1. Introduction
Organs and tissues consist of a precise arrangement of various cell types, working in concert to ensure optimal tissue function. Even small deviations from the tissue blueprint, for example in the composition of cell types or cellular phenotypes and behaviour, can lead to developmental defects, tissue malfunctions or the emergence of diseases. To prevent this, tissue quality control mechanisms facilitate the selective removal of defective cells. One such cell elimination mechanism is cell competition, through which cells of relatively lower fitness than their surroundings are removed (van Neerven and Vermeulen, 2022; Baker, 2020; Khandekar and Ellis, 2024) (Figure 1A). Competitive dynamics shape tissues during the whole animal’s lifespan. They are first observed at the blastocyst stage (Nichols et al., 2022), ensure robust pattern formation during morphogenesis (Tsai and Pinheiro, 2025) and may be an important factor in ageing-related processes (Marques-Reis and Moreno, 2021). Furthermore, cell competition can prevent the emergence of cancer through the removal of transformed cells (Cong and Cagan, 2024; Vishwakarma and Piddini, 2020). At the same time, mutated cells gaining a fitness advantage can eliminate the surrounding normal cells through competitive interactions, promoting cancer progression (Cong and Cagan, 2024; Vishwakarma and Piddini, 2020).
Cell competition shapes tissues by comparing relative cellular fitness levels. (A) General concept of cell competition: a population of cells shows higher fitness (winners) and is expanding at the cost of the population of lower fitness (losers). Loser cells are removed through cell elimination. (B) Cells compare their relative fitness to their surroundings. Multiple factors can influence which population wins and which one loses. (C) Loser cells are removed from the tissue via cell extrusion. This requires the coordinated exertion of forces within the removed cell and its neighbours. (D) Aim of this study: potential impact of force transmission differences on cell competition. Force transmission is mediated by cell–cell adhesion, which depends on the level of cadherin proteins.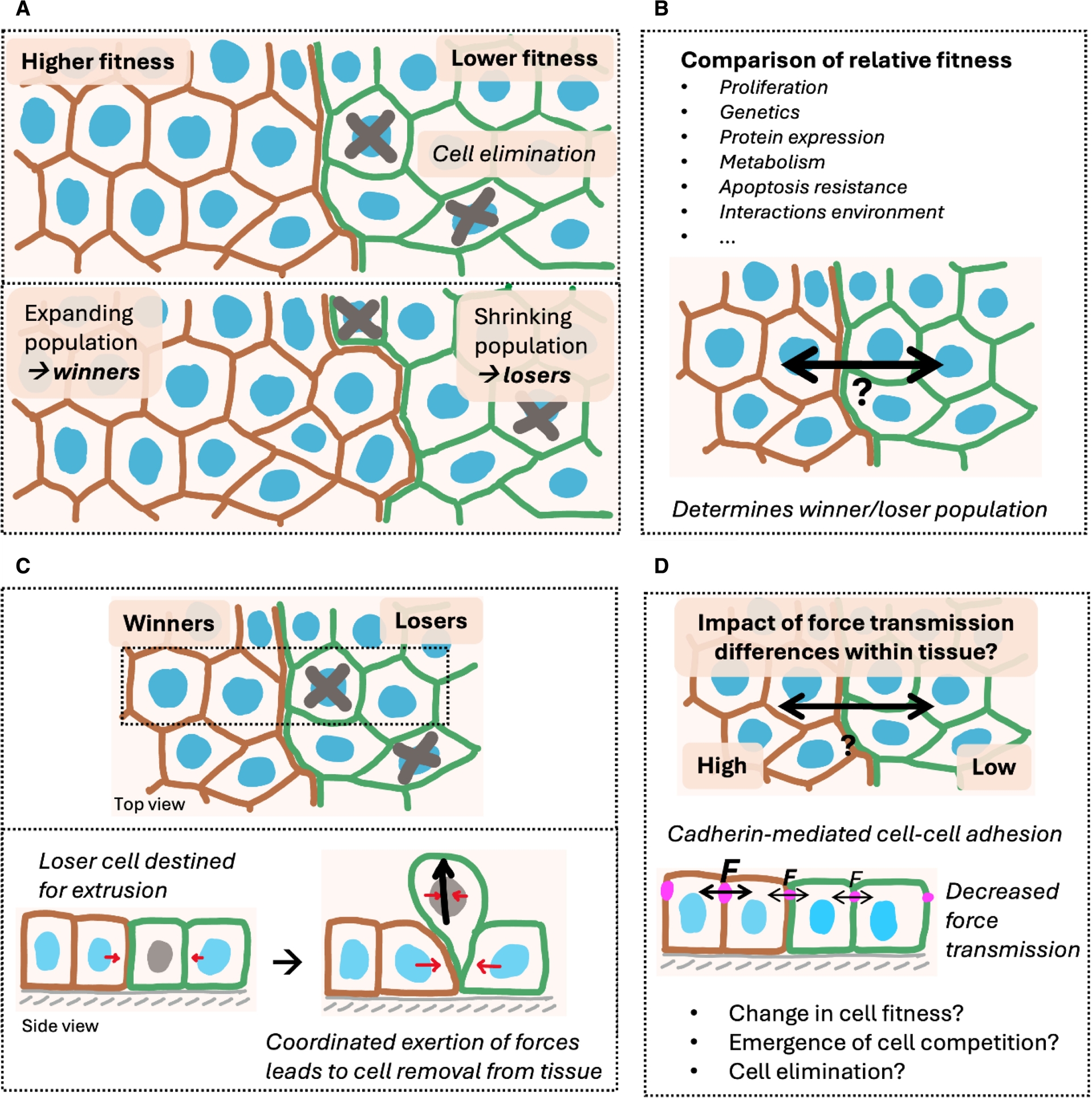
