1 Introduction
Fenugreek (Trigonella foenum-graecum L.) is a known medicinal plant with a long historical usage as a prevalent and natural medicine in Asia, Africa, Egypt, Middle East, and Europe. It is an annual plant belonging to Leguminosae or Fabaceae family. Egyptians use this herb to embalm mummies and as incense. Moreover, the Romans and Greeks used it for cattle fodder. The words foenum-graecum present the Latin name of fenugreek which means Greek-hay [1]. As stated by Morton [2], the height of this plant is up to 60 cm with leaves of 2–2.5 cm long. The brownish-yellow seeds grow inside long and narrow seedpods of 14 cm. Each pod contains 10–20 seeds with seed dimension of 3 × 4 mm. Owing to its high pharmaceutical and nutritional properties, different parts of this plant have attracted investigation by several researchers. The seeds and leaves of this plant have been reported to possess antidiabetic, antimicrobial, lactation aid, antiinflammation, anticancer, antifungal, and antioxidant activities [3,4]. In recent years, plants with high nutritional values have received more attention as a dietary supplement to prevent chronic immune disorders. Researchers have claimed that chronic ailments are treatable or could be averted by using the sources of biocompounds extracted from plants and fruits as plants contain different secondary metabolites and pharmacological components such as alkaloids, flavonoids, phenols, saponins, steroids, triterpenoids, and tannins [5]. Secondary metabolites (polyphenols and flavonoids) have been used as natural antioxidants to control oxidative stress or cell damage [6]. However, saponins are found in either steroid or triterpenoid glycosides. In terms of health benefit, both saponins and phenolic compounds existed in plants have demonstrated potential medicinal values for the treatment of numerous diseases such as cancers, heart-related illnesses, tumor, infections, and diabetes [7]. Nevertheless, the recoveries of bioactive phytochemical compounds from plants are potentially affected by the conditions of extraction methods and different solvent formulations.
Furthermore, several researchers have reported the existence of flavonoids, alkaloids, steroidal saponins, tannins, and phenolic compounds in fenugreek seeds. However, the determinations of saponins, diosgenin, steroidal sapogenins, phenolic compounds, and antioxidant compounds in these seeds are mostly carried out using conventional extraction methods such as Soxhlet and maceration extraction [8,9]. Besides, different solvents had been applied for the recovery of phytochemicals and bioactive compounds from fenugreek seeds.
To the best of our knowledge, no study has reported the optimization of microwave-assisted extraction (MAE) parameters including extraction time, ethanol concentration, microwave power, and feed-to-solvent ratio in achieving optimum recoveries of total phenolic content (TPC), total saponin content (TSC), 2,2-diphenyl-1-picrylhydrazyl (DPPH), and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) antioxidant capacities from fenugreek seeds. Therefore, this study aims to apply face-centered central composite design (FCCCD) subjected under response surface methodology (RSM) in optimizing the recoveries of TSC, TPC, DPPH, and ABTS antioxidant capacities of fenugreek seed extract.
2 Materials and methods
2.1 Materials used
The seeds of T. foenum-graecum. L. were obtained from the local markets located in Kuantan city of Pahang, Malaysia. The collected seeds were first separated from unknown species and dehumidified under 50°C in an oven. A grinder (Retsch ZM-200, Germany) was used to grind the seeds into powdered size. The crushed seeds were stored in a dark container at 4–5°C before extraction. High-purity analytical chemicals and reagents such as ethanol, methanol, DPPH, Folin-Ciocalteu reagent, ABTS+•, vanillin, diosgenin, gallic acid (GA), sodium carbonate, and sulfuric acid were received from Sigma Aldrich (M) Sdn. Bhd. Malaysia.
2.2 Extraction of fenugreek seeds
Ten grams of powdered fenugreek seeds was extracted using MAE method under different extraction parameters such as irradiation time (2–4 min), microwave power (500–700 W), ethanol concentration (40–80%), solid-to-solvent ratio (1:8–1:12 g/mL), and a constant microwave temperature of 70 °C. The factor levels for the optimization process were selected based on one-factor-at-one-time (OFAT) experimental method (Fig. 1). The levels of extraction parameters were designed based on the Design-Expert 7.0 (DOE) software® (Stat-Ease, Inc., USA). The seeds were extracted using a closed system of ethos microwave extractor (Frequency 2450, 1000 W; Milestone, Italy). The samples were pretreated and posttreated for 2 min before and after microwave extraction time. The purposes of pretreatment and posttreatment were to heat the samples efficiently while absorbing the heat; however, posttreatment was applied to cool up the samples at room temperature. After removing the samples from the microwave oven, the mixture was filtered via Advantec® qualitative No. 1 filter paper using a vacuumed filtration system, and the aqueous phase was transferred into a glass flask to evaporate the solvent using a rotary evaporator (R-200; Büchi, Germany) at 50°C. To prevent the biodegradation of bioactive compounds, the extract was kept at 4°C in a fridge for further analysis. All the experiments were triplicated.
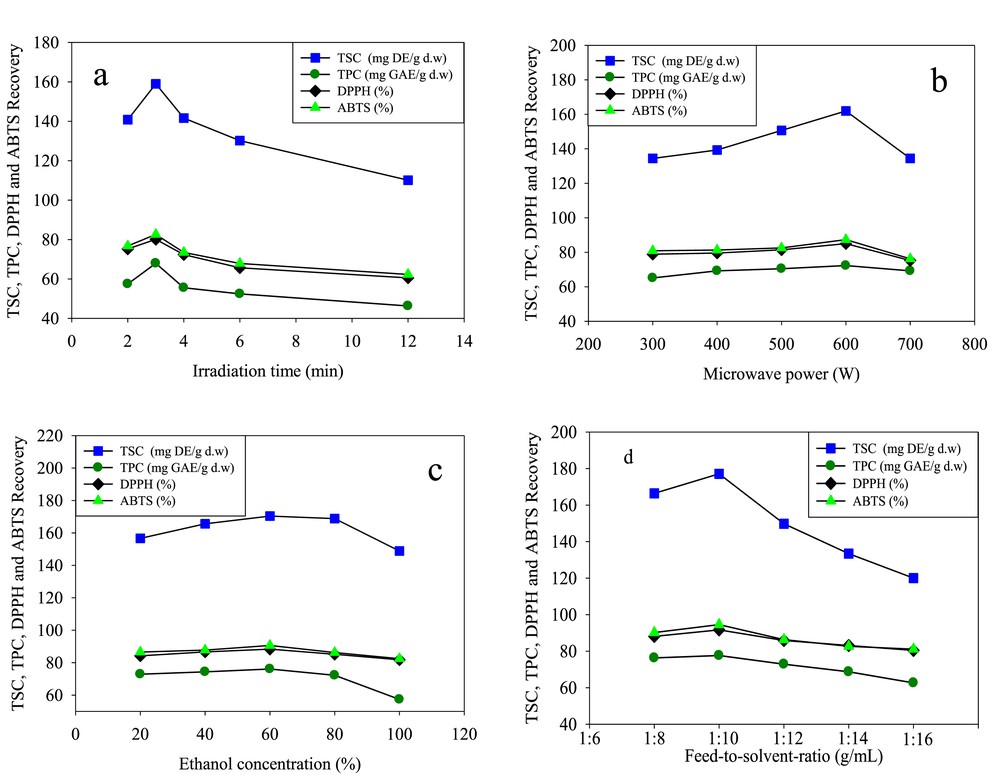
Experimental result of OFAT carried out before optimization process; (a) time of irradiation, (b) proposed microwave oven power, (c) ethanol concentration, (d) ratio of feed to solvent. OFAT, one-factor-at-one-time; TSC, total saponin content; TPC, total phenolic content; DPPH, 2,2-diphenyl-1-picrylhydrazyl; ABTS, 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid).
