1. Forest microbiota
Forests cover more than 30% of the Earth’s land surface and mainly consist of natural forest massifs, including primary and secondary forests. Boreal, temperate, and tropical woodlands (Figure 1) are home to over 3,100 billion trees (Crowther et al., 2015) and 80% of the plant, animal, and microbial biodiversity (Muys et al., 2022). They sequester substantial stocks of carbon in plantations and old-growth forests (Luyssarert et al., 2008) and lie at the heart of intricate networks of interactions between plants, animals, and microbes. Understanding how these forest ecosystems function entails measuring the flows of matter and energy that circulate within biogeochemical cycles, but also characterising the role of all the microorganisms — the microbiota — that influence soil fertility and carbon storage, as well as the vigour and health of trees (Baldrian et al., 2023). The composition and sustainability of forests are closely linked to the interdependence of plants and microorganisms (Baldrian, 2017; Peay et al., 2016). The forest microbiota — bacteria, archaea, fungi, and viruses — underpins this interdependence (Uroz et al., 2016); collectively, these organisms drive the decomposition of organic matter, nutrient cycling, soil formation, and plant growth, thereby regulating the biogeochemical cycles of carbon, nitrogen, and phosphorus that govern forest ecosystem dynamics. Their capacity to adapt to climate- and human-induced changes is a primary determinant of long-term soil fertility and forest sustainability (Peay et al., 2016; Uroz et al., 2016; Baldrian et al., 2023).

The boreal taiga of Alaska (A), beech-fir forests of Vosges (B), and primary mountain forests of north-western China’s Yunnan (C) are particularly rich in ectomycorrhizal fungal species.
It is now well established that trees host a complex community of microorganisms in their various organs, the microbiota, which is diverse and composed of thousands of species of viruses, bacteria, and fungi that influence the growth, development, and health of their host plants (Uroz et al., 2016; Baldrian, 2017; Li et al., 2023; Enea et al., 2025). This recognition has integrated forest microbiology and microbial ecology into a multidisciplinary research approach that aims to describe the biodiversity and functioning of forest ecosystems (F. Martin, 2021). However, to assess the ecological and economic services provided by the microbiota, it is necessary to develop molecular atlases that list and map the communities and populations of beneficial, commensal, and pathogenic microorganisms, monitor their dynamics in relation to changes in the forest, and assess the impacts of silvicultural management and climate change (Uroz et al., 2016). Identifying the species whose activity is essential for the proper functioning of the microbial community, its associated plants, and biogeochemical cycles is a central objective of current research (F. Martin and van der Heijden, 2024). Monitoring the presence and abundance of these keystone species would ensure the maintenance of optimal microbial biodiversity, thereby supporting tree growth, health, and long-term soil fertility. These microbiological indicators can then be incorporated into the suite of criteria used for certifying sustainably managed forests or establishing strict biological reserves. Sustainably managed forests are expected to constitute rich, stable reserves of terrestrial biodiversity, producing renewable resources, including wood, regulating water circulation and filtration, and functioning as critical carbon sinks.
2. Global changes
In recent decades, forests have been subjected to multiple pressures, including landscape fragmentation linked to human activities, increased atmospheric CO₂, global warming, recurrent droughts, megafires, and anthropogenic nitrogen deposition (Keenan, 2015; Seidl et al., 2017; Vacek et al., 2023). These factors directly alter the diversity and dynamics of plant communities and their associated microbiota, as well as critical processes such as nutrient cycling, carbon storage, and plant–microorganism interactions (Baldrian et al., 2023). For example, increased soil temperatures accelerate microbial biochemical processes, thereby altering the decomposition rate of organic matter and, consequently, the availability and acquisition of nutrients. This acceleration is particularly pronounced in boreal forests, where considerable stocks of carbon immobilised for hundreds of years are remobilised by increasingly active microbial communities (Hopkins et al., 2012). However, temperatures that exceed species-specific thermal tolerance thresholds have driven the local disappearance of numerous fungal taxa in temperate forests, notably ectomycorrhizal species with narrow climatic niches. Changes in precipitation patterns further govern soil moisture, which is a primary determinant of microbial survival and activity. Prolonged drought periods, as experienced across Europe from 2018 to 2020, suppress microbial activity, whereas episodes of excessive rainfall accelerate nutrient leaching from the soil profile. Elevated atmospheric CO₂ concentrations strongly stimulate plant growth, increasing organic matter inputs to the soil. Elevated CO₂ levels also alter the biochemical quality of organic matter, with cascading effects on microbial community dynamics and decomposition processes (Peay et al., 2016; Baldrian et al., 2023).