Those various examples highlight the need to understand what determines winning and losing, i.e. understanding how fitness changes are sensed and how they can lead to cell elimination through different mechanisms (Yusupova and Fuchs, 2023). Cellular fitness generally depends on the tissue context and can be influenced by multiple factors. Those include proliferation, genetics, protein expression, metabolic state, intrinsic resistance to death, but also the ways cells interact with their extracellular environment (van Neerven and Vermeulen, 2022; Baker, 2020; Khandekar and Ellis, 2024; Nichols et al., 2022; Tsai and Pinheiro, 2025; Marques-Reis and Moreno, 2021; Cong and Cagan, 2024; Vishwakarma and Piddini, 2020). They all could provide a competitive advantage or disadvantage conveying winner or loser status, respectively (Figure 1B). Loser cells are often eliminated via cell extrusion, which is an inherited mechanical process and requires the coordinated exertion of forces within the eliminated cell and its neighbors (Grata and Levayer, 2025; Ohsawa et al., 2018) (Figure 1C). However, frameworks integrating determinants of the loser cell status with the mechanism of cell elimination are often lacking.
Mechanical cell competition describes scenarios where cellular fitness and the elimination of loser cells are determined by tissue mechanics (Matamoro-Vidal and Levayer, 2019). There, compressive stresses lead to cell death and subsequent removal of loser cells. Compression can generally promote cell extrusion (Saw et al., 2017; Balasubramaniam et al., 2025) and can be mediated by collective migration (Wagstaff et al., 2016; Hundsdorfer et al., 2025), tissue crowding (Levayer et al., 2016), differential proliferation (Price et al., 2021), growth (Shraiman, 2005) and homeostatic cell densities (Gradeci et al., 2021; Basan et al., 2009) within mixed cell populations. Changing tissue mechanics has led to contradicting outcomes, e.g. when the mechanical properties of the extracellular environment were modulated (Aparicio-Yuste et al., 2022; Pothapragada et al., 2022) or when contractility was changed through the overexpression of Ras oncogenes (Levayer et al., 2016; Moitrier et al., 2019; Hogan et al., 2009; Porazinski et al., 2016). Thus, there is so far no consensus on how collective mechanical interactions and the cellular mechanical phenotype determine the competition outcome. Both critically depend on the fundamental process of force transmission, but its role in cell competition has not been explored yet.