2.3 Total saponin content measurement
The measurements of TSC were performed using combined and modified methods of Moyo et al. [10], Hu et al. [11], and Venegas-Calerón et al. [12]. Concisely, 0.2 mL of fenugreek seed extract was added to 0.80 mL of absolute methanol and 0.35 mL of vanillin (8% in ethanol) in a 15-mL glass test tube. Then, 1.25 mL of sulfuric acid (72%) was added and manually mixed for a few seconds while the test tube cap was closed. Afterward, the caps were loosed and test tubes were transferred into an electrical water heating bath at 60°C for 10 min. Then, ice crystals were used to cool the samples for 5 min. The TSC in the extract was then determined using a UV-vis spectrophotometer device (U-1800; Hitachi, Japan) at a wavelength of 544 nm. The standard curve of diosgenin (y = 0.0001x + 0.0408; R2 = 0.9945) was obtained based on different concentrations (100–600 mg/mL), and TSC in the extract was presented as milligram of diosgenin equivalent (DE) per gram of dry weight (mg DE/g d.w.). The absorbance was read against methanol (blank), and the measurements were repeated thrice. Then, the total content of saponin was calculated using Eq. 1.
| (1) |
2.4 Total phenolic content measurement
The determination of TPC in the extract was carried out using the technique described by Sookjitsumran et al. [13] and Nickel et al. [14] with little changes. Initially, 200 μL of the extract was mixed with 200 μL of Folin-Ciocalteu reagent in a test tube, and the mixture was stored in a dark place for 5 min at 26 ± 1°C. Afterward, 600 μL of 20% Na2CO3 solution was added into the test tube and incubated for 2 h at the same condition. A UV–vis spectrophotometer was used to measure the absorbance of the mixture at a wavelength of 765 nm against methanol (blank). The standard curve (100–500 mg/mL) was prepared from gallic acid to calculate the concentration of the sample. The obtained result was expressed as milligram of gallic acid equivalent (GAE) per gram of the extract (mg GAE/g d.w.). Eq. 2 was used to calculate the amount of TPC in the extract.
| (2) |
2.5 Antioxidant activities
2.5.1 DPPH radical–scavenging activity
The 2,2-diphenyl-1-picrylhydrazyl assay was accomplished using the technique illustrated by Alara et al. [15] with slight amendments. Initially, 200 μL of the extract diluted in distilled water and 200 μL of DPPH prepared solution (0.1 mM) were added into a glass test tube. The prepared mixture was kept for 30 min in the absence of light at constant room temperature. Finally, the absorbance of the samples was measured at 517 nm using a UV–vis spectrophotometer. The DPPH inhibition (%) was calculated using Eq. 3 against methanol (blank).
| (3) |
2.5.2 ABTS radical–scavenging activity of the extract
The ABTS+• assay was applied to evaluate the free radical–scavenging activity of the extract from fenugreek seeds based on the procedures explained by Cheng et al. [16] and Zielinski et al. [17] with slight variations. Initially, 7 mM ABTS stock solution in distilled water (solution a) and 2.45 mM K2S2O8 (potassium persulfate) in distilled water (solution b) were mixed in an equal ratio of 1:1 (v/v) and incubated for 12–16 h in the absence of light at room temperature. Then, 1 mL of the prepared solution was diluted in 60 mL of pure methanol to obtain a constant absorbance of 1.1 ± 0.02 at 734 nm via a spectrophotometric method. Afterward, 150 μL of fenugreek seed extract was mixed with 2850 μL of ABTS prepared solution. The mixture was then stored in a dark place for 2 h at 26 ±1°C, and the absorbance reading was taken at 734 nm. Ethanol was used as the blank. The capacity of antioxidants on ABTS radical was calculated using Eq. 4.
| (4) |
2.6 Experimental design (optimization)
The optimal conditions of MAE were determined using RSM, with FCCCD at three levels and four independent factors which include extraction time (X1), microwave oven power (X2), ethanol concentration (X3), and feed-to-solvent ratio (X4). The ranges of factors levels for optimization were selected through preextraction of OFAT experimental results. Table 1 shows the center points and different levels of independent variables based on this experiment. Four different responses such as total saponin content (YTSC), total phenolic content (YTPC), ABTS (YABTS), and DPPH (YDPPH) were recorded, respectively. To estimate the sum of square error, six center points with a total number of 30 experimental runs were used. The quadratic polynomial model was generated to correlate the responses. The generated second-order polynomial quadratic equation of the responses is as follows (Eq. 5):
| (5) |
Experimental factors with their levels used in the experiment.
| Factors (input) | Factor levels | ||
| −1 | 0 | +1 | |
| Extraction time, X1 (min) | 2 | 3 | 4 |
| Microwave oven power, X2 (W) | 500 | 600 | 700 |
| Ethanol concentration, X3 (%) | 40 | 60 | 80 |
| Feed-to-solvent ratio, X4 (g/mL) | 1:8 | 1:10 | 1:12 |
Analysis of variance (ANOVA) was applied to find the interaction between independent variables and responses. The significant terms of the model were also obtained through ANOVA. Adequacy of the model was identified based on R-squared, adj R-squared, predicted R-squared, F-value, and lack of fit. The ranges of predicted values to the average prediction errors were compared using adequate precision.
The four independent variables including X1, X2, X3, and X4 were optimized using FCCCD design under RSM. RSM is a useful statistical collection which is very effective for obtaining the influences of various factors on responses through a mathematical technique. Table 2 shows the central composite design of independent factors in coded values and obtained responses of actual and predicted values of YTSC, YTPC, YDPPH, and YABTS. Design-Expert (DOE), a statistical software, was used to design the experiment.