Describing, characterising, and modelling the multiple plant–microbe interactions that constitute the complex ecological networks of forest massifs is now an urgent research priority, both for developing silvicultural management scenarios that mitigate the foreseeable impacts of environmental change and for assessing anthropogenic threats to forest biodiversity, particularly in old-growth forests (F. Martin, 2021). Understanding these interdependencies is essential for anticipating future dieback and developing scenarios to mitigate the growing impact of human activities such as landscape fragmentation and deforestation (Uroz et al., 2016; Baldrian et al., 2023). Identifying the factors that govern the associations between plants and their microbiota should facilitate the use of beneficial microorganisms to stimulate tree growth, enhance their resilience to climatic and anthropogenic constraints, and limit the negative impacts of pathogens (Li et al., 2023; Brüssow et al., 2024). Elucidating the mechanisms that regulate these microbial responses and assessing their long-term implications for soil fertility, forest ecosystem productivity, and biodiversity conservation represent the central challenges for the next generation of forest microbiome research.
3. Different fungal communities in forests
Three broad functional guilds — positions along a nutritional continuum defined primarily by their mode of carbon acquisition — can be recognised in temperate, boreal, and tropical forests; these guilds are further differentiated by their degree of host dependency and trophic specialisation (Figure 2; Baldrian, 2017; Lebreton et al., 2021; F. Martin, 2021):
– Decomposers or saprotrophs: these fungi efficiently degrade polysaccharides and proteins from plant, microbial, and animal necromass through agents of white and brown rot as well as litter decomposers; some species, such as Heterobasidion annosum, are also facultative pathogens capable of infecting already-weakened trees.
– Mutualistic symbionts: these fungi obtain simple sugars, primarily glucose, from their host plant, reciprocally stimulating its growth and mineral nutrition, while their subterranean mycelial networks explore soil and litter to extract mineral elements, which are transferred to the host in exchange for imported carbon.
– Parasites or pathogens: these fungi infect plants and animals, exploiting the sugars and soluble amino acids accumulated in colonised tissues, which can ultimately weaken or kill their host.

Some species of woodland fungi: ectomycorrhizal fungi ((A) Brittlegill (Russula sp.); (B) Rooting bitter bolete (Caloboletus radicans)), and saprotrophic fungi, including an agent of white rot ((C) Hoof fungus (Fomes fomentarius)), a brown rot fungus ((D) Red-belted polypore (Fomitopsis pinicola)), a litter decomposer ((E) Clouded agaric (Clitocybe nebularis)) and a stump-forming fungus ((F) Crown-tipped coral (Artomyces pyxidatus)).
Decomposer fungi play a crucial role in ensuring the long-term productivity and sustainability of ecosystems, such as forests. Their mycelium contains an enzymatic repertoire (ligninases, cellulases, and pectinases) that degrades polysaccharides and lignin in wood and plant detritus, respectively. Notably, xylophagous and organic matter-degrading fungi release mineral elements and carbon from necromass, recycling them into biomass and sustaining the fertility that underpins primary productivity.
Unlike saprotrophs, symbiotic and pathogenic fungi interact directly with living host tissues, influencing tree metabolism, physiology, and growth, either positively or negatively.
Within the pathogenic guild, host fate diverges sharply along the biotrophic–necrotrophic axis of the host. Biotrophic fungi, such as poplar leaf rust (Melampsora spp.), sustain chronic parasitism without killing their hosts, whereas necrotrophic pathogens, such as honey fungus (Armillaria mellea), actively destroy host tissue, ultimately killing the tree.
Symbiotic mycorrhizal fungi colonise the root surface and intercellular space of the root epidermis and cortical parenchyma. In some mycorrhizal associations, such as arbuscular, orchid, and ericoid mycorrhizae, hyphae form intracellular structures. From the colonised root, hyphae often develop a dense network of filaments in the rhizosphere and soil, from which they extract mineral elements, particularly nitrates and inorganic phosphates. Some symbiotic fungi also degrade organic soil compounds (phytates and proteins), releasing bioavailable phosphates and soluble amino acids that are subsequently transferred to the host in exchange for carbon.