In epithelia, intercellular force transmission, but also force sensing and force exertion, are mainly mediated by the adherens junction complex (Ladoux and Mège, 2017). A key component of it is E-cadherin (E-cad), a transmembrane protein that links neighbour cells to one another (Harris and Tepass, 2010). It has been shown to be crucial for efficient mechanical coupling between cells, transmission of mechanical information and collective tissue behaviour (Kale et al., 2018; Noordstra et al., 2023; Schoenit, Giudice, et al., 2022; Bazellières et al., 2015; de Beco et al., 2015). Consequently, we hypothesized that modulating force transmission through changing cadherin-mediated cell–cell adhesion strength may affect cellular fitness and lead to the emergence of cell competition (Figure 1D).
2. Summary
2.1. Force transmission provides a competitive advantage
In our recent work (Schoenit, Monfared, et al., 2025), we studied the impact of force transmission differences on competitive interactions within tissues. Importantly, such differences can be found in pathological conditions, for example within metaplastic breast cancers, which are a subtype of highly aggressive triple-negative breast cancers. They present a major therapeutic challenge because they consist of at least two cellular subpopulations, one epithelial-like and the other mesenchymal-like (Reddy et al., 2020). While E-cad is absent in the mesenchymal cells, it is strongly expressed in the epithelial-like subpopulation (Figure 2A). Due to this intratumoral force transmission heterogeneity, we assessed the emergence of potential cell competition through cultivating and monitoring patient-derived xenografts. We found that the E-cad positive epithelial-like cells expanded over time, thereby removing the E-cad negative mesenchymal-like cells from the tissue (Figure 2B). This indeed suggested that force transmission differences lead to intratumoral cell competition, and that increased cell–cell adhesion might increase cellular fitness.