Central composite design of coded factors and responses based on actual and predicted values.
| Run | Factors | Responses | ||||||||||
| X1 | X2 | X3 | X4 | YTSC (Act) | YTSC (Pred) | YTPC (Act) | YTPC (Pred) | YDPPH (Act) | YDPPH (Pred) | YABTS (Act) | YABTS (Pred) | |
| 1 | 0 | 0 | 0 | 0 | 191.22 | 192.80 | 80.10 | 81.0 | 93.35 | 93.33 | 96.44 | 96.14 |
| 2 | 1 | −1 | −1 | −1 | 170.46 | 169.54 | 63.70 | 63.7 | 87.92 | 87.62 | 91.10 | 90.88 |
| 3 | 0 | 1 | 0 | 0 | 178.12 | 177.12 | 77.40 | 77.8 | 91.67 | 92.02 | 94.29 | 94.81 |
| 4 | −1 | −1 | −1 | 1 | 146.42 | 146.80 | 60.36 | 60.5 | 87.73 | 87.33 | 90.54 | 90.32 |
| 5 | −1 | 1 | −1 | −1 | 140.22 | 141.75 | 68.05 | 68.0 | 86.80 | 86.80 | 89.61 | 89.55 |
| 6 | 0 | 0 | 0 | 0 | 195.02 | 192.80 | 81.40 | 81.0 | 93.73 | 93.33 | 96.63 | 96.14 |
| 7 | 0 | 0 | 0 | −1 | 187.38 | 187.54 | 77.92 | 78.1 | 93.35 | 93.44 | 96.16 | 96.41 |
| 8 | 1 | 1 | 1 | 1 | 131.62 | 132.59 | 61.28 | 61.5 | 85.86 | 85.79 | 88.67 | 88.58 |
| 9 | 0 | 0 | 0 | 0 | 196.02 | 192.80 | 82.60 | 81.0 | 94.48 | 93.33 | 97.28 | 96.14 |
| 10 | −1 | 1 | 1 | −1 | 175.62 | 176.31 | 65.92 | 66.3 | 89.71 | 89.68 | 92.52 | 92.42 |
| 11 | 1 | 0 | 0 | 0 | 189.02 | 189.92 | 74.40 | 74.8 | 90.82 | 91.36 | 93.63 | 94.16 |
| 12 | −1 | −1 | 1 | −1 | 184.58 | 183.23 | 69.57 | 69.3 | 90.73 | 90.63 | 93.54 | 93.59 |
| 13 | 1 | −1 | 1 | −1 | 180.58 | 182.77 | 68.24 | 68.5 | 89.51 | 89.70 | 92.32 | 92.39 |
| 14 | 0 | 0 | 0 | 0 | 197.02 | 192.80 | 82.16 | 81.0 | 94.29 | 93.33 | 97.19 | 96.14 |
| 15 | 0 | 0 | 0 | 0 | 197.82 | 192.80 | 81.56 | 81.0 | 94.29 | 93.33 | 97.00 | 96.14 |
| 16 | 0 | −1 | 0 | 0 | 180.02 | 186.10 | 79.06 | 79.7 | 92.23 | 92.98 | 95.13 | 95.84 |
| 17 | 1 | 1 | 1 | −1 | 160.66 | 159.82 | 61.06 | 60.6 | 87.64 | 87.76 | 90.45 | 90.52 |
| 18 | 1 | −1 | −1 | 1 | 146.86 | 145.32 | 62.76 | 62.4 | 87.45 | 87.48 | 90.26 | 90.20 |
| 19 | 1 | 1 | −1 | 1 | 133.38 | 134.28 | 61.60 | 61.6 | 86.70 | 86.52 | 89.51 | 89.31 |
| 20 | −1 | 1 | −1 | 1 | 154.82 | 151.79 | 64.88 | 64.7 | 87.55 | 87.36 | 90.36 | 90.13 |
| 21 | −1 | 1 | 1 | 1 | 164.18 | 164.65 | 64.40 | 64.0 | 88.30 | 88.32 | 91.10 | 91.18 |
| 22 | −1 | 0 | 0 | 0 | 194.72 | 198.91 | 75.96 | 76.7 | 91.67 | 92.24 | 94.48 | 95.17 |
| 23 | −1 | −1 | 1 | 1 | 153.98 | 152.87 | 65.80 | 65.7 | 89.14 | 89.18 | 91.95 | 91.79 |
| 24 | 0 | 0 | −1 | 0 | 167.72 | 171.89 | 74.30 | 74.8 | 88.67 | 89.68 | 91.48 | 92.53 |
| 25 | 0 | 0 | 1 | 0 | 184.02 | 184.94 | 76.33 | 76.9 | 91.10 | 91.20 | 93.63 | 93.81 |
| 26 | 0 | 0 | 0 | 1 | 164.66 | 169.59 | 75.36 | 76.3 | 91.67 | 92.69 | 94.48 | 95.45 |
| 27 | 1 | 1 | −1 | −1 | 139.54 | 139.81 | 61.49 | 61.6 | 86.61 | 86.57 | 89.42 | 89.43 |
| 28 | 0 | 0 | 0 | 0 | 195.02 | 192.80 | 81.46 | 81.0 | 93.16 | 93.33 | 95.97 | 96.14 |
| 29 | 1 | −1 | 1 | 1 | 138.82 | 136.84 | 68.48 | 68.2 | 87.92 | 87.64 | 89.98 | 89.89 |
| 30 | −1 | −1 | −1 | −1 | 157.26 | 155.45 | 65.17 | 65.0 | 86.80 | 86.87 | 90.36 | 90.29 |
2.7 Model verification and statistical analysis
The adequacy of the model was validated by repeating the experimental run at optimum conditions of recovery yields (TSC, TPC, ABTS, and DPPH radical–scavenging activities of the extract). The obtained results were then verified by comparing the predicted value with actual values obtained from the experimental work. To verify the accuracy of the generated model, the experimental runs were triplicated at the optimum conditions.
2.8 Compound analysis via liquid chromatography–quadrupole–time-of-flight mass spectrometry
The analysis of liquid chromatography–quadrupole–time-of-flight mass spectrometry (LC–MS–QTOF) was performed using a mass spectrometer (Vion IMS QTOF; Waters, USA). The mobile phase was prepared by the composition of water and acetonitrile at different concentrations and operated with an injection volume of 20 μL and a flow rate of 0.5 mL/min. The mass spectrometry (MS) condition was set at 100–1000 m/z. The column, sample, and desolvation temperatures were set to 40, 15, and 550°C, respectively. The capillary voltage was 1.50 kV. Identifications of saponin and phenolic bioactive compounds of the extract were carried out in positive and negative ion modes.
