1. Introduction
In the α-CrPO4 structure [1], the Cr3+ and P5+ cations occupy four special positions of the Imma space group. Its corresponding framework is constructed on the basis of [CrO6] octahedra and [PO4] tetrahedra, revealing vacant channels along the [100] and [010] axis. Accordingly, this phosphate can be formulated as A1A2Cr1Cr22(PO4)3, with the channel vacant sites represented by A1, A2. As one of our points of interest is to elaborate new α-CrPO4-related open-framework phosphates, we have focused our research on the mixed substitution of di- and/or trivalent cations for Cr1 and/or Cr2. Such a substitution leads to the charge compensation requiring the localization of monovalent or divalent cations in the A1 and/or A2 channels. Hence, using the hydrothermal process, we have managed to isolate new phosphates Ag2M3(HPO4)(PO4)2 (M = Co or Ni) [2, 3], besides the rarely encountered mixed-valence manganese phosphates MMn(II)Mn(III)2(PO4)3 (M = Sr, Ba, or Pb) [4, 5, 6]. Moreover, the solid-state reaction technique has allowed us to isolate a variety of new other bi- and trivalent-cations-based phosphates with an α-CrPO4 structure type, namely α-Na2Ni2Fe(PO4)3 [7], NaCuCr2(PO4)3 [8], NaZnCr2(PO4)3 [9], BaNi2Fe(PO4)3 [10, 11, 12], and M′Co2Fe(PO4)3 (M′ = Sr or Ba) [13, 14].
These phosphates exhibit interesting physicochemical properties. For example, NaCoCr2(PO4)3 and its isotypes NaNiCr2(PO4)3 and Na2Ni2Cr(PO4)3 can serve as cathode materials for sodium-ion batteries, owing to their high discharge capacities of 352, 385, and 368 mAh⋅g−1, respectively [13]. Recently, Matsaev et al. have reported a novel series of iron-based phosphates, α-Fe1−xCrxPO4, adopting the α-CrPO4 structure type, capable of Li-ion intercalation in a high potential region.
The present work is devoted to the synthesis and crystal structure study of the two new magnesium phosphates AMg2Fe(PO4)3 (A = Sr or Ba) with the α-CrPO4 structure type.
2. Experimental
2.1. Crystal growth
Single crystals of the two compounds AMg2Fe(PO4)3 (A = Sr or Ba) were grown by solid-state reaction. The crystals were prepared from a stoichiometric mixture of ACO3 (A = Sr or Ba) (BaCO3: Sigma-Aldrich, 98%; SrCO3: BaCO3: Seelze–Hannover, 99%), Mg(NO3)2⋅6H2O (Sigma-Aldrich, 97%), Fe(NO3)3⋅9H2O (Panreac, 98%) and NH4H2PO4 (Alfa Aesar, 98%). The mixture was progressively heated in a platinum crucible to 573, 673, 773, and 873 K, maintaining each temperature for one night, combined with intermediate grindings. The products were then heated slowly at a rate of 5 °C/min, to 1473 K in the Sr case and to 1353 K for Ba. The final product was maintained at the final temperature for 10 min followed by cooling at room temperature at a rate of 5 °C/h. Finally, green single crystals were isolated from the Sr-containing preparation while brown crystals were obtained with Ba.
2.2. Crystal structure study
A single crystal of each preparation was chosen carefully under microscope with suitable dimensions for X-ray diffraction (XRD) studies. The XRD data were collected using a Bruker D8 Venture Super DUO diffractometer with a PHOTON100 CMOS area-detector and monochromatic MoKα radiation (𝜆 = 0.71073 Å) at room temperature. The APEX3 [15] software was used for data collection and the data were integrated with the SAINT-Plus program. Absorption corrections were performed by a multiscan semi-empirical method using SADABS [16]. The two crystal structures were solved by the Patterson method and refined by the SHELXT 2014 [17] and SHELXL 2018 [18] programs incorporated in the WinGX interface [19]. The structural graphics were drawn using the DIAMOND program [20].
3. Results and discussion
Table 1 summarizes the crystal data and structural refinement results. For both structures AMg2Fe(PO4)3 (A = Sr or Ba), the refinement of all ions in anisotropy led to good reliability factors (R1 = 0.018 and wR2 = 0.047 for SrMg2Fe(PO4)3, R1 = 0.012 and wR2 = 0.032 for BaMg2Fe(PO4)3).