Force transmission provides a competitive advantage. (A) Monolayer of heterogeneous patient-derived metaplastic breast cancer cells. E-cadherin in magenta marks the epithelial-like sub-population. Vimentin in cyan marks the mesenchymal-like sub-population. (B) Cluster development over time. The E-cad-expressing population is expanding. Red line shows the tissue boundaries after 20 h. (C) Top: cluster development in a mixed tissue consisting of MDCK WT and E-cad KO cells (green). Bottom: area quantification over time for different fractions of E-cad KO cells. (D) Left: map of the isotropic stress in the cancer tissue corresponding to (B). Right: quantification of the average isotropic stress within the two sub-populations. (E) Stress map (left) and stress quantification (right) of the competition between MDCK WT and E-cad KO corresponding to (C). Magenta outlines show the initial cluster boundaries. (D,E) P-values from unpaired t-tests. Every datapoint shows a single measurement in one frame out of n = 5 (D) or n = 14 (E) videos from N = 2 (D) or n = 4 (E) independent experiments. Data are presented as means ± s.d. Scale bars 200 μm (A,B,D), 100 μm (C,E). All figures reproduced from Schoenit, Monfared, et al. (2025).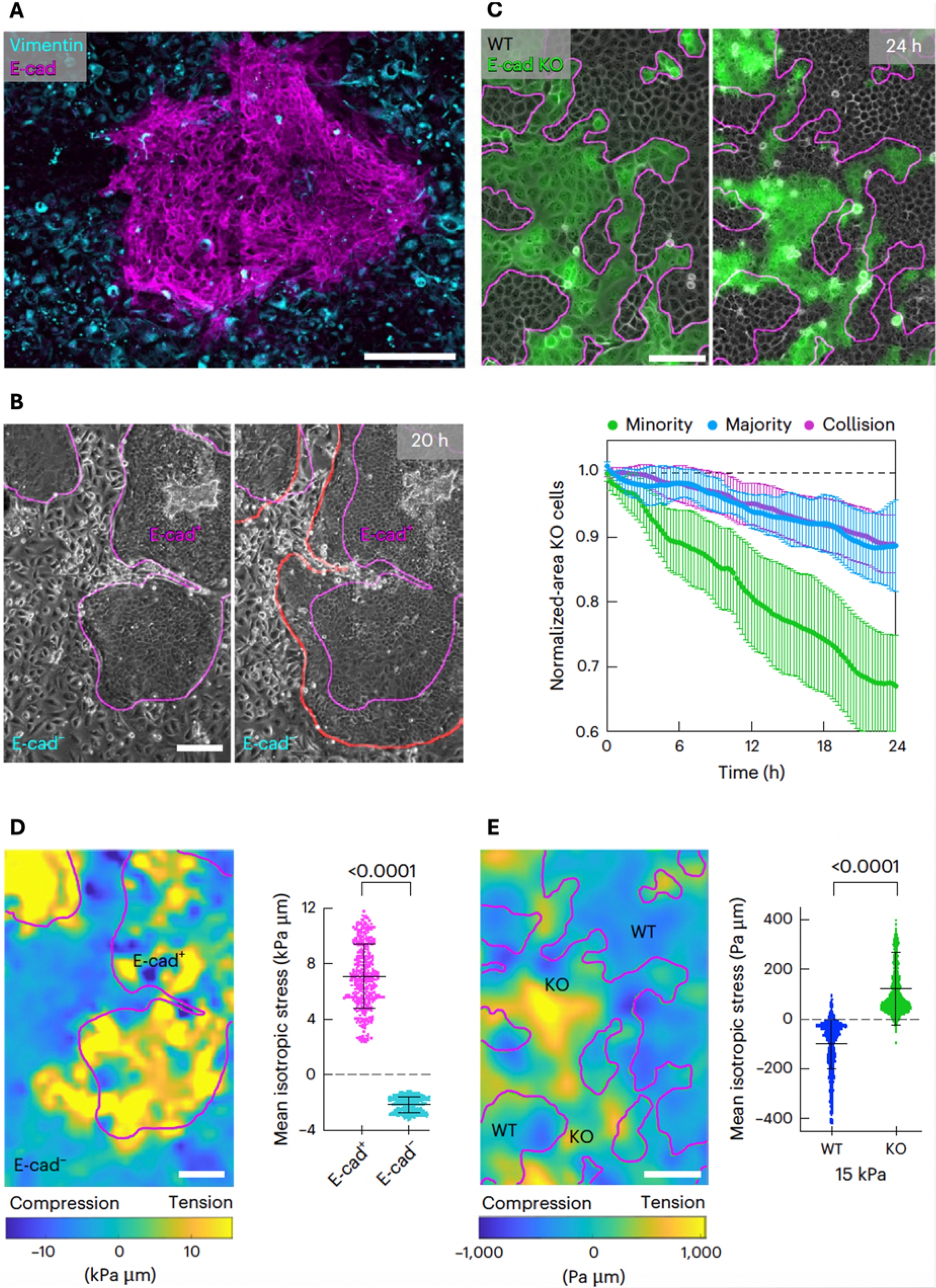
To investigate this hypothesis more systematically, we generated mixed cultures of epithelial cell lines, where the cell–cell adhesion strength of some cells was modulated through modifying the levels of cadherin expression. We tested various experimental conditions, including competition between MDCK and MCF10A WT versus E-cad KO cells, MDCK WT versus E-cad-overexpressing cells and MDCK E-cad KO versus E-cad/Cadherin-6 double-knockout cells. This experimental approach further offered the advantage that the ratios of competing cells, like MDCK WT competing against fluorescently labelled E-cad KO cells, can be precisely controlled (Figure 2C). Furthermore, boundary conditions like the interface geometry or the substrate mechanics can be modulated. We found across this wide range of experimental conditions that cells with relatively stronger force transmission mediated by their cell–cell adhesion levels are exclusively winning. This validated the observations in patient-derived tumours. It showed that force transmission increases cellular fitness and provides a crucial advantage in cell competition.
