1. Introduction
Double J catheters are one of the most commonly used indwelling ureteral devices to treat obstructive uropathy, to facilitate the drainage of urine through the urinary system. However, they can promote the appearance of urinary tract infections (UTIs) and the development of encrustations, which can hinder and even prevent its removal [1, 2, 3].
Degree of embedding of the catheters studied. Image obtained by stereoscopic microscopy.
Types of deposits observed. Images obtained by Scanning Electron Microscopy (SEM).
The introduction of a catheter into the urinary system implies the placement of a foreign body that facilitates the flow of urine between the renal pelvis and the urinary bladder. Therefore, the catheter will be permanently bathed in urine that will flow from the kidney to the urinary bladder. As it is a foreign solid in contact with urine, it is evident that it may act as a heterogeneous nucleant for those substances that are supersaturated in this medium. It is precisely for this reason that extensive research has been carried out to develop nonstick materials that avoid as much as possible the capacity that a catheter can present as a heterogeneous nucleant. In any case, urine is a medium rich in organic matter and proteins, mainly albumin and mucin in the absence of infections, which can adhere on the surface of the catheter covering it [4]. Once deposited, this material will act as an effective heterogeneous nucleant that will induce crystallization of those substances that are supersaturated in urine.
The main objective of this paper is to characterize and study the different types of encrustations that form on double J catheters, in order to be able to relate their characteristics to the urinary conditions that have induced them.
2. Material and methods
Ninety double J catheters (Angiomed, UROSOFT-ureteral-stent-set based on a soft-polyurethane compound, REF 57410030) of patients with an indwelling time approximately from 1 to 3 months were studied, processing the renal part and the bladder part separately. Thus each catheter is divided into two equal parts. This was an observational study, where 57.8% of the patients were males (age 57 ± 15) and 42.2% females (age 54 ± 13).
(A) Percentage of no deposit or low, medium and high degree of encrustation in the 90 stents. (B) Percentage of each type of deposit in the 90 stents.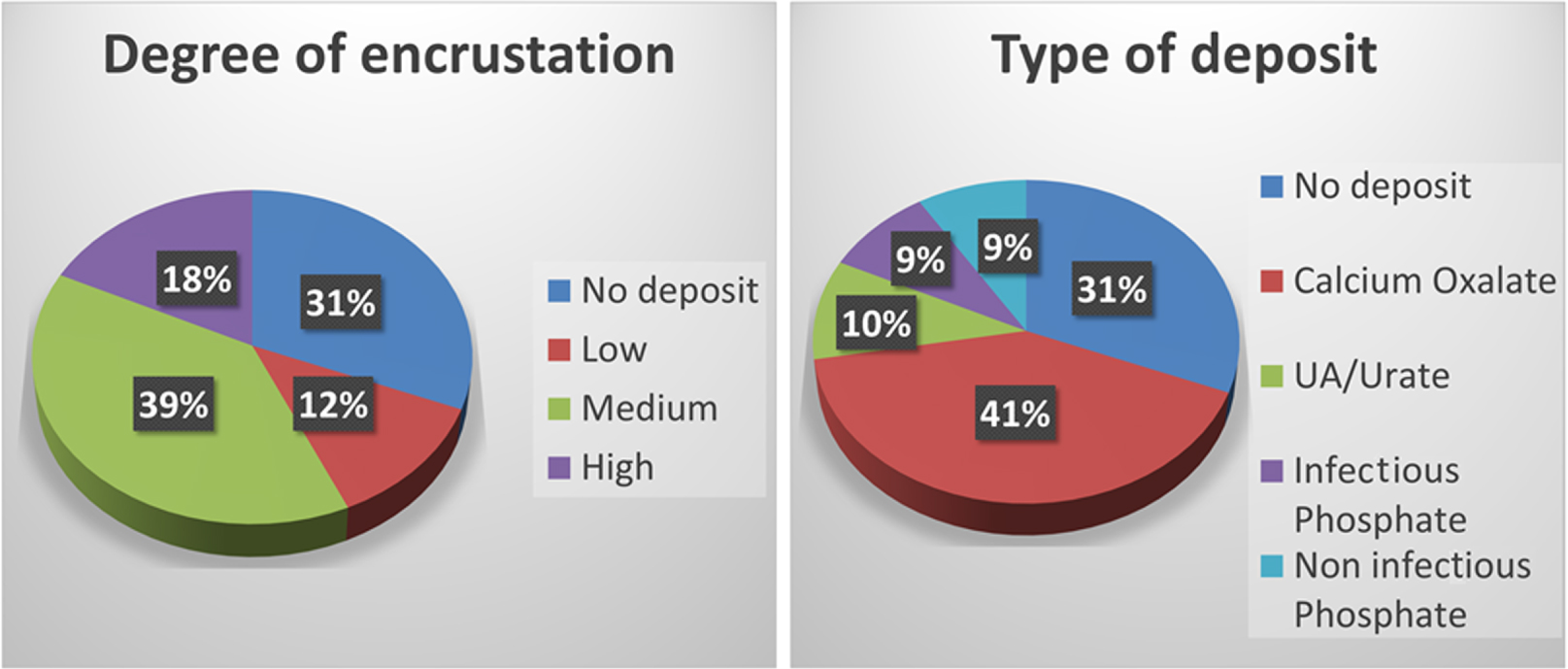
In all cases, the stent was placed as a consequence of ureteral obstruction due to benign pathology.