Crystal data and structure refinement of AMg2Fe(PO4)3 (A = Sr or Ba)
| Compound | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
|---|---|---|
| Crystal data | ||
| Crystal system | Orthorhombic | Orthorhombic |
| Space group | Imma | Imma |
| Cell dimension (Å) | 10.3998 (2) | 10.4996 (2) |
| 13.2000 (2) | 13.2696 (1) | |
| 6.5364 (1) | 6.6330 (2) | |
| Cell volume (Å3) | 897.30 (3) | 924.15 (3) |
| Multiplicity Z | 4 | 4 |
| Molecular weight (g/mol) | 477 | 526.72 |
| Density (g/cm−3) | 3.531 | 3.786 |
| Coefficient absorption 𝜇 (mm−1) | 8.31 | 6.53 |
| Data collection | ||
| Diffractometer | Brüker X8 APEX- CCD | Brüker X8 APEX- CCD |
| No. of measured, independent, and observed [I⩾2𝜎(I)] reflections | 9561, 892, 876 | 22556, 1671, 1595 |
| Rint | 0.035 | 0.032 |
| 𝜃min–𝜃max (°) | 3.1–32.5 | 3.1–41.5 |
| Refinement | ||
| R[F2 > 2𝜎(F2)] | 0.018 | 0.012 |
| wR2(F2) | 0.047 | 0.032 |
| S (Goodness-of-Fit): | 1.19 | 1.16 |
| No of refined parameters | 54 | 58 |
| No. of restraints | 0 | |
| Δ𝜌max, Δ𝜌min (e⋅Å−3) | 0.58, −0.82 | 1.38, −1.03 |
The two compounds crystallize in the Imma space group with the following unit cell parameters: a = 10.3998 (2) Å, b = 13.2000 (2) Å, c = 6.5364 (1) Å, and V = 897.30 (3) Å3 for the strontium-based phosphate and a = 10.4996 (2) Å, b = 13.2696 (1) Å, c = 6.6330 (2) Å, and V = 924.15 (3) Å3 for the barium-based phosphate.
The atomic coordinates and equivalent isotropic displacement parameters are given in Table 2. The atomic anisotropic displacement parameters are presented in Table 3. The bivalent cation (site = Ba1 or Sr1) is refined in the special position (4e). The P5+ ions (sites named P1 and P2) are located in the special Wyckoff positions (4e) and (8g), respectively. Two sites of the O2− ions, named O1 and O2, are refined in the special positions (8h) and (8i) respectively, while the other O2− ions (O3 and O4 sites) are in the general position (16j).
Atomic coordinates, occupancies, and equivalent isotropic displacement parameters (Å2) of SrMg2Fe(PO4)3 and BaMg2Fe(PO4)3*
| Atom | Site | Occupation | x | y | z | Uéq |
|---|---|---|---|---|---|---|
| Sr1 | 4e | 1 | 0.0000 | 0.7500 | 0.59942 (3) | 0.00871 (8) |
| Ba1* | 0.0000 | 0.7500 | 0.60778 (2) | 0.00789 (4) | ||
| Fe1/Mg1 | 4a | 0.880 (1)/0.120 (1) | 0.5000 | 0.5000 | 0.5000 | 0.00398 (9) |
| Fe1/Mg1* | 0.871 (1)/0.129 (1) | 0.5000 | 0.5000 | 0.5000 | 0.00384 (5) | |
| Mg2/Fe2 | 8g | 0.940 (1)/0.060 (1) | 0.7500 | 0.63349 (4) | 0.2500 | 0.0053 (1) |
| Mg2/Fe2* | 0.871 (1)/0.129 (1) | 0.7500 | 0.63293 (3) | 0.2500 | 0.00283 (7) | |
| P1 | 4e | 1 | 0.0000 | 0.7500 | 0.09209 (8) | 0.0038 (1) |
| P1* | 0.0000 | 0.7500 | 0.10104 (8) | 0.00341 (8) | ||
| P2 | 8g | 1 | 0.7500 | 0.42840 (3) | 0.2500 | 0.0043 (1) |
| P2* | 0.7500 | 0.42840 (3) | 0.2500 | 0.0043 (1) | ||
| O1 | 8h | 1 | 0.0000 | 0.65613 (9) | −0.0436 (2) | 0.0071 (2) |
| O1* | 0.0000 | 0.65524 (8) | −0.0294 (2) | 0.0065 (2) | ||
| O2 | 8i | 1 | 0.8822 (1) | 0.7500 | 0.2362 (2) | 0.0064 (2) |
| O2* | 0.8813 (1) | 0.7500 | 0.2390 (2) | 0.0059 (2) | ||
| O3 | 16j | 1 | 0.71114 (9) | 0.36450 (7) | 0.0665 (1) | 0.0081 (2) |
| O3* | 0.71689 (9) | 0.36643 (6) | 0.0661 (1) | 0.0082 (1) | ||
| O4 | 16j | 1 | 0.63860 (8) | 0.50528 (6) | 0.2950 (1) | 0.0062 (2) |