2.2. Loser cells are eliminated independently of compression at tissue interfaces
Compressive stress has been described as the main driver of cell elimination in mechanical cell competition (Wagstaff et al., 2016; Hundsdorfer et al., 2025; Levayer et al., 2016; Price et al., 2021; Shraiman, 2005; Gradeci et al., 2021; Basan et al., 2009). Therefore, we reasoned that increased cell–cell adhesion might provide a fitness advantage by allowing cells to collectively exert compressive stresses on their opponents. Unlike previous studies, our experimental approach enabled direct force and stress measurements using Bayesian Inversion Stress Microscopy (Nier et al., 2016). This method allows for a time-resolved and local readout of compression (negative isotropic stress) and tension (positive isotropic stress) within heterogeneous tissues (Anger et al., 2026).
In line with previous reports, we indeed observed in patient-derived tumour cultures that the winning E-cad positive cells were under tension, and the losing E-cad negative cells were under compression (Figure 2D). Surprisingly, however, we observed the opposite stress pattern between competing MDCK WT and E-cad KO cells, with the losing E-cad KO cells being under tension and the winning WT cells under compression (Figure 2E). Those measurements demonstrated that loser cells can be eliminated independently of compression, challenging the established consensus and suggest the existence of other, still unknown mechanisms of cell elimination.
Therefore, we investigated this competition scenario in detail. We first ruled out other previously conjectured fitness-increasing traits of winning cells, including cell-autonomous extrusion rates (Balasubramaniam et al., 2025), cell proliferation (Shraiman, 2005), crowding (Levayer et al., 2016) and homeostatic density (Gradeci et al., 2021; Basan et al., 2009), where we did not observe a difference between the subpopulations. Surprisingly, mechanical properties associated with increased fitness, namely cell stiffness and traction force generation (Hundsdorfer et al., 2025; Gradeci et al., 2021), were even increased in the losing E-cad KO cells. This suggests that they are overruled by increased cell–cell adhesion. The spatial distribution of cell extrusion events showed that losing E-cad KO cells were preferably eliminated close to the tissue interface (Figure 3A). Those interfaces were enriched in phospho-myosin, indicating increased mechanical activity (Figure 3B), suggesting a critical role of interface mechanical activity in cell elimination (Bielmeier et al., 2016).