Each catheter was submitted to the following procedure: First, it was photographed with a stereoscopic microscope. By observational study, they were classified into four categories according to the degree of encrustation observed (0–3, 0 being no deposit and 3 high degree of encrustation or deposit block, as shown in Figure 1). Subsequently, each stent was weighed and the total weight of deposit present in each catheter was evaluated, by comparison with an unused catheter. Later, by Scanning Electron Microscopy (SEM) the type of embedded deposit was identified: no deposit, calcium oxalate (monohydrate, dihydrate), uric acid (anhydrous, dihydrate) and/or urate, infectious phosphate (magnesium ammonium phosphate + hydroxyapatite) and noninfectious phosphate (hydroxyapatite + brushite) (Figure 2).
Finally, the catheters were cut into several fragments to facilitate dissolution of the deposits with 2 M HCl (except for those with uric acid and/or urate) to determine the amount of calcium, phosphorus and magnesium by ICP-OES.
2.1. Statistics
Normality graphs and plots were used to assess the data distribution. Data were represented as median and interquartile range or percentages. Intergroup comparisons (between deposit types and deposit grades) were performed using Kruskal–Wallis test and the Mann–Whitney U as post hoc test for continuous variables. For categorical variables, the Chi-square or Fisher’s exact test was performed. A two-tailed p-value less than 0.05 was considered statistically significant. Statistical analyses were performed using SPSS 25.0 (SPSS Inc., Chicago, IL, USA).
3. Results
Of the 90 catheters studied, most (39%) presented a medium degree of embedding (degree 2), and 41% had calcium oxalate (CaOx) deposits. 31% did not have any type of deposit on their surface (Figure 3).
Twenty patients (22%) were diagnosed with UTIs and 15% amongst them developed magnesium ammonium phosphate deposits.
Twenty four percent of the patients did not present deposits three months after catheter placement, while 68% formed medium/high deposits during this period.
Whereas magnesium ammonium phosphate + hydroxyapatite appeared in 9% of the catheters (Figure 3B), this composition corresponded more frequently to the high degree of encrustation group (43.8%, p value < 0.05; Figure 4), while in contrast CaOx was the most frequent composition of the low and medium degree of encrustation (63.3% and 80% respectively, p value < 0.05, Figure 4). Hydroxyapatite with no infection was also identified in 9% of catheters (Figure 3B) equally distributed at low, medium and high degree of encrustation (Figure 4), and uric acid was detected in 10% of catheters, mainly with high degree of encrustation (31%, Figure 4).
