA) for 1 hour at RT. The cells were then incubated in the dark for 1 hour at RT with a cytoskeleton stain containing phalloidin (25 μL/mL) (phalloidin-X5, Fluoprobes) and DAPI (1 μg/mL, Sigma-Aldrich) diluted in PBS. Cells were mounted in Mowiol (PolySciences) and observed with a fluorescence microscope (DMI600, Leica System).
The cells were observed with a Scanning Electron Microscope (SEM). Forty-eight hours post-seeding cells were fixed with 4% formaldehyde for 24 hours. The HUVEC were then covered with a layer of gold (about 50 nm) for 3 minutes under 25 mA, using a SEM coating unit E5100 (Polaron Equipment Ltd) and viewed under the SEM (ESEM FEG XL30 Philips).
2.8 ECM protein domain accessibility
The primary antibodies used to determine protein domain accessibility were polyclonal Fn (pFn; A0245, Dako), polyclonal Coll (pColl; AB749, Millipore) and monoclonal RGD (mRGD, MAB 1926, Millipore). The freshly coated 6-well polystyrene culture plates were saturated with 0.1% Tween20 + 0.25% BSA in PBS for 30 minutes at 37 °C. PBS + 0.1% Tween20 was used as the wash solution following each incubation. Primary antibodies were diluted 1/5000 in PBS and incubated for 1 hour at 37 °C. After washing, the horseradish peroxidase-conjugated secondary antibodies (goat anti-Rabbit and anti-mouse, Interchim) were diluted at 1:10,000, incubated at 37 °C for 1 hour on the corresponding antibodies. The wells were then incubated for 2 minutes in the substrate, composed of ortho-phenylene-diamine (0.5 mg/ml, Sigma-Aldrich) and H2O2 (0.4 μL/mL) diluted in citrate buffer. The reaction was blocked with 100 μL of 1 M HCl. The absorbance was obtained at 490 nm in a microplate reader (Biorad Laboratories). The results were represented as the absorbance of the coated wells, subtracted by the control well (all components of the assay, except the primary antibody). The results were expressed as the mean absorbance of two samples per experiment, over three individual experiments ± SE (n = 6).
2.9 Statistical analysis
Analysis of the data was conducted using the statistical program GraphPad Instat (version 3.01). Unless otherwise stated, the data were represented as the mean ± SE of 9 measurements, corresponding to 3 samples of coated polystyrene for each ECM protein repeated 3 times. Comparisons across the 4 experimental groups were determined by a one-way ANOVA followed by the Newman-Keuls Multiple Comparison post hoc test. A value of P < 0.05 was considered significant.
3 Results
3.1 Seeding of HUVEC on extracellular matrix coatings
Cultured HUVEC were seeded on different extracellular matrix proteins and the proliferation was measured at 24 and 48 hours post-seeding. As shown in Fig. 1, the proliferation of HUVEC at 24 hours was similar across all four conditions; there was no significant difference in proliferation at 24 hours. However, a significant difference (P < 0.05, one-way ANOVA) became visible after 48 hours. Fig. 1 shows that endothelial cell proliferation was significantly higher on a double coating (3.53 ± 0.62) compared to polystyrene (2.66 ± 0.45) and both collagen and fibronectin single coatings (Coll: 2.66 ± 0.31; Fn: 2.73 ± 0.17). These results show that HUVEC prefer to grow on a double-coated surface of collagen + fibronectin.