| O4* | 0.63800 (7) | 0.50498 (6) | 0.2930 (1) | 0.0062 (1) |
Anisotropic displacement parameters (Å2) of SrMg2Fe(PO4)3 and BaMg2Fe(PO4)3*
| Atom | U11 | U22 | U33 | U12 | U13 | U23 |
|---|---|---|---|---|---|---|
| Sr1 | 0.0091 (1) | 0.0114 (1) | 0.0056 (1) | 0.000 | 0.000 | 0.000 |
| Ba1* | 0.00716 (6) | 0.01205 (6) | 0.00447 (5) | 0.000 | 0.000 | 0.000 |
| Fe1/Mg1 | 0.0034 (2) | 0.0041 (2) | 0.0044 (2) | 0.000 | 0.000 | 0.0003 (1) |
| Fe1/Mg1* | 0.0034 (1) | 0.0040 (1) | 0.0041 (1) | 0.000 | 0.000 | 0.00011 (8) |
| Mg2/Fe2 | 0.0051 (2) | 0.0040 (2) | 0.0067 (2) | 0.000 | 0.0002 (2) | 0.000 |
| Mg2/Fe2* | 0.0027 (2) | 0.0013 (2) | 0.0045 (2) | 0.000 | 0.0003 (1) | 0.000 |
| P1 | 0.0038 (2) | 0.0034 (2) | 0.0043 (2) | 0.000 | 0.000 | 0.000 |
| P1* | 0.0033 (2) | 0.0033 (2) | 0.0036 (2) | 0.000 | 0.000 | 0.000 |
| P2 | 0.0047 (2) | 0.0038 (2) | 0.0044 (2) | 0.000 | 0.0005 (1) | 0.000 |
| P2* | 0.0040 (1) | 0.0039 (1) | 0.0040 (1) | 0.000 | 0.0005 (1) | 0.000 |
| O1 | 0.0088 (5) | 0.0050 (5) | 0.0074 (5) | 0.000 | 0.000 | −0.0018 (4) |
| O1* | 0.0086 (4) | 0.0039 (3) | 0.0069 (4) | 0.000 | 0.000 | −0.0018 (3) |
| O2 | 0.0051 (5) | 0.0071 (5) | 0.0070 (5) | 0.000 | 0.0015 (4) | 0.000 |
| O2* | 0.0042 (4) | 0.0076 (3) | 0.0059 (4) | 0.000 | 0.0016 (3) | 0.000 |
| O3 | 0.0101 (4) | 0.0074 (4) | 0.0067 (3) | −0.0018 (3) | 0.0002 (3) | −0.0022 (3) |
| O3* | 0.0096 (3) | 0.0085 (3) | 0.0065 (3) | −0.0021 (2) | 0.0003 (2) | −0.0027 (2) |
| O4 | 0.0053 (3) | 0.0059 (3) | 0.0075 (3) | 0.0012 (3) | 0.0015 (3) | 0.0006 (3) |
| O4* | 0.0047 (3) | 0.0064 (2) | 0.0073 (3) | 0.0011 (2) | 0.0017 (2) | 0.0008 (2) |
However, the position refinement of cations Fe3+ and Mg2+ in the special positions (4e) and (8g), respectively, results in negative atomic displacement parameters for Mg2+, a high-density residue in the vicinity of Mg2+, and a density deficit in the vicinity of Fe3+, despite the very good R1 and wR2 factors obtained. Those facts suggest the concomitant localization of Fe3+ and Mg2+ in the same site. Such an assumption is established by the last refinement assuming, firstly, the localization of both Fe3+ and Mg2+ in the same crystallographic Wyckoff position (4a), initially assigned only to Fe3+. The occupancy rate obtained in this site, hereafter named Fe1/Mg1 site, corresponds to 0.880 (1) for Fe3+ and 0.120 (1) for Mg2+ in the strontium compound. Likewise, the remaining amount of Fe accommodates the site of Mg2 at the special Wyckoff position (8g), hereafter called Mg2/Fe2 site in the strontium phosphate. Such refinement led to an occupancy of 0.940 (1) for Mg2+ and 0.060 (1) for Fe3+ at the Mg2/Fe2 site. Therefore, refining the Fe/Mg site occupancy ratios in the case of the Sr compound, taking into account the electrical neutrality of the molecule, leads to a stoichiometric compound with a Mg/Fe ratio of two and therefore a SrMg2Fe(PO4)3 formula. In contrast, in the case of the Ba compound, the Mg/Fe ratio is slightly less than two (7.482 (1)/4.518 (1)), and the electrical neutrality of the molecule is no longer maintained. This can be explained by the presence of a small amount of Fe2+, and consequently, the formula of the resulting compound will be BaMg1.87Fe0.13Fe(PO4)3.