Percentage of type of deposit in each grade of deposit group. (a) Significant differences with the Low deposit group. (b) Significant differences with the Medium deposit group. (c) Significant differences with the High deposit group. Differences were considered significant if p value < 0.05 in the Chi-square or Fisher’s exact test.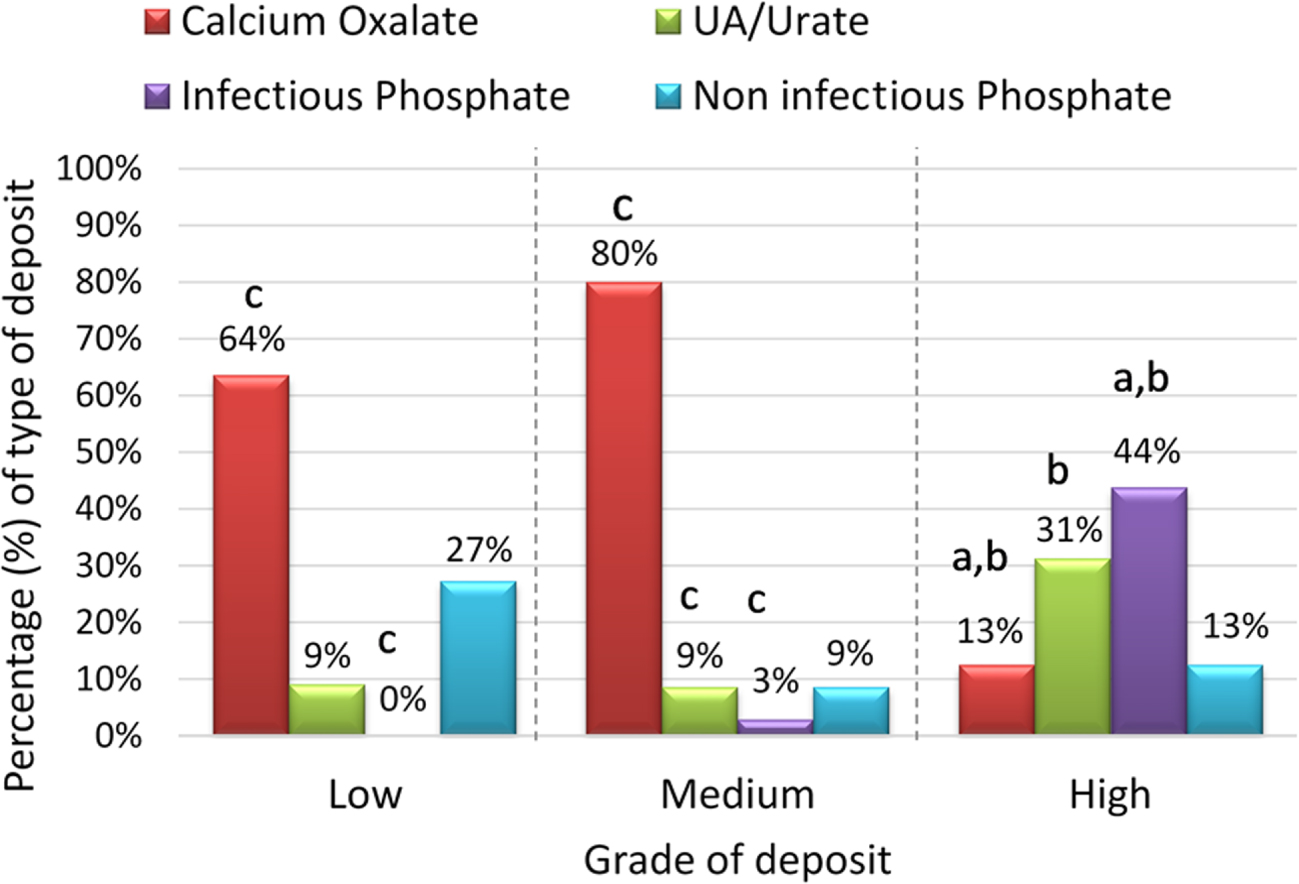
The qualitative classification was compared with the total weight of the stents (Figure 5). A positive and high correlation between the deposit weight of the 90 stents and the degree of deposit is observed (rs = 0.777, p value < 0.001), with greater the weight of the deposit on the stent, the higher the degree of encrustation (Figure 5).
Deposit weight for each degree of encrustation group. Medians are represented in the figure. * Significant differences with the no deposit and low deposit group, p-value < 0.05 Kruskal–Wallis test and the Mann–Whitney U as post hoc test. #Significant differences with the medium group, p-value < 0.05 Kruskal–Wallis test and the Mann–Whitney U as post hoc test. Correlation between the two variables was assessed with the Spearman test, rs = 0.777, p value < 0.001.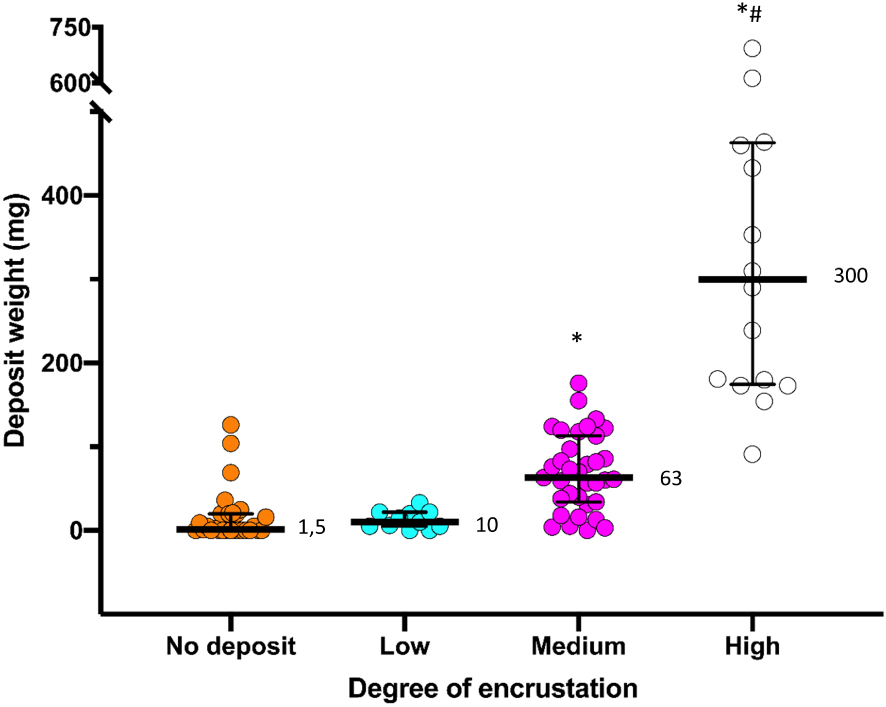
In concordance with these findings, it is also seen that the higher the degree of encrustation, the greater the amount of calcium, phosphorus and magnesium quantified for each type of deposit (Table 1). Catheters with greater weight of deposited material (median = 393 mg, Figure 6) presented mostly an infectious phosphate-type deposit. When comparing the renal and bladder parts of the catheters, no difference between the degree of encrustation or the type of deposit was observed (Tables 2 and 3).