Mycorrhizal fungi also interconnect multiple plants through underground hyphal networks (Rog et al., 2020). Several studies suggest that fungal networks facilitate resource exchange among plants within a community (Figure 3) (Simard et al., 1997; Selosse, Richard et al., 2006; Klein et al., 2016), but the ecological importance of these common mycorrhizal networks (CMNs) is currently disputed (Karst et al., 2023; Robinson et al., 2024). These criticisms do not contest the existence of underground mycorrhizal networks per se, but rather the evidence for their functional roles in inter-plant nutrient exchange. Given this scientific controversy, several field experiments have been conducted in mature forest stands to quantify the nutrient flows and signals circulating within CMNs (van der Heijden et al., 2015; Rillig et al., 2025). Carbon flows transiting through ectomycorrhizal fungal networks are sometimes 'plundered' by fungi associated with mycoheterotrophic and mixotrophic plants, such as orchids, which then act as parasites of the mycorrhizal network (Selosse, Richard et al., 2006, Figure 3).
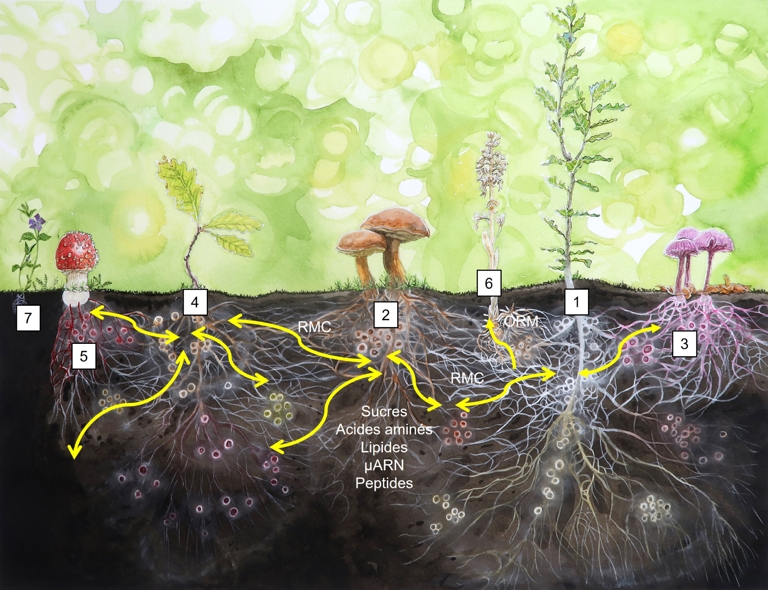
Hypothetical representation of the soil mycorrhizal community in a forest dominated by oak species associated with different types of mycorrhizal fungi that form intermingled mycelial networks (yellow arrows) through which amino acids, carbohydrates, lipids, microRNAs, and signalling molecules may be exchanged between host plants. A holm oak (Quercus ilex) (1) forms ectomycorrhizal roots with the hyphae of the red-cracking bolete (Xerocomellus chrysentheron) (2) and the amethyst deceiver (Laccaria amethystina) (3), whereas a sessile oak (Quercus petraea) (4) forms ectomycorrhizal associations and hyphal networks with X. chrysentheron (2) and the fly agaric (Amanita muscaria) (5). Ectomycorrhizae are represented by coloured dots on the root systems. The hyphal networks of X. chrysentheron link Q. ilex and Q. petraea through a common mycorrhizal network (CMN). The non-photosynthetic bird's nest orchid (Neottia nidus-avis) and its orchid mycorrhizal network (ORM) (6) parasitise the mycorrhizal network of the holm oak. The lesser periwinkle (Vinca minor) is colonised by arbuscular mycorrhizal fungi, forming an independent mycorrhizal network (7). Different colours represent different fungal networks, with mycorrhizal roots represented as dots of the corresponding colour. Other network configurations are possible; for example, boreal forests with ectomycorrhizal trees often support an undergrowth of ericaceous plants (e.g. rhododendrons, bilberries, and cranberries) that form ericoid mycorrhizal associations. Illustration by Florian Gadenne. Adapted with permission from F. Martin and van der Heijden (2024).