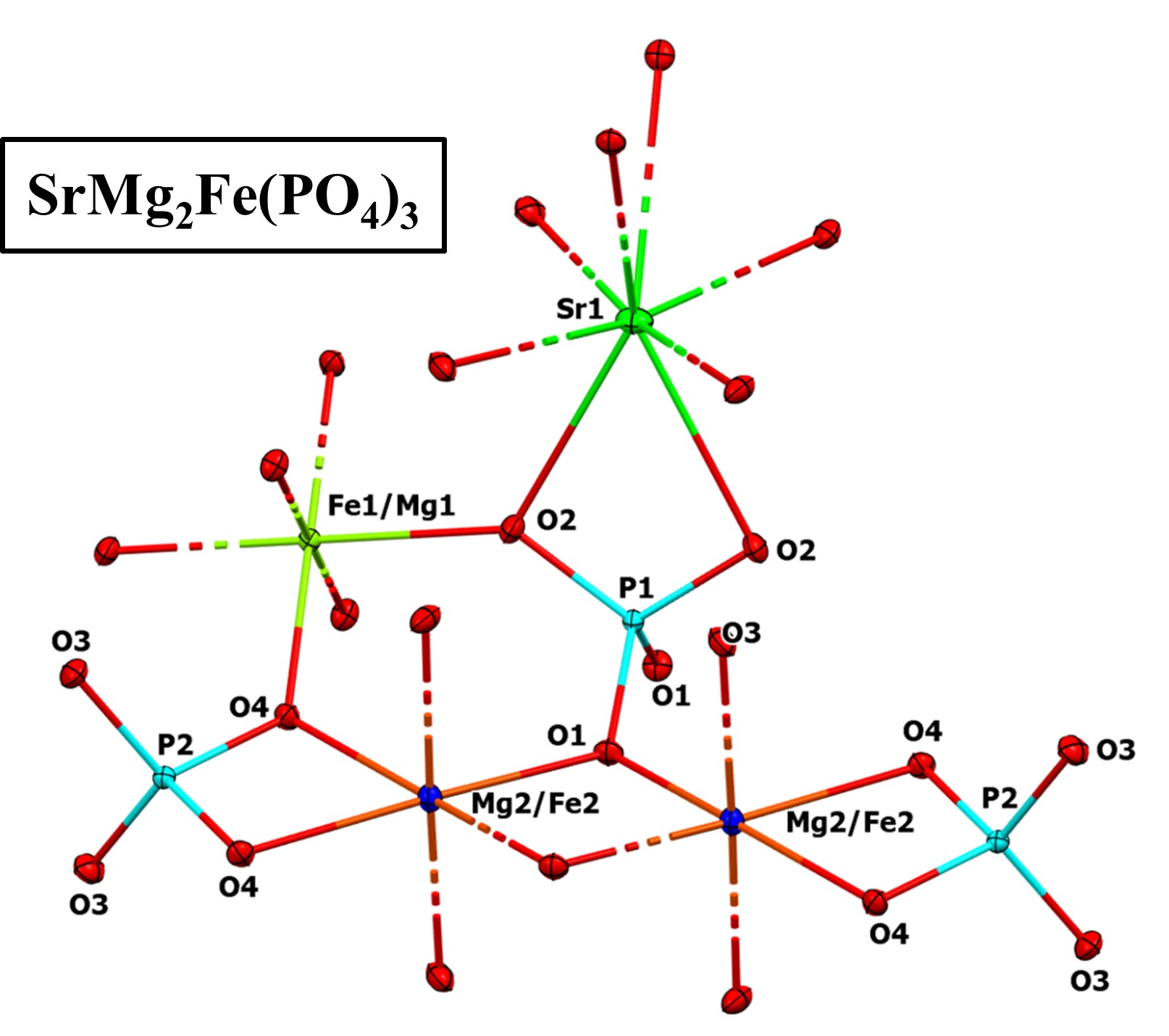
Selected bond lengths and angles in both compounds AMg2Fe(PO4)3 (A = Sr or Ba) are listed in Table 4.