3 Results and discussion
3.1 Investigation of extraction parameter influences
3.1.1 Influence of microwave irradiation time
Studies have revealed that the percentage of extraction yield tends to increase as the irradiation time increases in an MAE technique; however, higher exposure to microwave radiation can cause the degradation of biologically active compounds [18]. Therefore, the selection of proper time in an MAE is necessary to obtain high recovery in terms of quality and quantity. In this study, the recoveries of TSC, TPC, DPPH, and ABTS antioxidant capacities were examined at different microwave extraction times (2–4 min), while the four other factors including irradiation time, microwave power, concentration of ethanol, and feed-to-solvent ratio were considered at center points 600 W, 60%, and 1:10 g/mL, respectively. The results revealed that irradiation time has a significant influence on the recovery of bioactive compounds. As it is seen in Table 2 and Figs. 2–5 (a, b and c), at the beginning of extraction time (2 min), the recoveries of TSC, TPC, DPPH, and ABTS were recorded to be 194.72 (mg DE/g d.w.), 75.96 (mg GAE/g d.w.), 91.67%, and 94.48%, respectively. These show significant enhancement in the recoveries of TSC, TPC, DPPH, and ABTS yields attained at 3 min (center point) in an average base of six center points which were 195.32 (mg DE/g d.w.), 81.55 (mg GAE/g d.w.), 93.88%, and 96.75%, respectively. However, once the extraction time reached 4 min, the yields declined to 189.02 (mg DE/g d.w.), 74.40 (mg GAE/g d.w), 90.82%, and 93.63%, respectively. This might be due to the degradation of bioactive compounds when the sample is exposed to microwave radiation beyond 3 min. The results obtained from this study is in good agreement with those found by Xu et al. [18] and Maeng et al. [19] as they also reported 3 min as an optimum irradiation time for MAE of saponins, phenolics, and antioxidant capacities from Pulsatilla turczaninovii and the Coriolus versicolor mushroom, respectively.
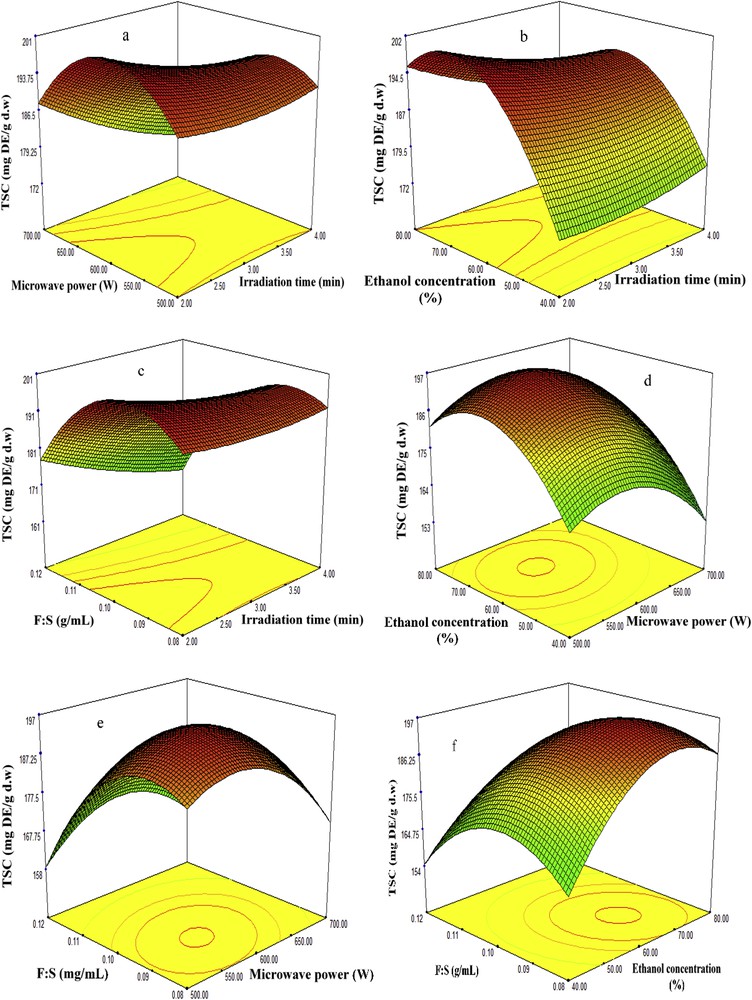
Three-dimensional plots of fenugreek seed extract showing the influence of MAE factors on recovery of TSC. MAE, microwave-assisted extraction; TSC, total saponin content.
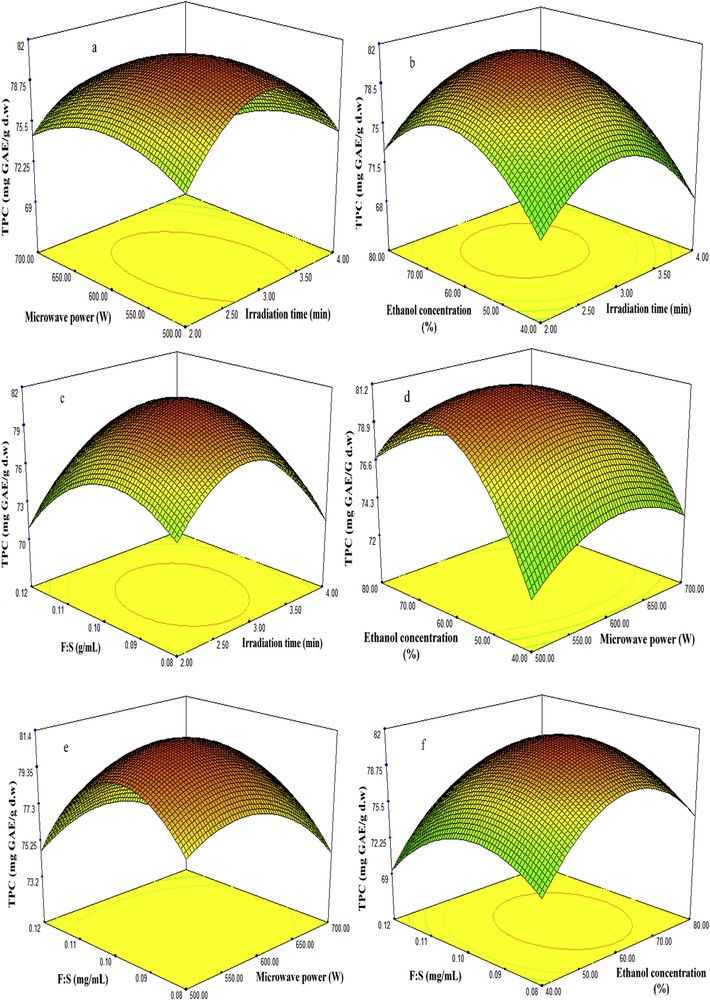
Three-dimensional plots of fenugreek seed extract showing the influence of MAE factors on recovery of TPC. MAE, microwave-assisted extraction; TPC, total phenolic content.