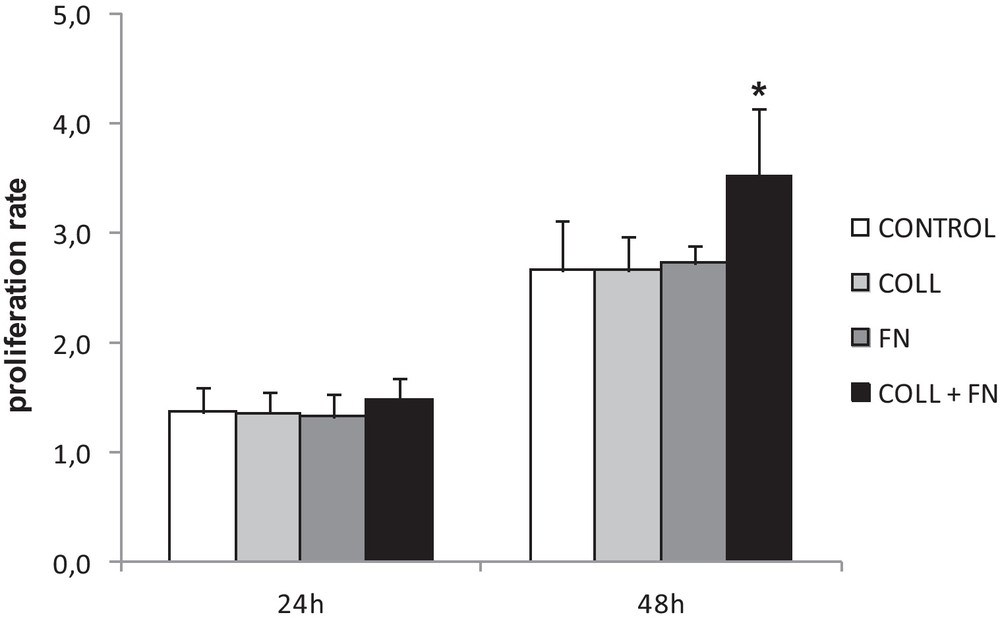
Proliferation rate on various ECM proteins at 24 and 48 h post-seeding. A significant difference in proliferation of HUVEC was only observed after 48 h in culture on double coating. The data represent the mean cell proliferation rate of 3 experiments in triplicate ± SEM (*P < 0.05).
3.2 Cell migration of HUVEC on extracellular matrix
Fig. 2 shows that a significant increase (P < 0.001; one-way ANOVA) in HUVEC migration was observed on both a single collagen coating and the collagen + fibronectin coating compared to all other conditions. This result suggests that collagen plays a role in supporting HUVEC migration across a surface. Fig. 2 also indicates that the migration property observed in a collagen + fibronectin coating is similar to that seen in the collagen-only coating, suggesting collagen can somehow interact with the HUVEC even though it is coated underneath the fibronectin.
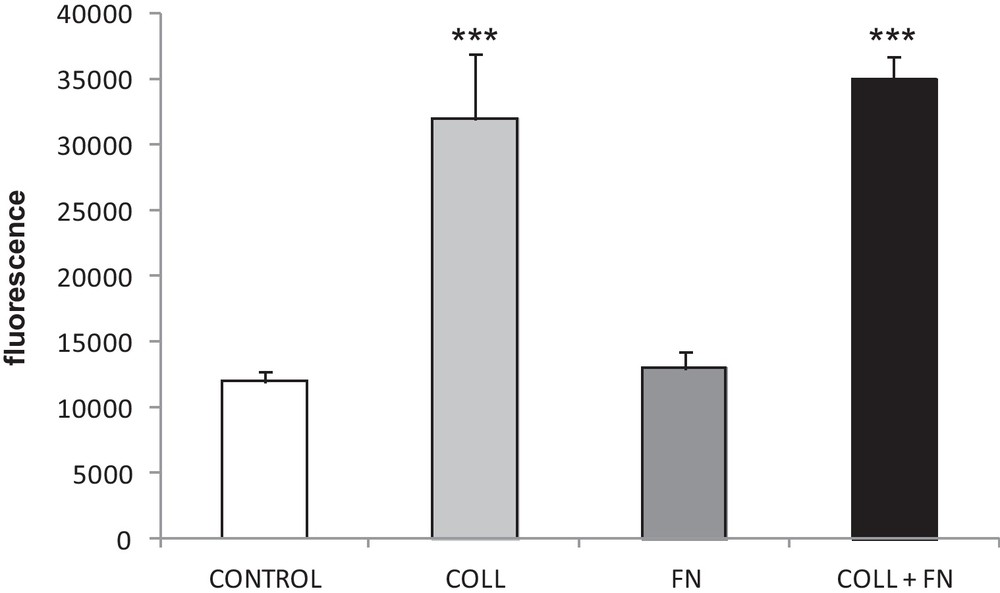
Migration, determined by the Oris™ cell migration assay, of HUVEC seeded on different extracellular matrix coating. The resulting graph represents the measurements of the fluorescence emission of the migrating cells into an unpopulated area. Data are expressed as the mean ± SEM from 9 wells for each condition. A significant increase is observed on collagen and collagen + fibronectin coating (***P < 0.001).