Interatomic distances (Å) and angles (°) for AMg2Fe(PO4)3 (A = Sr or Ba)
| P–O distances (Å) and O–P–O angles (°) | ||
| Distance (Å) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| P1–O1 | 1.524 (1) | 1.526 (1) |
| P1–O1ix | 1.524 (1) | 1.526 (1) |
| P1–O2ix | 1.546 (1) | 1.546 (1) |
| P1–O2 | 1.546 (1) | 1.546 (1) |
| Average distances | 1.535 | 1.536 |
| P2–O3 | 1.521 (1) | 1.521 (1) |
| P2–O3vii | 1.521 (1) | 1.521 (1) |
| P2–O4vii | 1.568 (1) | 1.570 (8) |
| P2–O4 | 1.568 (1) | 1.570 (8) |
| Average distances | 1.544 | 1.545 |
| Angle (°) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| O1–P1–O1ix | 108.8 (1) | 109.61 (3) |
| O1–P1–O2ix | 110.77 (3) | 110.94 (9) |
| O1ix–P1–O2ix | 110.77 (3) | 109.61 (3) |
| O1–P1–O2 | 110.77 (3) | 109.61 (3) |
| O1ix–P1–O2 | 110.77 (3) | 109.61 (3) |
| O2ix–P1–O2 | 104.91 (9) | 107.40 (9) |
| O3–P2–O3vii | 112.62 (7) | 112.15 (4) |
| O3–P2–O4vii | 114.04 (5) | 113.05 (7) |
| O3vii–P2–O4vii | 108.10 (5) | 109.00 (5) |
| O3–P2–O4 | 108.10 (5) | 109.00 (5) |
| O3vii–P2–O4 | 114.04 (5) | 112.15 (4) |
| O4vii–P2–O4 | 99.34 (7) | 100.87 (6) |
| Fe1/Mg1–O distances (Å) and O–Fe1/Mg1–O angles (°) | ||
| Distance (Å) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| Fe1/Mg1–O4i | 1.9692 (8) | 1.9975 (8) |
| Fe1/Mg1–O4ii | 1.9692 (8) | 1.9975 (8) |
| Fe1/Mg1–O4iii | 1.9692 (8) | 1.9975 (8) |
| Fe1/Mg1–O4 | 1.9692 (8) | 1.9975 (8) |
| Fe1/Mg1–O1iv | 2.081 (1) | 2.069 (1) |
| Fe1/Mg1–O1v | 2.081 (1) | 2.069 (1) |
| Average distances | 2.007 | 2.021 |
| Angle (°) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| O1iv–Fe1/Mg1–O1v | 180.0 | 180.000 (6) |
| O4ii–Fe1/Mg1–O1v | 86.67 (3) | 88.18 (3) |
| O4i–Fe1/Mg1–O1v | 93.33 (3) | 91.82 (3) |
| O4iii–Fe1/Mg1–O1v | 93.33 (3) | 91.82 (3) |
| O4–Fe1/Mg1–O1v | 86.67 (3) | 88.18 (3) |
| O4–Fe1/Mg1–O1iv | 93.33 (3) | 91.82 (3) |
| O4i–Fe1/Mg1–O1iv | 86.67 (3) | 88.18 (3) |
| O4iii–Fe1/Mg1–O1iv | 86.67 (3) | 88.18 (3) |
| O4ii–Fe1/Mg1–O1iv | 93.33 (3) | 91.82 (3) |
| O4i–Fe1/Mg1–O4 | 85.90 (5) | 87.00 (5) |
| O4i–Fe1/Mg1–O4iii | 94.10 (5) | 93.00 (5) |
| O4ii–Fe1/Mg1–O4iii | 85.90 (5) | 87.00 (5) |
| O4ii–Fe1/Mg1–O4 | 94.10 (5) | 93.00 (5) |
| Mg2/Fe2–O distances (Å) and O–Mg2/Fe2–O angles (°) | ||
| Distance (Å) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| Mg2/Fe2–O2 | 2.0647 (9) | 2.0784 (8) |
| Mg2/Fe2–O2vi | 2.0647 (9) | 2.0784 (8) |
| Mg2/Fe2–O4 | 2.0719 (9) | 2.0849 (9) |
| Mg2/Fe2–O4vii | 2.0719 (9) | 2.0849 (9) |
| Mg2/Fe2–O3v | 2.1083 (9) | 2.1252 (8) |
| Mg2/Fe2–O3viii | 2.1083 (9) | 2.1252 (8) |
| Average distances | 2.082 | 2.096 |
| Angle (°) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| O3v–Mg2/Fe2–O3viii | 178.56 (6) | 179.55 (5) |
| O4–Mg2/Fe2–O3v | 88.74 (4) | 87.75 (3) |
| O4vii–Mg2/Fe2–O3v | 92.44 (4) | 92.62 (3) |
| O2–Mg2/Fe2–O3v | 84.60 (4) | 85.59 (4) |