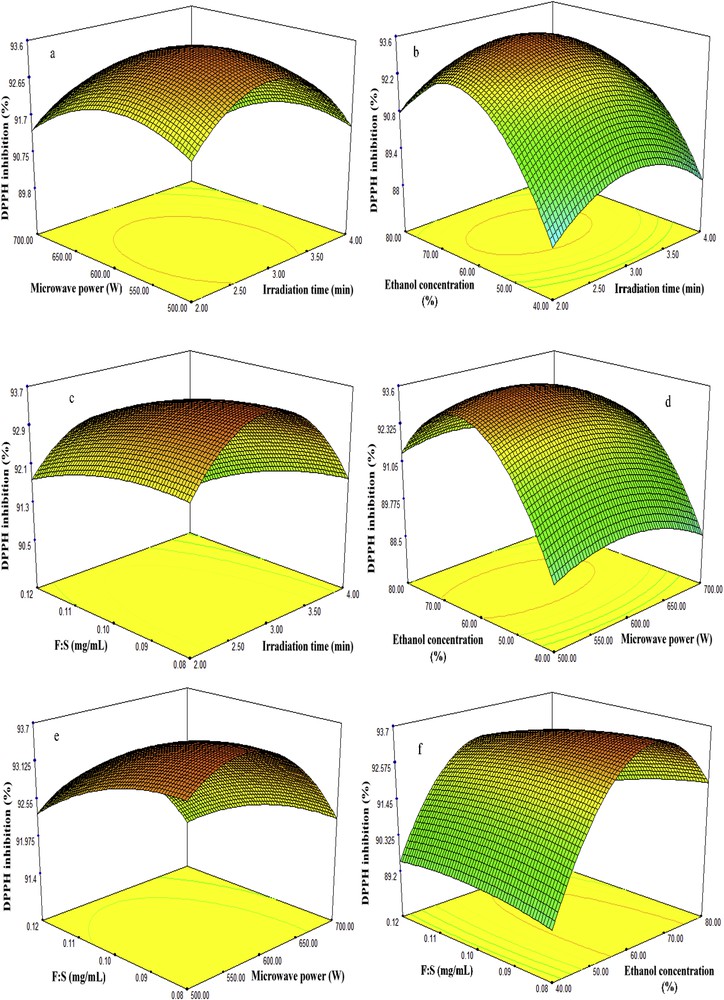
Three-dimensional plots of fenugreek seed extract showing the influence of MAE factors on recovery of DPPH antioxidant capacity. MAE, microwave-assisted extraction; DPPH, 2,2-diphenyl-1-picrylhydrazyl.
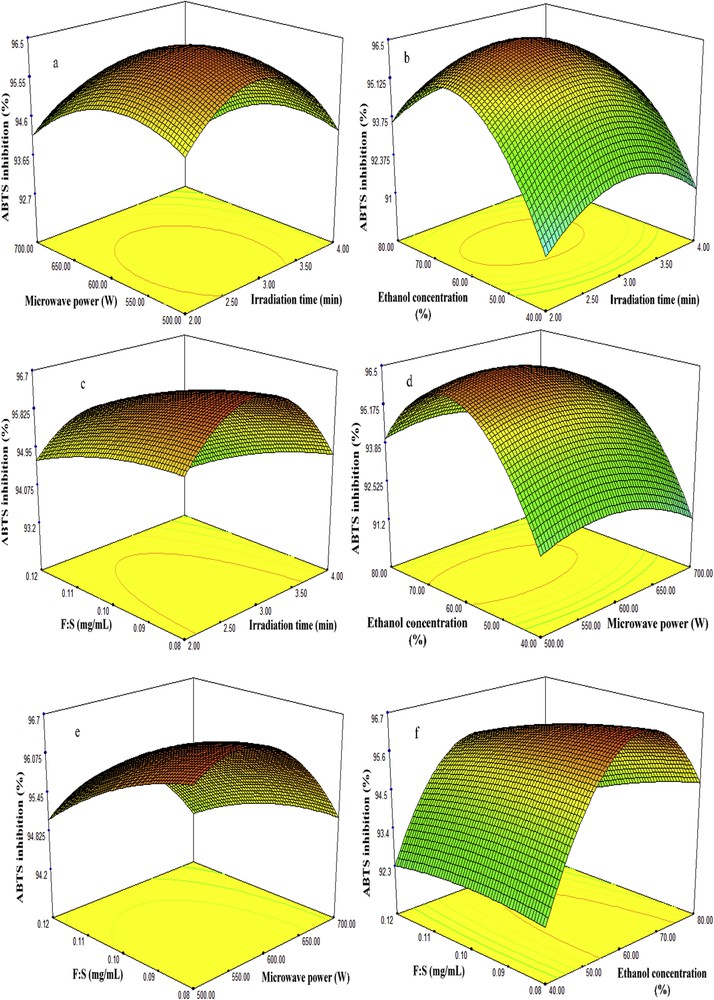
Three-dimensional plots of fenugreek seed extract showing the influence of MAE factors on recovery of ABTS antioxidant capacity. MAE, microwave-assisted extraction; ABTS, 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid).
3.1.2 Influence of microwave oven power
The effects of microwave power on the recovery of TSC, TPC, DPPH, and ABTS from fenugreek seed extract were investigated under different ranges (500–700 W). Table 2 and Figs. 2–5 (a, d, and e) reflected that at 500 W, the recoveries of TSC, TPC, DPPH, and ABTS were 180 mg DE/g d.w., 79.06 mg GAE/g d.w., 92.23%, and 95.13%, respectively. As the microwave power reached 600 W, significant increase of 195.32 mg DE/g d.w., 81.55 mg GAE/g d.w., 93.88%, and 96.75% was observed, respectively; these values were obtained from the average of six center points in central composite design. However, the recoveries declined significantly to 178.12 mg DE/g d.w., 77.40 mg GAE/g d.w., 91.67%, and 94.26% when the level of microwave power was increased to 700 W. The reduction of these bioactive compounds and antioxidant capacities beyond 600 W was also reported by Karabegovic et al. [20], Ling et al. [21], and Shao et al. [22] from MAE of Prunus laurocerasus leaves, Radix astragali, and Perilla Frutescens leaves, respectively. They claimed that higher microwave power results in the thermal degradation of plant matrix and thus declined the yields.
3.1.3 Influence of ethanol concentration
Ethanol has been extensively used for the extraction of biologically active compounds from different plants because of its low toxicity. To enhance the extraction efficiency in MAE, ethanol is usually used at different concentrations in water. The high dielectric property of water turned it to a good cosolvent for the extraction of phytochemicals using MAE. Figs. 2–5 (b, d and f) illustrate the effect of 40–80% ethanol on the recoveries of TSC, TPC, DPPH, and ABTS. As seen, the recoveries of these bioactive compounds at 40% ethanol were 167.72 mg DE/g d.w., 74.30 mg GAE/g d.w., 88.67%, and 91.48%. Further increase in ethanol concentration to 60% enhanced the yields of recoveries to 195.32 mg DE/g d.w., 81.55 mg GAE/g d.w., 93.88%, and 96.75%. However, when ethanol concentration reached 80% in the extraction medium, the yields declined to 184.02 mg DE/g d.w., 76.33 mg GAE/g d.w., 91.10%, and 93.63%.