Mycorrhizal symbiosis occupies a central position in the biology, ecology, and management of forests, governing plant health, productivity, and diversity while underpinning the carbon, nitrogen, and phosphorus cycles of terrestrial ecosystems. Worldwide, mycorrhizal plants store over 350 gigatonnes of carbon in their above-ground biomass alone, a figure that underscores the role of these fungi as keystone ecosystem engineers whose services extend across carbon sequestration, nutrient cycling, and soil structure (Peay et al., 2016; Baldrian et al., 2023). Harnessing this knowledge in programs for the conservation of old-growth forests, reforestation, assisted migration, and the restoration of disturbed or polluted sites now requires a deeper characterisation of tree microbiota and their functional roles.
4. Biodiversity of mycorrhizal symbioses
Mycorrhizal associations, which first arose approximately 450 Ma, coinciding with the colonisation of land by plants, are ubiquitous in the plant kingdom. An estimated 72% of plant species establish symbiosis with arbuscular mycorrhizal fungi of the class Glomeromycetes, approximately 2% of plants — predominantly trees — form ectomycorrhizal associations, a further 10% — principally orchids — form orchid mycorrhizae, and 1.5% — primarily Ericaceae — form ericoid mycorrhizae (van der Heijden et al., 2015; Brundrett and Tedersoo, 2018; F. Martin and van der Heijden, 2024).
Ericaceae, such as rhododendrons, cranberries, and azaleas, and their symbiotic fungi drive carbon sequestration by generating recalcitrant plant and microbial litter that is highly resistant to decomposition, thereby promoting the accumulation of organic carbon in boreal and montane soils (Ward et al., 2022). Consequently, ecosystems dominated by Ericaceae and their ericoid fungi are estimated to account for almost 20% of the planet's terrestrial carbon storage (Perotto et al., 2018; Soudzilovskaia et al., 2019).
Some species, such as poplars and eucalyptus, can associate with both arbuscular and ectomycorrhizal fungi, although these two symbiont types do not colonise roots simultaneously; arbuscular mycorrhizal fungi (Glomeromycetes) typically predominate in the juvenile stage, whereas ectomycorrhizal fungi come to dominate as trees mature. Mycorrhizal plants dominate almost all ecosystems (Brundrett and Tedersoo, 2018; Steidinger et al., 2019), except in intensively cultivated arable land and extremely phosphorus-poor soils, where non-mycorrhizal families, such as Proteaceae, prevail. Approximately 8% of plant species, notably aquatic, carnivorous, and parasitic lineages, are non-mycorrhizal.
5. Main factors that control diversity in mycorrhizal communities
A central objective in the ecology of mycorrhizal fungi is to identify the genetic and environmental factors that govern the spatial and temporal structures of their communities and populations. This involves inventorying the species diversity of mycorrhizal fungi across terrestrial ecosystems from the tropics to the poles, mapping their spatial and temporal distributions, and characterising the drivers of genetic structuring within these communities (Tedersoo et al. (2014), Větrovský, Kohout et al. (2019), Větrovský, Morais et al. (2020), Větrovský, Kolaříková et al. (2023), Once species richness and spatial distribution have been determined on a seasonal and/or annual basis, microbiologists seek to characterise the functions expressed by these fungi, such as those involved in the decomposition of organic matter or the uptake of mineral nitrogen (Zeng et al., 2023; Auer et al., 2024). The composition of fungal communities is closely coupled to their abiotic and biotic environments and shifts with the age of the ecosystem. Numerous biotic (e.g. vegetation type) and abiotic (e.g. pH and precipitation) factors shape this diversity at spatial scales ranging from continental and regional to local and even individual root levels. These drivers frequently act in concert or in sequence, encompassing plant community composition and structure, reproductive strategies, mycelial and spore dispersal, resource exploitation, and resistance and resilience to biotic and abiotic disturbances.