Deposit weight for each type of encrustation group. Medians are represented in the figure. * Significant differences with all the other groups, p-value < 0.05 Kruskal–Wallis test and the Mann–Whitney U as post hoc test.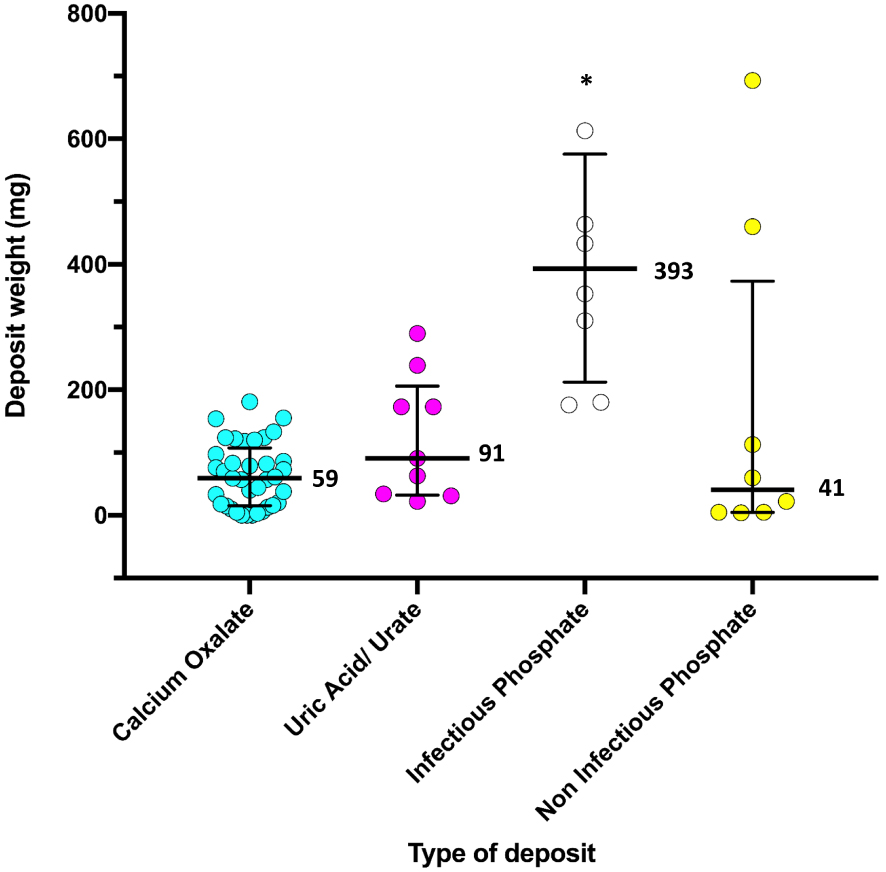
Ca, P and Mg, expressed as median value, detected for each degree of deposit per type of deposit
| Type of deposit | Degree of deposit | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ca (μmol) | P (μmol) | Mg (μmol) | |||||||
| L | M | H | L | M | H | L | M | H | |
| Calcium oxalate | 46.4 | 351 | 876 | 3.74 | 12.8 | 30.3 | 0.23 | 0.87 | 1.27 |
| Infectious phosphate | — | 486∗ | 1687 | — | 784∗ | 1202 | — | 366∗ | 235 |
| Noninfectious phosphate | 4.99 | 390 | 3607 | 8.1 | 298 | 2674 | 0.43 | 10.4 | 112 |
Degree of deposit: L = Low, M = Medium, H = High.