Ethanol has low polarity, while water has high polarity; these two solvents can be mixed at any concentration depending on the extraction condition of the plant matrix. Ethanol alone is not capable of extracting more bioactive compounds as most of the bioactive components such as saponins, phenolics, and flavonoids are high in polarity. Thus, a polar solvent is required to enhance the recovery yield. On the other hand, more than 45% of the water in ethanol can decline the recovery of TSC and TPC [23]. As fenugreek seed contains water-soluble gums, with higher water content, more gums will be extracted rather than TSC and TPC, and this can generate problem during the filtration process after the MAE. The results from this study are similar to those of the study carried out by Amid and Mirhosseini [23] where biopolymer was obtained from Durio zibethinus seed. It was reported that the protein fractions are more soluble in alcohols, while polysaccharide fractions (gums) have better solubility in water rather than alcohols. Therefore, the yields of TSC and TPC increased by increasing the concentration of ethanol to 60%. As the concentration of ethanol exceeded 80%, significant declination was observed in both TSC and TPC of fenugreek seeds because of protein denaturation in the extraction medium [24]. Indeed, the reduction of TPC means less OH group in the extract and hence lower antioxidant activity. This result was in good correlation with the findings of Ling et al. [21], who studied the MAE of polyphenols and saponins from R. astragali and reported that there was no significant difference between 60 and 70% ethanol concentration of bioactive compounds.
3.1.4 Influence of feed-to-solvent proportion
Feed-to-solvent ratio is one of the important parameters of MAE technique. In industrial processes, it is essential to maximize the recovery yield of extraction and reduce the consumption of extraction solvent [25]. In this study, the recoveries of TSC and TPC from fenugreek seed extract increased from 187.38 mg DE/g d.w. and 77.92 mg GAE/g d.w. to 195.32 mg DE/g d.w. and 81.55 mg GAE/g d.w., respectively, as the feed-to-solvent ratio increased from 1:8 to 1:10 g/mL. However, further increment in the ratio to 1:12 g/mL minimized the recoveries of TSC and TPC to 164.66 mg DE/g d.w. and 75.36 mg GAE/g d.w., respectively (Figs. 2–5c, e and f); at the same time, the DPPH and ABTS antioxidant capacities declined. An increase in the ratio from 1:8 to 1:10 g/mL provided a better wettability and microwave adsorption of plant matrix during the microwave heating. On the other hand, further enhancement of the ratio to 1:12 g/mL means more ethanol and water content in the extraction medium, which results in more adsorption of microwave energy because of the dielectric property of water and hence reduced the yields. Besides, higher solvent generates more heat within the microwave system which may also lead to the evaporation of thermolabile compounds. The obtained findings were in correlation with previous results reported by Alara et al. [26] on MAE of phytochemicals from Vernonia amygdalina leaf at a feed-to-solvent ratio of 1:10 g/mL as an optimized condition.
3.2 Optimization process
3.2.1 Model fitting using RSM
The ranges of factors for optimization of MAE process parameters were selected based on OFAT experiments. The independent variables affecting the recoveries of TSC, TPC, DPPH, and ABTS were irradiation time X1 (2–12 min), microwave oven power X2 (300–700 W), ethanol concentration X3 (20–100%), feed-to-solvent ratio X4 (1:8–1:16 g/mL), and a constant temperature of 70 °C. The factor levels for optimization of TSC, TPC, DPPH, and ABTS based on OFAT results (Fig. 1) were selected to be X1 (2–4 min), X2 (500–700 W), X3 (40–80%), and X4 (1:8–1:12 g/mL). The extraction temperature was considered constant at 70°C as it indicated no considerable influence on the recovery yields. The obtained optimal responses were 195.32 mg DE/g d.w., 81.55 mg GAE/g d.w., 93.88%, and 96.75%, respectively, based on the average of six center points (optimum conditions). The results of experimental design based on central composite design and corresponding responses are shown in Table 2. The second-order polynomial equations of optimized conditions for TSC, TPC, DPPH, and ABTS are shown in Eqs. 6–9.
| (6) |
| (7) |
| (8) |
| (9) |
Analysis of variance of quadratic model for optimization of bioactive compounds from fenugreek seeds.
| Factors | TSC | TPC | DPPH | ABTS | |||||||||
| Source | Sum of squares | F-value | p-value, Prob > F | Sum of squares | F-value | p-value, Prob > F | Sum of squares | F-value | p-value, Prob > F | Sum of squares | F-value | p-value, Prob > F | |
| Model | 13174.54 | 71.34 | <0.0001 | 1709.256 | 201.23 | <0.0001 | 209.2423 | 31.03 | <0.0001 | 209.0103 | 30.12 | <0.0001 | Significant |
| X1 | 363.2411 | 27.54 | <0.0001 | 16.2450 | 26.78 | 0.0001 | 3.527314 | 7.32 | 0.0163 | 4.592228 | 9.26 | 0.0082 | |