The distribution of mycorrhizal types across terrestrial biomes is not random but is closely linked to climate (Read, 1991; Read and Perez-Moreno, 2003); arctic and alpine plant communities are dominated by ericoid mycorrhizal plants, boreal regions by ectomycorrhizal conifers, and temperate forests by a mixture of woody plants associated with both ectomycorrhizal and arbuscular mycorrhizal fungi. Subtropical and tropical forests mainly comprise arbuscular mycorrhizal plants, although some ecosystems dominated by Dipterocarpaceae are notably rich in ectomycorrhizal fungi. Meta-analyses of fungal community distribution on a continental scale underscore these patterns (Steidinger et al., 2019; Větrovský, Kohout et al., 2019; Větrovský, Morais et al., 2020), revealing close associations between latitude, altitude, climate, soil properties, and mycorrhizal type. One example of this global approach is the GlobalFungi database (https://globalfungi.com), which compiles and maps the diversity of soil fungi from over 85,000 samples drawn from 846 molecular inventories, totalling more than 3.5 billion fungal ribosomal DNA sequences (Větrovský, Kohout et al., 2019; Větrovský, Morais et al., 2020). Climate emerged as the dominant driver of soil fungal distribution, with climatic factors accounting for 65% of the variance, soil-related variables for 24%, and vegetation for 11%. Within the climatic component, temperature (39%) and precipitation (27%) carried the greatest explanatory weight, whereas bulk density (14%) outweighed soil pH (9.5%) among the edaphic predictors. Notably, because of their close coupling with plant hosts, symbiotic mycorrhizal fungi are considerably more sensitive to rapid climate change than saprotrophic or pathogenic guilds.
6. Impact of global warming
Global warming is profoundly reshaping plant and microbial communities in forests (Seidl et al., 2017; Jansson and Hofmockel, 2020; Baldrian et al., 2023). Increasing temperatures progressively shift the geographical distribution of plants, and this range displacement directly affecting their associated fungi. For example, beech (Fagus sylvatica), a dominant tree in European montane forests, is gradually migrating to higher altitudes (at a rate of 1 m per year since 1925), thereby shifting its distribution area and consequently that of its fungal partners. Such compositional shifts risk compromising key ecosystem services, such as organic matter recycling, nutrient cycling, and long-term soil fertility, while threatening the persistence of range-restricted fungal species. A study conducted in the Alps demonstrated that mycorrhizal fungi migrate to higher altitudes at a rate of 0.6 to 2.2 m per year, depending on their plant hosts (Diez et al., 2020); such altitudinal migration disrupts the ecological equilibrium of local communities, particularly in high-altitude ecosystems where the growing season is already severely compressed. In the United States, studies on pine forests have demonstrated that rising temperatures favour decomposing fungi at the expense of mycorrhizal fungi, thereby accelerating litter decomposition rates and progressively depleting soil carbon stocks (Treseder et al., 2016).
In temperate forests, prolonged soil warming experiments, such as those conducted at the Harvard Forest Experimental Station (Worcester County, Massachusetts, USA), have highlighted significant shifts in microbial community structure, including a marked decrease in ectomycorrhizal fungi, changes that directly compromise carbon storage in soil organic matter and disrupt nutrient cycling (Pec et al., 2021; Baldrian et al., 2023). For instance, rising temperatures accelerate the fungal and bacterial decomposition of polysaccharides accumulated in soil organic matter, thereby increasing CO₂ and CH₄ emissions. Moreover, long-term studies (Melillo et al., 2017) underscore that prolonged soil warming triggers successive phases of rapid carbon loss, microbial community reorganisation, and depletion of easily degradable organic matter stocks, dynamics that metatranscriptomic approaches are now beginning to resolve at the community level.
7. Effect of drought on microbial communities
Edaphic drought is a primary determinant of soil microbiota composition. It reduces microbial biomass, shifts the fungus-to-bacteria ratio, and disrupts biogeochemical processes, such as nitrification and organic matter decomposition (Meisner et al., 2018). Several studies have established that fungal communities and their networks are more resilient than bacterial communities during droughts (de Vries et al., 2018). This resilience is rooted in the physiological and morphological traits that confer resistance to desiccation. Many species can strengthen their cell walls or upregulate osmolyte synthesis (e.g. glycerol) to regulate cell water potential (Jansson and Hofmockel, 2020; Wang et al., 2025). However, these adaptations are energetically costly and may be insufficient during prolonged droughts. Drought tolerance varies markedly among ectomycorrhizal species; during desiccation events, sensitive taxa, such as Hygrophorus sp., decline sharply, whereas tolerant species, such as Cenococcum geophilum, dominate (Baldrian et al., 2023; Wang et al., 2025).
In boreal forests, recurrent droughts reduce ectomycorrhizal fungal diversity and favour saprotrophic communities capable of rapidly decomposing organic matter (Baldrian et al., 2023). A study of California oak forests revealed that prolonged drought reduced ectomycorrhizal fungal populations, compromising mineral element uptake by the host plants (Allen et al., 2010). These effects are often exacerbated by soil compaction and reduced organic matter content, which impair the resilience of mycelial networks. Nonetheless, ongoing field experiments have begun to identify the fungal taxa that are most critical to post-drought ecosystem recovery.