| O2vi–Mg2/Fe2–O3v | 94.32 (4) | 94.07 (4) |
| O4–Mg2/Fe2–O3viii | 92.44 (4) | 92.62 (3) |
| O4vii–Mg2/Fe2–O3viii | 88.74 (4) | 87.75 (3) |
| O2–Mg2/Fe2–O3viii | 94.32 (4) | 94.07 (4) |
| O2vi–Mg2/Fe2–O3viii | 84.60 (4) | 85.59 (4) |
| O4–Mg2/Fe2–O4vii | 70.47 (5) | 70.95 (4) |
| O2–Mg2/Fe2–O4vii | 103.29 (3) | 103.28 (3) |
| O2–Mg2/Fe2–O2vi | 83.70 (5) | 83.27 (5) |
| O2vi–Mg2/Fe2–O4 | 103.29 (3) | 103.28 (3) |
| A1–O distances (Å) (A1 = Sr or Ba) | ||
| Distance (Å) | SrMg2Fe(PO4)3 | BaMg2Fe(PO4)3 |
| A1–O1xi | 2.642 (1) | 2.715 (1) |
| A1–O2 | 2.672 (1) | 2.715 (1) |
| A1–O2ix | 2.672 (1) | 2.745 (1) |
| A1–O3v | 2.6744 (9) | 2.745 (1) |
| A1–O3xii | 2.6744 (9) | 2.7658 (9) |
| A1–O3xiii | 2.6744 (9) | 2.7658 (9) |
| A1–O3xiv | 2.6744 (9) | 2.7658 (9) |
| A1–O1xi | 2.642 (1) | 2.7658 (9) |
| Average distances | 2.666 | 2.740 |
Symmetry codes: (i) x, − y + 1, − z + 1; (ii) − x + 1, y, z; (iii) − x + 1, − y + 1, − z + 1; (iv) x − 1/2, y, − z + 1/2; (v) − x + 3/2, − y + 1, z + 1/2; (vi) − x + 3/2, − y + 3/2, − z + 1/2; (vii) − x + 3/2, y, − z + 1/2; (viii) x, − y + 1, − z; (ix) − x + 2, − y + 3/2, z; (x) − x + 2, − y + 3/2, z + 1; (xi) x, y, z + 1; (xii) − x + 3/2, y + 1/2, z + 1/2; (xiii) x + 1/2, y + 1/2, z + 1/2; (xiv) x + 1/2, − y + 1, z + 1/2.
In the two compounds SrMg2Fe(PO4)3 and BaMg2Fe(PO4)3, the P5+ ions located at the Wyckoff positions (4e) and (8g) for P1 and P2 sites, respectively, have a tetrahedral environment (P1O4 and P2O4) with a mean distance ⟨P–O⟩ of 1.540 Å.
In both phosphates AMg2Fe(PO4)3 (A = Sr or Ba), the cations Fe3+ and Mg2+ located in the Fe1/Mg1 site, corresponding to the crystallographic Wyckoff position (4a), are surrounded by six O2− ions forming an octahedron (Fe1/Mg1)O6 with a mean distance ⟨Fe1/Mg1–O⟩ of 2.007 Å for SrMg2Fe(PO4)3 and 2.021 Å for BaMg2Fe(PO4)3.
For the Mg2/Fe2 site, the Mg2+ and Fe3+ cations are statistically distributed in the Wyckoff position (8g) and adopt an octahedral environment (Mg2/Fe2)O6 with a mean distance ⟨Mg2/Fe2–O⟩ of 2.082 Å for SrMg2Fe(PO4)3 and 2.096 Å for BaMg2Fe(PO4)3.
Finally, for each compound, the A cation (Sr2+ or Ba2+) is surrounded by eight O2− ions with a distance A–O ranging from 2.642(1) Å to 2.674 (1) Å for SrMg2Fe(PO4)3 and from 2.715 (1) Å to 2.766 (1) Å for BaMg2Fe(PO4)3.
In the crystal structure of AMg2Fe(PO4)3 phosphates (A = Sr or Ba), (Fe1/Mg1)O6 octahedra and P1O4 tetrahedra are connected via common vertices O1 alternately to form linear infinite chains (Fe1/Mg1)P1O9 along the b axis (Figure 1A), while the octahedra (Mg2/Fe2)O6 share the O2–O2 edge to form dimers (Mg2/Fe2)2O10. Each dimer (Mg2/Fe2)2O10 is linked to the two tetrahedral groups P2O4 by an O4–O4 edge to form the entity (Mg2/Fe2)2P22O14. Those later entities share the O3 vertices to form sheets (Mg2/Fe2)4P24O26 parallel to the (ac) plane (Figure 1B).