| X2 | 362.8818 | 27.51 | <0.0001 | 16.18173 | 26.67 | 0.0001 | 4.113543 | 8.54 | 0.0105 | 4.764064 | 9.61 | 0.0073 | |
| X3 | 765.4480 | 58.03 | <0.0001 | 19.56599 | 32.25 | <0.0001 | 10.39646 | 21.58 | 0.0003 | 7.380760 | 14.89 | 0.0015 | |
| X4 | 1448.799 | 109.83 | <0.0001 | 14.64968 | 24.15 | 0.0002 | 2.531952 | 5.26 | 0.0367 | 4.131467 | 8.34 | 0.0113 | |
| X1X2 | 256.9609 | 19.48 | 0.00050 | 25.23388 | 41.59 | <0.0001 | 0.971184 | 2.02 | 0.1761 | 0.496444 | 1.00 | 0.3328 | |
| X1X3 | 211.7025 | 16.05 | 0.00110 | 0.324900 | 0.54 | 0.4756 | 2.848446 | 5.91 | 0.0280 | 3.173269 | 6.40 | 0.0231 | |
| X1X4 | 242.4249 | 18.38 | 0.00060 | 10.38988 | 17.13 | 0.0009 | 0.367567 | 0.76 | 0.3961 | 0.489869 | 0.99 | 0.3359 | |
| X2X3 | 46.10410 | 3.49 | 0.08120 | 34.37868 | 56.66 | <0.0001 | 0.787075 | 1.63 | 0.2206 | 0.175567 | 0.35 | 0.5606 | |
| X2X4 | 349.3161 | 26.48 | 0.00010 | 1.512900 | 2.49 | 0.13520 | 0.008334 | 0.02 | 0.8971 | 0.312992 | 0.63 | 0.4392 | |
| X3X4 | 471.3241 | 35.73 | <0.0001 | 0.993344 | 1.64 | 0.22010 | 3.693379 | 7.67 | 0.0143 | 3.342255 | 6.74 | 0.0202 | |
| 6.768690 | 0.51 | 0.48480 | 72.57962 | 119.63 | <0.0001 | 6.039004 | 12.54 | 0.0030 | 5.655431 | 11.41 | 0.0041 | ||
| 324.0574 | 24.57 | 0.00020 | 13.03207 | 21.48 | 0.00030 | 1.761142 | 3.66 | 0.0752 | 1.750631 | 3.53 | 0.0798 | ||
| 536.0341 | 40.63 | <0.0001 | 68.96887 | 113.68 | <0.0001 | 21.55557 | 44.75 | <0.0001 | 22.93975 | 46.28 | <0.0001 | ||
| 524.9124 | 39.79 | <0.0001 | 38.12659 | 62.84 | <0.0001 | 0.178754 | 0.37 | 0.5515 | 0.117972 | 0.24 | 0.6327 | ||
| Residual | 197.873 | 9.100621 | 7.225233 | 7.434844 | |||||||||
| Lack of fit | 171.2597 | 3.22 | 0.1045 | 5.485621 | 0.76 | 0.6688 | 5.723131 | 1.91 | 0.2471 | 6.178222 | 2.46 | 0.1664 | Not significant |
| Pure error | 26.61333 | 3.615 | 1.502102 | 1.256622 | |||||||||
| Cor total | 13372.41 | 1718.357 | 216.4676 | 216.4451 | |||||||||
| C.V. % | 2.137829 | 1.095657 | 0.770905 | 0.758244 | |||||||||
| PRESS | 738.8179 | 25.91467 | 20.99762 | 20.45925 | |||||||||
| R2 | 0.9852 | 0.994704 | 0.966622 | 0.96565 | |||||||||
| Adj R2 | 0.9714 | 0.989761 | 0.935469 | 0.93359 | |||||||||
| Pred R2 | 0.9448 | 0.984919 | 0.902999 | 0.905476 | |||||||||
| Adeq precis | 25.8237 | 37.25518 | 15.57944 | 15.73003 |
3.3 Validation of the model
Validations of the models were carried out based on the optimum extraction condition of RSM at 2.84 min, 572.50 W, 63.68%, and 0.09 g/mL. Based on the optimum condition, the responses of TSC, TPC, DPPH, and ABTS were 196.48 mg DE/g d.w., 81.01 mg GAE/g d.w., 93.61%, and 96.48%, respectively. The experimental runs were performed in triplicate based on the suggested optimum conditions, and the obtained results were 195.89 ± 1.07 mg DE/g d.w., 81.85 ± 0.61 mg GAE/g d.w., 92.86 ± 0.56%, and 95.85 ± 0.81%, respectively. This indicated that there is a desirable agreement between predicted and experimental (actual) values. By applying the paired t-test, no significant variance between actual and predicted values (p < 0.05) was observed. Therefore, it has been proved that the generated response model was adequate in predicting optimal conditions which is suitable for the study.
3.4 LC–MS–QTOF analysis of extracted compounds
The identification of saponin and phenolic bioactive compounds of fenugreek seed extract was performed using LC–MS–QTOF analysis. A total of 50 compounds containing saponins, phenolics, alkaloids, flavonoids, triterpenes, and steroid glycosides were identified. These natural biologically active compounds are responsible for many activities in the human body such as antioxidant, antiinflammatory, antidiabetic, and anticancer activities [28]. As seen in Table 4, most of the components are belonging to steroid and terpenoid saponins. It is also seen that different types of phenolic compounds such as phenolic aldehydes, phenolic acids, polyphenols, and flavonoids were present in the extract. Phenolic compounds including protocatechuic acid, cistanoside C, campneoside, forsythoside E, and steroidal saponins such as terrestrosin, timosaponin, markogenin, protodiosgenin, yamogenin, and sarsaponin have been reported to possess anticancer, antidiabetic, antiinflammatory, antibiotic, antioxidant, hormone-balancing, and antidepressant properties [28–30]. Cistanoside C is used to repair DNA damage [31]. Figs. 6 and 7 show the identified compounds of fenugreek seeds through LC–MS–QTOF analysis based on retention time and observed intensity of positive and negative ion modes, respectively. It is seen in Fig. 6 that protodiosgenin, timosaponin B-2, and cimicifugic acid B are the most representative compounds with the intensity of more than 1 × 106 in positive ion mode. Fig. 7 indicates (−)-suspensaside B, kuzubutenolide A, and smilaxin are the compounds with the highest intensity of more than 1 × 105 in negative ion mode.
Saponin and phenolic compounds of fenugreek seed extract identified by LC–MS–QTOF.
| No. | Compound name | Chemical formula | Observed RT (min) | Observed m/z | Adducts | Total fragments found |