8. Effect of increased atmospheric nitrogen deposition
Atmospheric nitrogen deposition, driven primarily by intensive agriculture, livestock operations, and industrial emissions across Western Europe, is altering the structure of forest communities. In European forests, plant species are progressively shifting their ranges northward and toward higher elevations in response to warming, and a concurrent westward displacement is strongly correlated with excess soil nitrogen transported by westerly atmospheric currents toward Central Europe (Sanczuk et al., 2024). These range shifts in plant communities modify the associated microbiota, favouring nitrophilic bacteria and reducing the abundance of certain ectomycorrhizal fungi, such as the golden chanterelle (Cantharellus cibarius). Mycorrhizal fungi adapted to nutrient-poor soils lose their competitive edge under nitrogen-enriched conditions. Studies in European deciduous forests have demonstrated that excess nitrogen favours saprotrophic fungi at the expense of symbiotic ectomycorrhizal fungi (Lilleskov, Hobbie and Fahey, 2002; Lilleskov, Hobbie and Horton, 2011), a competitive shift that destabilises soils and amplifies CO₂ emissions through accelerated organic matter decomposition. Forest ecosystems in Europe and North America have been particularly affected, with lasting consequences for mycorrhizal fungal diversity and the long-term carbon sequestration capacity of soils (Lilleskov, Hobbie and Fahey, 2002; Erik et al., 2024).
9. Effect of forest fires
Forest fires, exacerbated by warming and drought, profoundly disrupt the soil microbiota (Koster et al., 2021; Nelson et al., 2022). In Canadian boreal forests, recurrent fires reduce microbial biomass, alter fungal community composition, and diminish long-term carbon storage (Holden, Gutierrez et al., 2013; Holden, Rogers et al., 2016; Koster et al., 2021). In Europe, studies on Mediterranean maquis have demonstrated that recurrent fires favour opportunistic fungal species at the expense of symbiotic species, thereby disrupting ecosystem dynamics over the long term (Bastias et al., 2006). Fires act as selective agents, reshaping the phylogenetic composition of ectomycorrhizal communities associated with pines during natural regeneration and favouring lineages potentially adapted to fire disturbance (Rincón et al., 2014). These disturbances increase soil vulnerability to erosion and reduce its capacity to support effective plant recolonisation.
10. Adaptations and ecological responses
Remarkably, despite the scale of climate-driven disturbances, certain microbial communities display an adaptive capacity that partly buffers ecosystem disruption. Fungi adjust their metabolism, gene expression, and spatial distribution in response to water stress and nutrient fluctuation. Three response modes are well documented:
- Genetic plasticity and transcriptomic response: Studies on ectomycorrhizal fungi have revealed the upregulation of drought tolerance genes during water deficit conditions. Notably, the ascomycete Cenococcum geophilum upregulates soluble sugar accumulation in response to osmotic shock, thereby maintaining turgor and hyphal integrity (Wang et al., 2025).
- Altitudinal migration: in the Alps, ectomycorrhizal fungi migrate to higher elevations with their hosts; several webcap (Cortinarius spp.) and brittlegill (Russula spp.) taxa, typically present at mid-elevations, gradually colonise higher subalpine zones where cooler, moister conditions more closely match their climatic optima (Diez et al., 2020).
- Colonisation of disturbed niches: In areas affected by forest fires, opportunistic fungi such as Pezizales develop rapidly in carbon-rich post-fire soils, stabilising substrate conditions and thereby facilitating the long-term recolonisation of burned sites by forest species (Glassman et al., 2016).
11. Conservation initiatives and outlook
Preserving the ecological integrity of minimally disturbed ecosystems, including forests, and expanding areas dedicated to integral biological reserves and protected zones (Natura 2000, Zones of Ecological and Floristic Interest — ZNIEFF) are prerequisites for long-term fungal conservation. Increasing the land area devoted to old-growth forests or forests under non-intervention management will further promote microbial richness in forest stands. In production forests, sustainable management must be encouraged through regulations to preserve biodiversity, soil quality and fertility, and carbon storage. Forest management plans should endeavour to limit clear-cutting, plot fragmentation, and the use of heavy machinery that exerts lasting impacts on plant and microbial diversity (Hartmann et al., 2014; Vennin et al., 2025).