Interface stress fluctuations lead to E-cad KO cell extrusion. (A) Left: representative phase contrast image of MDCK WT cells competing against E-cad KO cells (green). Red circles highlight cell extrusions. Right: probability distribution of cell extrusion as a function of the distance from the interface. Cells have a typical diameter of 10–15 cells. n = 14,729 KO and n = 11,031 WT extrusions from N = 4 independent experiments. (B) Immunostaining of phosphomyosin (red). E-cad KO cells in cyan. (C) Example simulation snapshot of the multiphase field model. (D) Fluctuations quantified as susceptibility of the 2D stress, i.e. the in-plane isotropic stress field and the zz, or out-of-plane component as a function of distance from the interface in simulations. (E) Susceptibility of the in-plane isotropic stress in experiments. (F) Example snapshots of a WT cell cluster undergoing strong area fluctuations. (G) Mirror situations. Extrusions are highlighted with red circles. (H) Schematic of the proposed mechanism of cell competition. Scale bars 50 μm (A), 25 μm (B). All figures from Schoenit, Monfared, et al. (2025).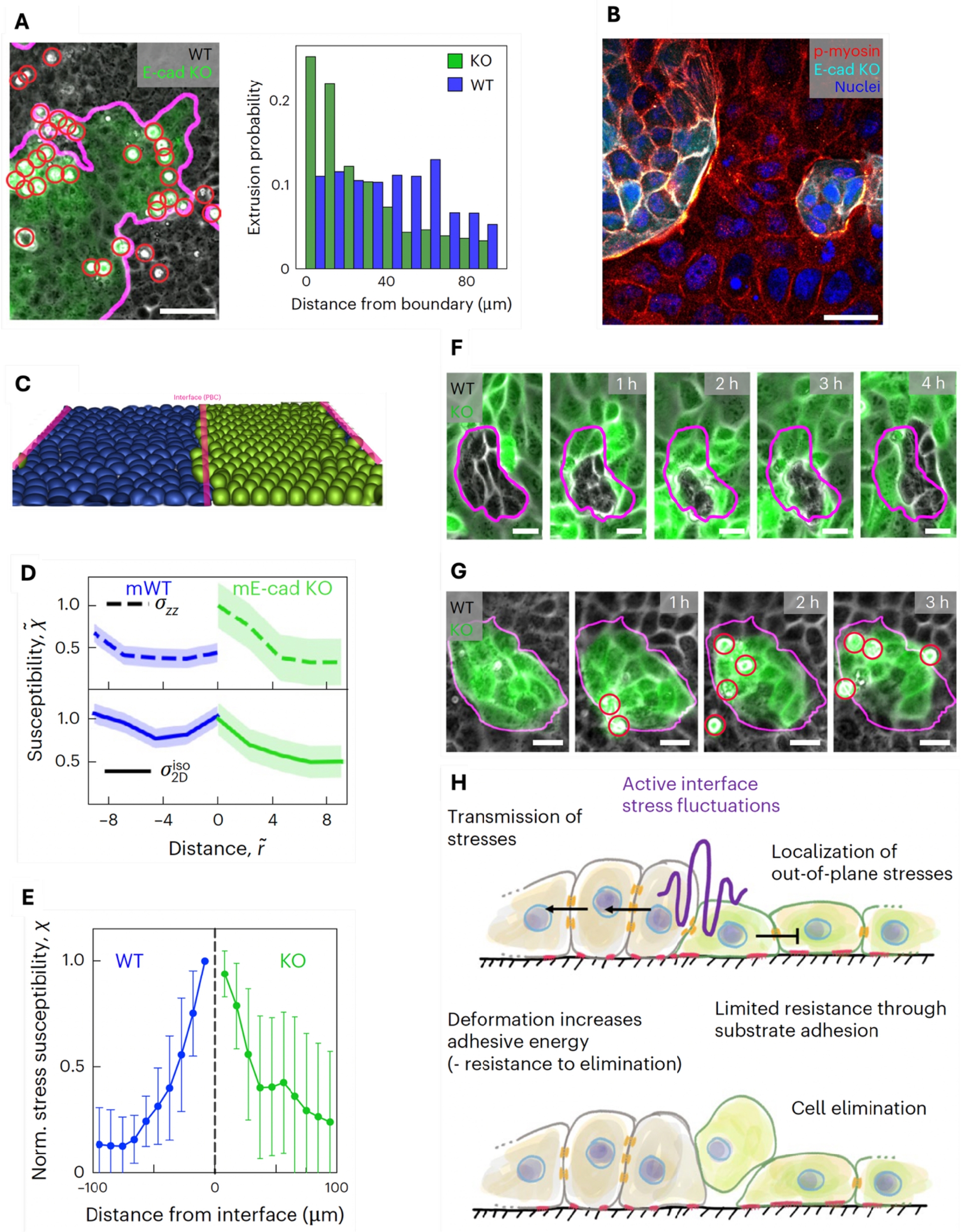
2.3. Stress fluctuations at active interfaces lead to E-cad KO cell elimination
To understand the contribution of interface mechanics to governing the cell competition outcome, we turned to 3D physical modelling using a multiphase field approach (Monfared et al., 2023) (Figure 3C), which provides access to the 3D stress. Simulations of competing model tissues showed that increased fluctuations, i.e. extreme tensile or compressive values of the in-plane isotropic stress, resulted in increased out-of-plane stresses leading to cell extrusion. To quantify those fluctuations, we measured the susceptibility X, and found that they occurred predominantly at the interface of the competing populations (Figure 3D). To test these predictions, we assessed stress fluctuations in our experimental data and indeed observed the same striking stress fluctuation increase at the interface, correlating with the location of cell eliminations (Figure 3E). Based on this combination of simulations and experiments, we proposed a new mode of cell extrusion through increased stress fluctuations. Importantly, it is not based on compression, and extruded cells can be under tension.