3.3 Uniform extracellular matrix coverage
To determine if a uniform layer of extracellular matrix proteins was adsorbed when the polystyrene was coated, the samples were analysed by fluorescence microscopy. Prior to coating, the markers FITC and Rhodamin were chemically linked to fibronectin and collagen respectively. After 2 hours in culture, the coatings were observed, and the only protein visible was the one coated and not the one produced by the cells. As shown in Fig. 3a and b, Rhodamin and FITC were visible on their respective extracellular matrix molecules as collagen and fibronectin molecules were evenly distributed across the surface of the polystyrene, visible by the uniform coverage of the fluorochrome. Fig. 3c and d demonstrate that on the double coating, collagen and fibronectin were co-distributed across the surface and could be found at identical locations (Fig. 3e) on the polystyrene. The results of Fig. 3 also show that the concentration of collagen and fibronectin was higher around the perimeter of the HUVEC, in both the single and double coatings.
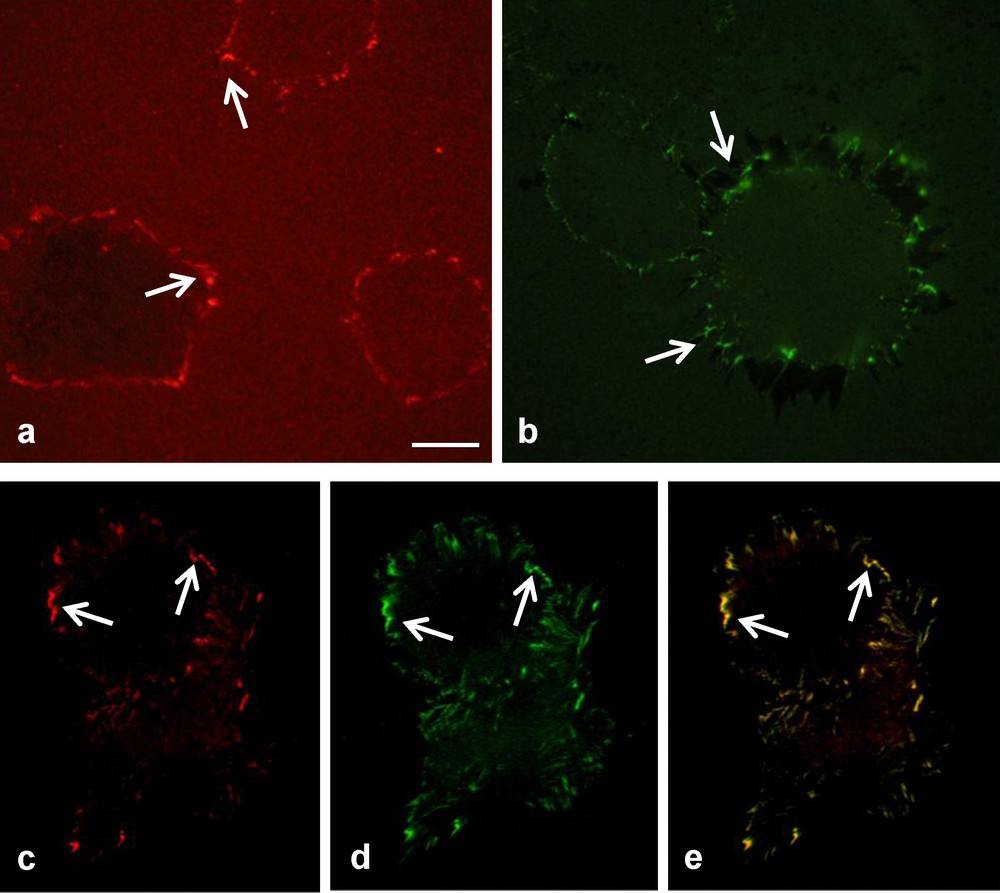
Visualization using fluorochrome coupled ECM molecules of collagen and fibronectin remodelling by HUVEC, 4 h after being seeded. In the presence of a single coating only rhodamine-collagen (a) and FITC-fibronectin (b) could be detected. On a double coating (c–e), rhodamine-collagen (c) and FITC-fibronectin (d) were both visible at identical locations (e, see arrows) at the cell periphery (bar 25 μm).