In addition to rising atmospheric CO₂, current climate change is driven by recurrent soil droughts, anomalous spring heat waves, and intensifying storm regimes, which are accelerating forest decline across Europe. Between 2021 and 2023, the proportion of damaged forest trees exceeded 15% in northeastern France, reducing biological growth and diminishing forest carbon uptake (Inventaire forestier national, 2024). Innovative approaches, including microbiota transplantation (Allsup et al., 2023) and controlled mycorrhization (Le Tacon et al., 1997) represent concrete tools for mitigating forest decline and restoring disturbed areas. These microbial engineering techniques operate by manipulating community composition to improve essential ecosystem functions, including soil fertility, seedling establishment, and resistance to abiotic stress. The immediate challenge is to integrate microbial processes into forest adaptation scenarios and to deploy microbiota-based solutions — improving soil fertility, establishing young seedlings, and enhancing soil carbon capture — in restoration programs.
Experimental inoculation with mycorrhizal fungi, such as the symbiont Rhizophagus irregularis, has demonstrated its capacity to enhance phosphorus and nitrogen uptake in depleted soils (Onyeaka et al., 2024; F. Martin and van der Heijden, 2024). Controlled mycorrhization, the inoculation of soils with mycorrhizal fungi selected for their symbiotic efficacy, has been deployed on a large scale to support the establishment of Douglas fir seedlings colonised by Laccaria bicolor (Le Tacon et al., 1997). Selosse et al. (1999) demonstrated that introduced ectomycorrhizal mycelium persisted for many years on adult plantation trees. The symbiotic association thrives in undisturbed forest conditions, where tillage and high fertility are absent, and controlled mycorrhization can be used in forest-management scenarios. In contrast, intensive tillage destroys mycelial networks in agricultural soils, and elevated fertility disrupts symbiosis.
Controlled mycorrhization is widely practiced in regions affected by desertification and soil depletion. For example, in maritime pine (Pinus pinaster) plantations in Spain, inoculation with the ectomycorrhizal symbiont Pisolithus tinctorius markedly promoted tree growth under drought conditions (Requena et al., 2001). Controlled mycorrhization has also been applied to inoculate oak and hazelnut trees with truffle species, such as the Périgord truffle (Tuber melanosporum), Burgundy truffle (T. aestivum uncinatum) (Figure 4) and white Piedmont truffle (T. magnatum) (Murat et al., 2017). Truffle cultivation is particularly effective when orchards are established on former agricultural land or vineyards, where the local soil fungal inoculum is low. In future controlled mycorrhization programs, the introduction of exotic microbial strains must be avoided to safeguard local microbial diversity.

Experimental truffle farm in Boncourt-sur-Meuse, France. Controlled inoculation of hazelnut seedlings with Burgundy truffle mycelium enables the production of fruiting bodies a few years after planting. Most truffle farms are now equipped with irrigation systems to mitigate summer soil droughts.
Allsup et al. (2023) demonstrated that in Illinois (USA), tree seedlings inoculated with complex microbial communities from drier, warmer, or colder sites exhibited significantly higher survival rates under drought, heat, or cold stress, respectively. Drought tolerance was correlated with increased arbuscular mycorrhizal fungal diversity, whereas cold tolerance was correlated with lower fungal richness, likely reflecting a reduced load of non-adapted taxa in the cold.
Molecular inventory and mapping of microbial diversity, transplantation of drought-adapted microbiota, and controlled mycorrhization of forest seedlings are currently being developed as part of forest restoration projects (e.g. the oak forest on the Chantilly estate in the Oise Department, France). These approaches, together with silvicultural management promoting species diversification through natural regeneration and assisted migration, must be integrated into comprehensive sustainable land management programs; their implementation, for example, within the network of regional nature parks, will require close collaboration between scientists, managers, and policymakers to optimise deployment and maximise impact at the landscape scale. These methods of microbial ecological engineering offer a coherent framework for mitigating the effects of ongoing environmental change; however, they must be accompanied by a robust policy framework for ecological transition aimed at limiting the impact of human activities on biodiversity and, more broadly, natural ecosystems.
12. Conclusions and further research
Forest fungi — decomposers and mycorrhizal symbionts alike — emerge from this review as indispensable regulators of biogeochemical cycles, tree health, and ecosystem resilience; their biology and ecology now constitute a central frontier of forest science.