(A) Infinity chain of (Fe1/Mg1)P1O9 formed by sharing vertices of [Fe1Mg1O6] octahedra and P1O4 tetrahedra (B) Sheet showing the (Mg2/Fe2)4P24O26 unit along [010].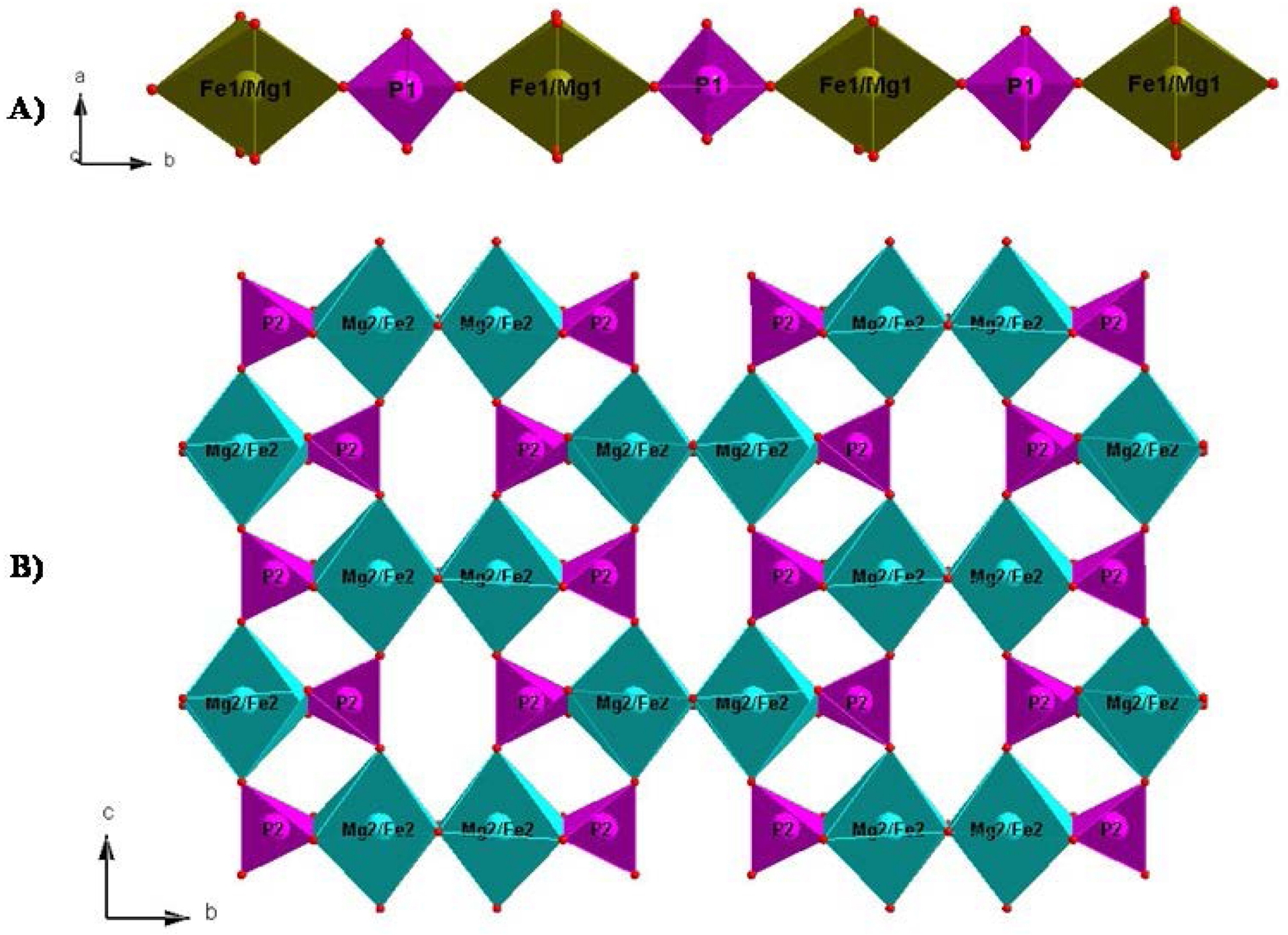
Vertices shared between sheets and chains lead to a three-dimensional framework delimiting two types of tunnels in which the Sr2+ or Ba2+ cations are located (Figure 2).
Projection view along the b direction of the structure showing the hexagonal tunnels that accommodate Sr or Ba in AMg2Fe(PO4)3 (A = Sr or Ba).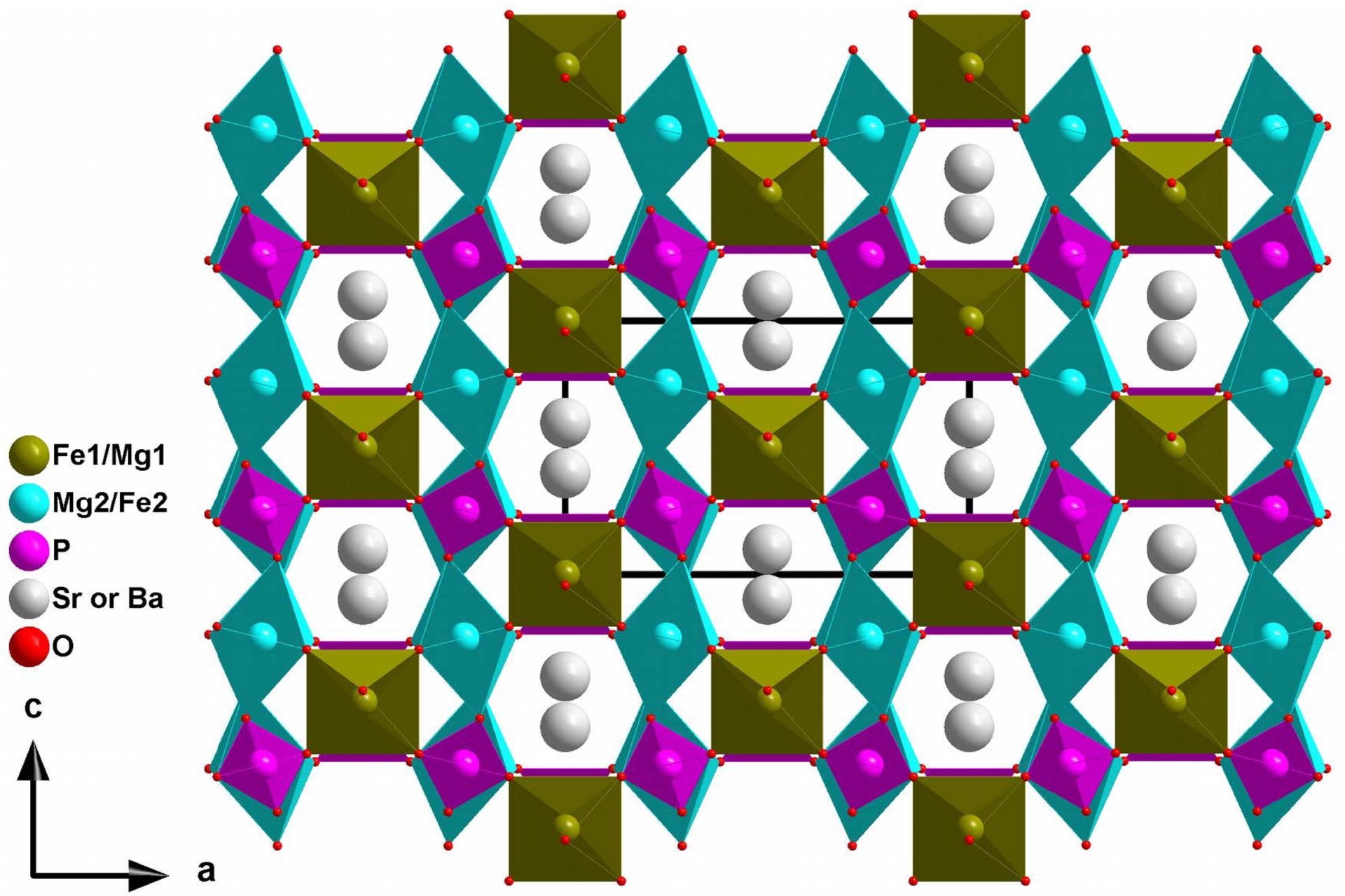
Exploration of the crystallographic database shows that the structures of both orthophosphates are similar to structures of the α-CrPO4 type mentioned in literature such as SrNi2Fe(PO4)3 [11], SrCo2Fe(PO4)3 [13], SrMn$^{\mathrm{II}}_{2}$MnIII(PO4)3 [4], BaNi2Fe(PO4)3 [10], and BaMn$^{\mathrm{II}}_{2}$MnIII(PO4)3 [5].
4. Conclusion
The single crystal X-ray diffraction study of these two new orthophosphates, SrMg2Fe(PO4)3 and BaMg2Fe(PO4)3 showed that these compounds crystallize in an orthorhombic system (Imma space group) with unit cell parameters a = 10.3998 (2) Å, b = 13.2000 (2) Å, c = 6.5364 (1) Å for the strontium compound and a = 10.4996 (2) Å, b = 13.2696 (1) Å, c = 6.6330 (2) Å for the barium composition. Their structures are isotype to α-CrPO4. The crystal