3.4 The formation of filopodia on an extracellular matrix
To further explore the behaviour of HUVEC in the presence and absence of an extracellular coating, phalloidin-staining of the actin cytoskeleton was examined. Because the HUVEC proliferation rate at 24 hours was not significantly affected by an extracellular matrix coating, it was important to determine whether cell morphology was affected. As shown in Fig. 4, an extracellular matrix coating stimulated the HUVEC to rearrange their cytoskeleton and form filopodia. The reorganization of the HUVECs cytoskeleton caused the actin filaments to protrude from their lamellipodium frontier, to form filopodia. The presence and quantity of filopodia did not differ depending on the extracellular matrix coating (Fig. 4b, c and d). However, in the absence of coating there was no filopodia formed, only lamellipodia were visible on the HUVEC (Fig. 4a).
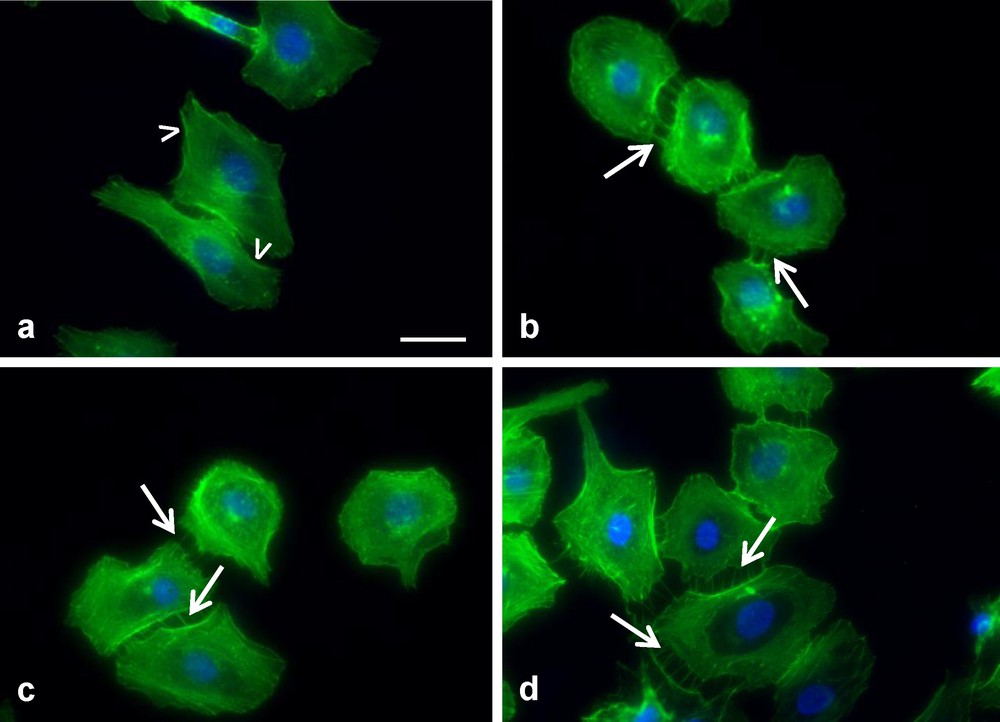
Observation of HUVEC cytoskeleton grown on (a) polystyrene, (b) collagen, (c) fibronectin, (d) collagen + fibronectin. In the presence of an ECM coating the cells produced filopodia (arrows in b, c and d), however in the absence of coating lamellipodium frontier is observed (arrowheads in a) (bar 25 μm).
3.5 Protein coating cause morphological changes to the cell
To determine whether the protein binding interaction caused changes in cell morphology, HUVEC were seeded on the different proteins and examined using SEM. As displayed in Fig. 5, HUVEC shape was affected by the extracellular matrix coating. In Fig. 5a and c, it is seen that the morphology is similar for non-coated and fibronectin-coated polystyrene. The cells were flat, round with a large appearance. However, HUVEC grown on a collagen or collagen + fibronectin surface had an elongated shape and a raised appearance (Fig. 5b and d). This suggests that collagen plays some role in directing cell morphology. These differences on the collagen + fibronectin coating compared to the other coatings correspond to cord-like structures forming, that could be precursors of angiogenesis [22].