Les techniques de séquençage à haut débit permettent désormais de recenser la diversité et la richesse des communautés de champignons sylvicoles, de cartographier leur distribution spatiale et de suivre leur dynamique dans divers types de forêts (Auer et al., 2024). These inventories enable the assessment of a major component of forest biodiversity and provide an empirical foundation for monitoring soil quality, evaluating silvicultural management practices and tracking the health of protected forests.
Comparative genomics of hundreds of saprotrophic and mycorrhizal fungi have significantly advanced our understanding of the evolutionary transitions that gave rise to mutualistic symbionts from saprotrophic ancestors (Lebreton et al., 2021). These genomic resources facilitate the study of the biological, genetic, and evolutionary processes governing soil fungal communities and, through metatranscriptomics, the characterisation of functions expressed by these communities across forest types (Auer et al., 2024). These approaches of environmental genomics are clarifying the role of fungi in plant growth and evolution, biogeochemical cycles, soil microbiology, and the trajectory of forests under environmental change — insights that are beginning to inform concrete management and restoration strategies.
Mycorrhization has a demonstrably beneficial impact on the growth of forest seedlings in reforestation and plantation programs. Symbiotic fungi stimulate mineral nutrition in young plants, provide bioprotection against soil pathogens, and improve soil structure, making the management of mycorrhizal symbiosis a major asset in ecological approaches to plantation management and in forest restoration. A central frontier for the coming years is the characterisation of the mechanisms governing microbiota composition and dynamics, and in particular, the functional role of mycorrhizal fungi within those communities across the full range of forest ecosystems and climate trajectories. Within the coming decade, it should become practicable to establish trees pre-fortified with selected microbiota, equipping them to withstand the increasingly adverse abiotic conditions imposed by ongoing climate change — a prospect that positions forest microbiome science at the interface of fundamental ecology and applied silviculture.
Glossary
Anthropogenic: Relating to human activities or their impact on the environment.
Arbuscular mycorrhizae: mutualistic symbiosis between glomeromycete fungi and plant roots, characterised by the formation of arbuscules (intracellular exchange structures) and vesicles (storage organs) in the root cortex.
Assisted migration: A conservation strategy involving the intentional movement of species, populations, or genotypes to more favourable habitats, usually in response to climate change.
Biogeochemical cycles: natural processes involving the circulation and transformation of chemical elements (carbon, nitrogen, phosphorus, etc.) between living (biosphere) and nonliving (lithosphere, hydrosphere, atmosphere) components of ecosystems.
Ectomycorrhizae: mutualistic symbiotic associations between a fungus (typically an ascomycete or basidiomycete) and the fine roots of a plant, characterised by the formation of a fungal mantle around the root tip and a Hartig net — an intercellular hyphal network penetrating between epidermal and cortical cells — but without intracellular penetration.
Ericoid mycorrhizae: a type of mycorrhiza associated with plants of the Ericaceae family, characterised by intracellular colonisation of the roots by fungal hyphae forming hyphal pelotons (tightly coiled intracellular hyphae).
Ligninase: An enzyme that degrades lignin; principal examples include class II peroxidases.
Managed forests: forest ecosystems subject to planned human interventions (silviculture) aimed at achieving specific production, conservation, or protection objectives.
Microbiota: all microorganisms (bacteria, archaea, fungi, and viruses) inhabiting a specific environment, host, substrate, or ecosystem.
Mixotrophic plants: plants that supplement autotrophic carbon gain (photosynthesis) with heterotrophic carbon obtained via their mycorrhizal fungal partners, such as certain partially achlorophyllous orchids and some members of Ericaceae and Pyrolaceae.
Mycoheterotrophic plants: Achlorophyllous plants that depend entirely on their mycorrhizal fungal partners for their carbon supply in a relationship that is functionally parasitic on fungi that are associated with autotrophic plants.
Orchid mycorrhizae: specific and obligatory mycorrhizal associations in orchids, where the fungus provides essential carbon compounds and mineral nutrients for seed germination and nutrition of adult plants (notably achlorophyllous species).
Osmolyte: a small soluble organic molecule (such as proline, glycine betaine, or polyol sugars) that accumulates in cells to maintain water balance and protect against osmotic stress.
Declaration of interests
The author does not work for, advise, own shares in, or receive funds from any organisation that could benefit from this article and has declared no affiliations other than their research organisation.




 CC-BY 4.0
CC-BY 4.0