∗ n = 1, value in μmol of the case.
Percentage of each degree of encrustation in the renal and bladder parts
| Degree of encrustation (%) | |||||
|---|---|---|---|---|---|
| No deposit | Low | Medium | High | Total | |
| Renal | 35.6% | 21.1% | 34.4% | 8.9% | 100.0% |
| Bladder | 32.2% | 13.3% | 40.0% | 14.4% | 100.0% |
4. Discussion
The proposed sample processing method is effective when observing a good correlation between the degree of deposit and the weight obtained for each deposit (Figure 5).
Percentage of each type of encrustation in the renal and bladder parts
| Type of deposit (in %) | ||||||
|---|---|---|---|---|---|---|
| No deposit | CaOx | Uric acid/urate | Infectious phosphate | Noninfectious phosphate | Total | |
| Renal | 33.3 | 40.0 | 10.0 | 7.8 | 8.9 | 100.0 |
| Bladder | 32.2 | 41.1 | 8.9 | 8.9 | 8.9 | 100.0% |
From this study it is deduced that the deposit that generates the most trouble, due to its magnitude, is that of magnesium ammonium phosphate/hydroxyapatite. In the presence of ureolytic bacteria, large amounts of uromucoid and a significant increase in urinary pH (as a consequence of the formation of NH3) are produced. Under these conditions, hydroxyapatite and magnesium ammonium phosphate precipitate on the catheter [5, 6, 7, 8]. Therefore, the presence of urinary infection, which causes this type of deposits, should be controlled and avoided as far as possible. Precisely, as a very long treatment with antibiotics is not advisable, all those prophylactic measures to avoid urinary infection should be taken into account [9]. Among these measures, the most important are to avoid persistent high urinary pH which promotes bacterial infection, since acidic urinary pH values facilitate the transformation of nitrite generated by bacteria (from nitrate present in the urine) into NO, and this compound is toxic to bacteria [10, 11]; it is advisable to drink large amounts of water and maintain strict hygienic conditions. It is interesting to observe that though 22% of the patients were diagnosed with UTIs, only 15% of them developed magnesium ammonium phosphate deposits. This is probably because these patients were initially treated with antibiotics.
It is important to observe how in a large number of patients (80%, Figure 4), calcium oxalate dihydrate was detected in medium degree of encrustation (degree 2) and even in a small percentage (13%, Figure 4) in the high encrustation (degree 3) group. This circumstance shows that in patients with hypercalciuria, control of calciuria should be carried out during the ureter implantation period, avoiding as far as possible high concentrations of urinary calcium, which should be maintained below 170 mg/L. In the absence of ureolytic bacteria, it is necessary to consider that, since it is advisable to keep double J catheters for short periods (maximum 2–3 months), only substances with very considerable supersaturations will crystallize massively [12, 13].
It has also been observed that in the absence of infection, hydroxyapatite deposits in low degree of encrustation (27%) or medium/high grade of encrustation (9% and 13% respectively; Figure 4) are generated on the catheter in a certain group of patients, which could compromise its easy removal. In these cases, it is clear that the formation of these deposits is a consequence of the existence of persistently high urinary pH values. Therefore, as far as possible, attempts should be made to acidify these urines when it is possible and, in the presence of distal tubular acidosis, which precludes acidification, appropriate prophylactic measures should be taken [14].
The formation of large uric acid/urate deposits (31%) or medium and low grade of encrustation (9%, in both cases; Figure 5) has also been observed. These deposits must be clearly attributed to the persistence of urinary pH values below 5.5 and also to the possibility of elevated urinary uric acid concentration, when uric acid dihydrate was observed. Therefore, excessively acidic urinary pH values below 5.5 should also be avoided.
As can be deduced, with the exception of high consumption of liquids, the other preventive measures must be adapted to the urinary composition of each patient, to avoid the implementation of contraindicated treatments.
The degree of encrustation on the stents is quite high, even for time periods shorter than one month, since 11% developed high grade deposits during this period. This shows that polyurethane, which is the material composing these stents, develops important deposits with relative ease, which is consistent with a recently published study that shows that this material generates deposits more easily than others such as hydrocoated silicone [15].
5. Conclusion
To prevent the development of large encrustations in the double J catheters, strict control should be carried out to prevent urinary infections, high urinary pH values (above 6.2) or too low values (below 5.5). In addition, control of excessive urinary calcium levels is also important.




 CC-BY 4.0
CC-BY 4.0