Since all cells at the tissue interfaces were subjected to those increased stress fluctuations, we then wondered why only E-cad KO cells were eliminated. We reasoned that, under such mechanically challenging conditions, increased force transmission might increase cellular fitness, as it could allow a more efficient redistribution of stresses across the tissue. Consequently, forces are shared among many cells, which improves the mechanical resistance of the collective. Indeed, we observed that small groups of WT cells could collectively sustain drastic area fluctuations through cell deformation without being extruded (Figure 3F). In mirror situations where E-cad KO islands were surrounded by WT cells, cells could not deform and stresses were released through cell elimination (Figure 3G). Taken together, we showed that increased force transmission increases mechanical cellular fitness because it allows winning cells to collectively resist elimination forces caused by stress fluctuations (Figure 3H).
3. Discussion and outlook
In this study (Schoenit, Monfared, et al., 2025), we discovered that cell–cell adhesion differences within epithelia can directly lead to cell competition. Cells with increased intercellular adhesion are exclusively winning, and we established adhesion-mediated force transmission as a crucial regulator of cell fitness across different scenarios of cell competition, including within patient-derived breast cancer xenografts. This principle could have broad implications in development and disease. Indeed, recent studies have shown that E-cad deficient cells are eliminated during zebrafish development to ensure correct pattern formation (Aoki et al., 2024). Moreover, increased adhesion allows winning cells to collectively exert compression on loser populations in vivo and in the context of cancer-associated mutations (Valon et al., 2025; Gupta et al., 2025).
Importantly, we demonstrated that the force transmission capacity can overrule other mechanical cellular properties like stiffness or force generation, and even allows winners to persist in a compressive environment, which was before considered the main driver of loser cell elimination (Wagstaff et al., 2016; Hundsdorfer et al., 2025; Levayer et al., 2016; Price et al., 2021; Shraiman, 2005; Gradeci et al., 2021; Basan et al., 2009). The surprising observation that cells under tension were in turn eliminated and became losers led to the identification of stress fluctuations as a new, compression-independent cell elimination mechanism. Reports of cell extrusion mediated by tension in intestinal organoids were recently published (Krueger et al., 2025), suggesting that similar mechanisms could govern cell elimination in physiological contexts. To withstand such cell fluctuations and to resist elimination, we showed that increased force transmission provides an inherently collective competitive advantage, which allows to dissipate elimination forces more effectively throughout the tissue.
The differences in force transmission studied here were particularly pronounced due to binary expression levels of adhesion molecules. However, biological and particularly pathological tissues, including different cancers, can exhibit more heterogeneous protein expression levels (Zhang et al., 2012). Thus, future studies need to address competition dynamics in such scenarios. Furthermore, research needs to address if and how the principles of mechanical cell competition proposed here affect the fate of cells on a longer timescale; that is, if invasion and subsequent metastasis of specific subpopulations could be promoted. Finally, we speculate that interface stress fluctuations might fulfil an important function in maintaining tissue boundaries by preventing the mixing of cell types and by removing undesired cells, which may have left their fate compartments.
Declaration of interests
The authors do not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and have declared no affiliations other than their research organizations.






 CC-BY 4.0
CC-BY 4.0