HUVEC morphology observed by SEM. Cells were grown for 48 h on (a1, a2) polystyrene, (b1, b2) fibronectin, (c1, c2) collagen and (d1, d2) collagen + fibronectin. Note the difference in morphology between the round and large cells seen in (a) and (c) and the more fusiform cells, corresponding to cord-like structures (panels b and d).
3.6 Protein adsorption and accessibility of fibronectin cell binding domains
The accessibility of fibronectin RGD domain (arginin-glycin-aspartic acid sequence), when coated alone or combined with collagen, was determined by ELISA. Our results show that there was a significant decrease in adsorbance with both monoclonal and polyclonal fibronectin antibodies on the double coating compared to the single coating. The decrease of adsorbance read from polyclonal fibronectin (pFn) and monoclonal RGD (mRGD) was 26.5% (P < 0.001) and 82.4% (P < 0.001), respectively. There was no significant difference in the adsorbance read from polyclonal collagen (pColl) on a double or single coating (Table 1). These results suggest that collagen and fibronectin interact and/or bind in a RGD dependent manner.
Absorbance data of ECM proteins detected by ELISA. Polyclonal antibodies (pColl and pFn) and monoclonal antibody directed against fibronectin RGD domain (mRGD) were used. The fibronectin absorbance with both polyclonal and monoclonal antibodies significantly decreased in a double coating compared to a single coating. The collagen absorbance did not significantly change between the single and the double coating. The results are expressed as the mean of absorbance ± SEM (n = 6).
| ECM Coating | |||
| Antibody | Coll | FN | Coll + FN |
| pColl | 0.918 ± 0.196 | nd | 0.976 ± 0.077 |
| pFn | nd | 1.202 ± 0.153 | 0.830 ± 0.145*** |
| mRGD | nd | 0.224 ± 0.096 | 0.041 ± 0.026*** |
4 Discussion
To successfully develop a biosynthetic blood vessel that comes as close as possible to the characteristics of native blood vessels, it is essential to understand the important roles that the ECM and vascular cells have in maintaining normal vessel function. The cells of a blood vessel are essential for tissue regeneration and repair. They have the ability to proliferate, differentiate, migrate, communicate with the cells through cell signaling, and synthesize and secrete ECM components [1]. The ECM, and among them fibronectin and collagen, are responsible for tensile strength, elasticity and compressibility of blood vessels [23].
A recent study has shown that when cells interact with fibronectin it causes the fibronectin molecule to polymerise and change in structure [24]. Fibronectin matrix polymerisation has been shown to strengthen collagen-based tissue constructs [25] and promote type I collagen deposition [26]. Several authors have suggested that the fibronectin–collagen interactions are fundamental to the structure of an ECM and the behaviour of cells on materials [4,27]. Fibronectin is able to bind to collagen via its collagen-binding domain, located adjacent to the amino-terminal region [28]. The collagen-binding domain has four Type I and two Type II homologous repeats in the sequence (I6-h1-II2-I7-I8-I9). Nakamuna [29] showed that the adhesive properties of collagen-binding domain and full-length fibronectin are similar. The results from this study also suggest that endothelial cells interact with the fibronectin's collagen-binding domain and may therefore increase cell viability. The ability of collagen to bind to fibronectin depends on the collagen type and the availability of its fibronectin binding site, which is said to be located on the α1 chain of collagen [30]. Fibronectin has the highest affinity to bind to collagen type III [27], while collagen I is the most abundant form of collagen in the body [31]. The collagen used in this study was predominantly type I with traces of type III, and it was assumed that when coated together, collagen and fibronectin would interact and bind forming a matrix. Evidence of collagen and fibronectin binding is well-documented, but the mechanism by which they bind is not completely understood. However, it is known that their interaction results in significant conformational changes in both collagen and fibronectin [11]. Furthermore, the ability of fibronectin-collagen-binding is dependent on the structure of both molecules [27,32]. The integrity of disulphide bonds in the type I and type II repeats of fibronectin affects its ability to bind to collagen [27]. Shimiizu [28] found that when the disulfate bonds were cleaved, the binding affinity of fibronectin to collagen (type I and type IV) was reduced. The tertiary structure of collagen is thought to modulate its interaction but it is not essential