| 1 | 6′-O-Galloylhomoarbutin | C20H22O11 | 0.51 | 437.1090 | -H | 1 |
| 2 | Meliadanoside B | C15H20O8 | 0.59 | 327.1082 | -H | 2 |
| 3 | Protocatechuic aldehyde | C7H6O3 | 0.59 | 137.0249 | -H | 0 |
| 4 | 2,4,6-Trihydroxyacetophenone−2,4-di-O-β-d-glucopyranoside | C20H28O14 | 0.65 | 491.1414 | -H | 24 |
| 5 | Cistanoside C | C30H38O15 | 0.65 | 637.2150 | -H | 20 |
| 6 | 2,4,6-Trihydroxyacetophenone−2,4-di-O-β-d-glucopyranoside | C20H28O14 | 0.76 | 491.1415 | -H | 23 |
| 7 | (−)-Suspensaside B | C33H44O16 | 0.87 | 695.2563 | -H | 29 |
| 8 | Campneoside I | C30H38O16 | 0.89 | 653.2090 | -H | 34 |
| 9 | Forsythoside E | C20H30O12 | 0.97 | 461.1677 | -H | 6 |
| 10 | Osmanthuside H | C19H28O11 | 1.18 | 431.1564 | -H | 1 |
| 11 | Decaffeoylacteoside | C20H30O12 | 1.50 | 461.1671 | -H | 8 |
| 12 | Quercetin-3-O-neohesperidoside | C27H30O16 | 1.79 | 611.1615 | +H | 10 |
| 13 | Estrone | C18H22O2 | 2.09 | 293.1492 | +Na | 1 |
| 14 | (+)-Suspensaside A | C29H34O15 | 2.16 | 621.1833 | -H | 6 |
| 15 | Deacetylmatricarin-8-O-β-d-glucopyranoside | C21H28O9 | 2.41 | 425.1809 | +H | 12 |
| 16 | Cimicifugic acid B | C21H20O11 | 2.50 | 449.1076 | +H | 34 |
| 17 | Brazilein | C16H12O5 | 2.61 | 283.0606 | -H | 2 |
| 18 | Erigoster A | C27H26O13 | 2.70 | 557.1304 | -H | 45 |
| 19 | 2,3,5,4′-Tetrahydroxystilbene-2,3-O-β-d-glucopyranoside | C26H32O14 | 2.72 | 567.1711 | -H | 20 |
| 20 | 5,7,2′,5′-Tetrahydroxyflavone | C15H10O6 | 2.74 | 287.0551 | +H | 2 |
| 21 | Kuzubutenolide A | C23H24O10 | 2.80 | 459.1304 | -H | 25 |
| 22 | Dihydroresveratrol | C14H14O3 | 2.80 | 229.0872 | -H | 1 |
| 23 | Smilaxin | C17H16O6 | 2.86 | 315.0875 | -H | 10 |
| 24 | Terrestrosin A | C45H74O18 | 2.90 | 903.4948 | +H | 11 |
| 25 | Terrestrosin E | C45H74O19 | 2.98 | 919.4905 | +H | 66 |
| 26 | Timosaponin D | C45H74O19 | 2.99 | 919.4892 | +H | 66 |
| 27 | Prosapogenin 2 | C32H48O8 | 3.00 | 599.2983 | +K | 19 |
| 28 | 25(S)-Ruscogenin | C27H42O4 | 3.00 | 431.3163 | +H | 12 |
| 29 | Markogenin | C27H44O4 | 3.04 | 433.3314 | +H | 33 |
| 30 | Protodiosgenin | C33H54O9 | 3.04 | 595.3839 | +H | 64 |
| 31 | Zingiberogenin | C27H42O4 | 3.08 | 431.3165 | +H | 17 |
| 32 | Timosaponin B-2 | C45H76O19 | 3.10 | 943.4872 | +Na | 80 |
| 33 | Diosgenone | C27H40O3 | 3.16 | 413.3051 | +H | 17 |
| 34 | Yamogenin | C27H42O3 | 3.25 | 415.3204 | +H | 58 |
| 35 | Sarsaponin | C27H44O3 | 3.25 | 417.3350 | +H | 25 |
| 36 | Prosapogenin A | C39H62O12 | 3.25 | 745.4150 | +Na | 25 |
| 37 | Oleuropein | C25H32O13 | 3.28 | 579.1494 | +K | 12 |
| 38 | epi-Smilagenin | C27H44O3 | 3.32 | 417.3359 | +H | 2 |
| 39 | Atroposide E | C45H74O18 | 3.40 | 925.4777 | +Na | 22 |
| 40 | Cimifoetiside VII | C43H70O16 | 3.48 | 843.4740 | +H | 45 |
| 41 | Abrisaponin I | C48H74O20 | 3.53 | 971.4845 | +H | 45 |
| 42 | Soyasaponin βg | C47H74O17 | 3.67 | 911.4982 | +H | 56 |
| 43 | Ophiogenin | C27H42O5 | 3.69 | 447.3105 | +H | 11 |
| 44 | Kingianoside A | C39H60O14 | 3.82 | 753.4063 | +H | 31 |
| 45 | Hecogenone | C27H40O4 | 3.90 | 429.2999 | +H | 5 |
| 46 | Gracillin | C45H72O17 | 3.92 | 885.4846 | +H | 63 |
| 47 | 25(S)-Ruscogenin | C27H42O4 | 4.23 | 431.3154 | +H | 8 |
| 48 | Hookeroside C | C38H62O15 | 4.29 | 781.3985 | +Na | 32 |
| 49 | Celosin C | C42H66O13 | 4.74 | 779.4591 | +H | 37 |
| 50 | 2-Octylphenol | C14H22O | 5.50 | 205.1601 | -H | 0 |
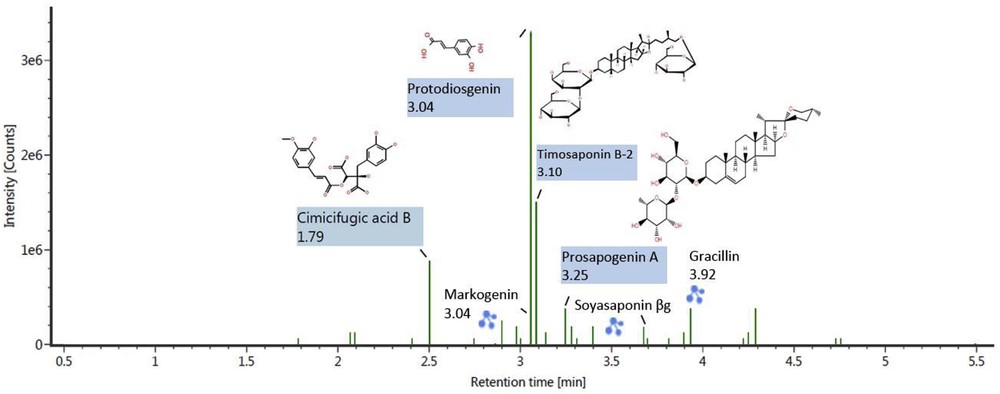
LC–MS–QTOF chromatogram of tentatively assigned compounds of fenugreek seed extract in positive ion mode. LC–MS–QTOF, liquid chromatography–quadrupole–time-of-flight mass spectrometry.
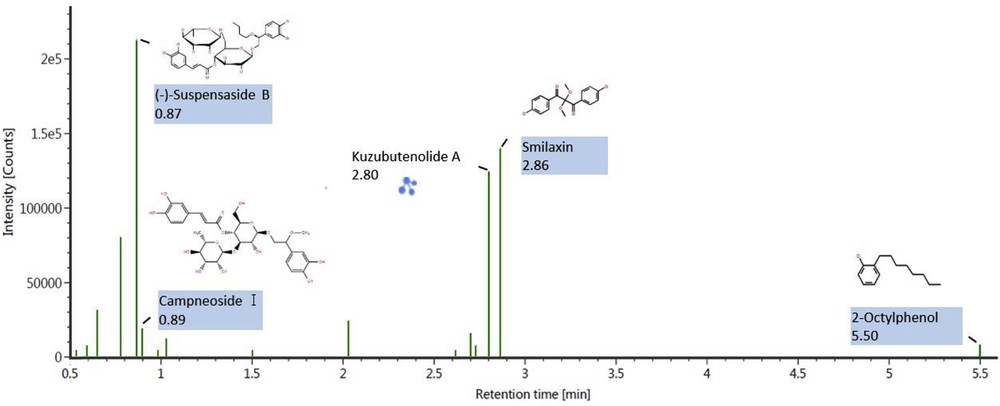
LC–MS–QTOF chromatogram of tentatively assigned compounds of fenugreek seed extract in negative ion mode. LC–MS–QTOF, liquid chromatography–quadrupole–time-of-flight mass spectrometry.
4 Conclusions
In the present study, MAE of TSC, TPC, DPPH, and ABTS antioxidant capacities from fenugreek seeds was optimized. The optimal conditions of parameters were found to be 2.84 min of irradiation time, microwave oven power of 572.50 W, ethanol concentration of 63.68%, and feed-to-solvent ratio of 0.09 g/mL. Based on the suggested optimum conditions, the obtained responses were 195.89 ± 1.07 mg DE/g d.w., 81.85 ± 0.61 mg GAE/g d.w., 92.86 ± 0.56%, and 95.85 ± 0.81%, respectively. The results demonstrated no significant observation between actual and predicted values obtained from the experiment. The results obtained from LC–MS–QTOF analysis confirmed the presence of steroids, triterpenoids, saponins, phenolics, and other natural antioxidants. This study recommends that the models obtained from this study can be applied for further industrial application in the production of saponins, phenolics, and antioxidants in both small and large scales from fenugreek seeds.
Acknowledgements
All authors would like to thank the financial assistance received from Universiti Malaysia Pahang (UMP) under Grant No. PGRS 1803105.



Vous devez vous connecter pour continuer.
S'authentifier