structure of both compounds is composed of MgO6 octahedra, FeO6 octahedra, and PO4 tetrahedra. The sharing of vertices between those polyhedra leads to a three-dimensional network delimiting hexagonal channels that are occupied by Ba2+ or Sr2+ cations.
CRediT authorship contribution statement
Ahmed Ould Saleck: Investigation, Writing—Original draft, Writing—Review & editing.
Claudine Follet-Houttemane: Investigation, Writing—Original draft, Writing—Review & editing.
Cyrille Albert-Mercier: Investigation, Writing—Original draft, Writing—Review & editing.
Lahcen El Ammari: Formal analysis, Writing—Original draft, Writing—Review & editing.
Mohamed Saadi: Formal analysis, Writing—Original draft, Writing—Review & editing.
Abderrazzak Assani: Formal analysis, Writing—Original draft, Writing—Review & editing.
Declaration of interests
The authors do not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and have declared no affiliations other than their research organizations.
Data availability
All the data generated or analyzed during this study are included in this published article.
The data can be obtained free of charge from the Cambridge Crystallographic Data Centre at http://www.ccdc.cam.ac.uk/structures with deposition number CCDC 2454547 for BaMg2Fe(PO4)3 and CCDC 2454549 for SrMg2Fe(PO4)3.






 CC-BY 4.0
CC-BY 4.0