for fibronectin recognition [27]. The affinity of collagen to bind to fibronectin is higher in denatured collagen than native collagen [33], and it has been suggested that during collagen denaturation, its triple helix unfolds, exposing RGD sequences. This enables RGD dependent fibronectin–collagen-binding [32]. In similar contexts, Ohashi [34] found that cells routinely elongate adhered fibronectin and this elongation may expose binding sites for collagen on the fibronectin molecule. Other authors suggest that fibronectin identifies a location on the α1 chain of collagen, which is located near the collagenase cleavage site [11]. Although the exact ways in which collagen and fibronectin interact and bind are not known, it is clear that their interaction is fundamental to the integrity of the ECM and its ability to increase cell adherence [16]. We suggest that the increased proliferation and growth of HUVEC on a double coating is due to the interaction between fibronectin, collagen and the cells. It can be assumed that the interaction and joining of these adhesion molecules results in an ECM formation that mimics that found in vivo. The fibronectin + collagen support structure has an enhanced adhesion property that is more suitable for the growth and proliferation of HUVEC. This effect was confirmed by recent in vivo experiments demonstrating the ability for collagen modules coated with fibronectin to increase vessel formation based on an increase of proliferation [35].
The HUVEC seeded on ECM molecules had reorganized their actin filament to spread and form connections with neighboring cells. Among ECM molecules, fibronectin has a role in regulating cell adhesion, migration, motility, and due to these characteristics, fibronectin for example plays an important role in wound healing [23]. The interaction between the endothelial cells and the ECM proteins is fundamental. We have demonstrated that each ECM coating initiated cell migration by causing the HUVEC to reorganize its cytoskeleton and that only ECM coated samples induced visible filopodial projections, which protruded from the lamellipodium frontier. Cell migration requires actin filament polymerization, which initiates the development of membrane protrusions in the direction of migration [4]. Cell migration and motility is a tightly organized process and depends on certain intracellular molecules, including the Rho GTPases. This family of proteins plays an important role in the organization of cells cytoskeleton. Three of the Rho GTPases have key roles in cell movements, namely Cdc42, Rac1 and RhoA. Cdc42 is said to initiate filopodia projections, Rac1 is responsible for the development of lamellipodia, while RhoA is involved in stress fiber formation and focal adhesions [36]. Furthermore, Menager [36] reports that human endothelial cells, which have adhered to collagen or fibronectin, recruit RhoA to specific actin focal domains. These results suggest that a collagen or fibronectin coating initiates and enhances endothelial cell spreading and motility by activating important intracellular molecules, including the Rho GTPases.
Even if the positive effects of ECM coatings are known, there is limited research that examines the effect of single and double coatings on different cell types. As shown in our results, the adsorption of the antibodies directed against fibronectin (pFn) and only the triplet RGD (mRGD) was significantly higher on a single fibronectin coating than a double coating. However, the adsorption of the collagen antibody was similar for both the single collagen and double coating. Vandenberg [32] reports Albelda's results [37] suggesting that denatured collagen expose cell binding sites which can be bound also by fibronectin and Vn integrin receptors. ELISA and HUVEC proliferation results suggest that fibronectin is secreted by cells [38], and cells then bind endogenous secreted fibronectin with the coated collagen and fibronectin in a RGD dependent manner. Therefore, our results indicate that collagen had almost the same binding sites available when coated as a single or double coating.
Taken together, these results demonstrate that the presence on specific materials surface of more than one ECM molecule could represent a novel approach to improve the fast re-endothelialization of this support and lead to better biocompatibility.
Disclosure of interest
The authors declare that they have no conflicts of interest concerning this article.
Acknowledgement
We would like to thank all our colleagues at Université de technologie de Compiègne for their support and guidance. A special thanks to the Service d’analyse physicochimique at UTC for their technical assistance.


