1. Introduction
Primary hyperoxaluria (PH) is one of the most severe hereditary kidney stone diseases and is caused by defects in glyoxylate metabolism, resulting in overproduction of oxalate and eventually end stage renal disease (ESRD). Three different genetic forms have been identified to date, related to mutations on three genes coding for three enzymes, namely alanine glyoxylate aminotransferase (PH1) [1, 2], glyoxylate reductase hydroxypyruvate reductase (PH2) [3, 4] and 4-hydroxy-2-oxo-glutarate aldolase 1 (PH3) [5, 6]. In all cases, the high production of oxalate, mainly by the liver, is the cause of recurrent urolithiasis and nephrocalcinosis which are the main markers of the disease. At the stage of severe chronic kidney disease, plasma oxalate increase leads to a systemic oxalosis characterized by calcium oxalate deposits in all tissues, in particular bones and the cardiovascular system [7, 8]. Furthermore, it accounts for 1 to 2% of cases of pediatric ESRD, according to registries from Europe, the United States, and Japan [1, 9, 10], but it appears to be more prevalent in countries in which consanguineous marriages are common (with a prevalence of 10% or higher in some North African and Middle Eastern nations), but it is certainly underestimated [11]. From an epidemiological point of view, PH1 is the most frequent (60–75% of cases) and unfortunately the most severe form of the disease. Theoretically, the diagnosis of PH should be based on the measurement of oxalate in urine and/or blood and confirmed by genetic determination of the gene mutations. Unfortunately, for economic reasons and logistics, these tools are not available in many countries around the world, thus inducing an absence of diagnosis or a delay in the diagnosis of this pathology with dramatic consequences for the patient [1, 12, 13]. Moreover, due to inadequate pre-analytical conditions by untrained laboratories, oxalate measurement in urine may fail to demonstrate elevated levels because of oxalate loss as a result of massive calcium oxalate crystalluria. Even in western countries, the diagnosis may be missed because the clinical course of the disease is only calcium oxalate stone disease, the composition of calculi being very common since they are made of calcium oxalate, the main component of about 70–90% of the stones throughout the world. Thus, a number of PH cases are diagnosed in patients with ESRD and even after a failed kidney transplant [13, 14, 15]. Actually, data from PH cohorts in industrialized countries show that the diagnosis was only made after progression to ESRD in more than 30% of patients [1, 9, 12, 16].
The number of PH cases in Morocco is currently unknown because PH is under-diagnosed. The objective of this study on a group of Moroccan children is to show that calcium oxalate monohydrate (COM) stone with type Ic morphology is associated with high oxalate excretion, which should lead to PH investigation and genetic assessment.
2. Material and methods
2.1. Study population
This study was based on the current morpho-constitutional stone analysis [17, 18] performed routinely in the Laboratoire de Recherches et d’Analyses Médicales de la Gendarmerie Royale in Morocco. From 1999 to 2020, we analyzed urinary calculi from 614 children aged less than 18 years old, i.e., 419 boys and 195 girls. Because type Ic morphology of stones has been described as a strong marker suggesting PH [18, 19], we focused our attention on patients who formed COM stones with a type Ic morphology. Of our 614 patients, 85 children (53 boys, 32 girls) met this criterion. However, urine samples required for determining oxalate concentration and the oxalate to creatinine ratio were not available for all patients. Only 43 patients who formed Ic-type calculi were able to benefit from additional biochemical exploration including the study of crystalluria and oxalate assay in urine samples. They were compared to a control group of 92 children (55 boys, 37 girls aged from 1 month to 17 years old) who produced stones with other morphological types including only 10 patients with type Ia stones. Ideally, the control group should have included pure calcium oxalate stones with morphologies other than type Ic, but this group, very common in adult patients, hardly exists in children in our series where many stones have an infectious component and include either phosphates or ammonium urate. None of the patients presented with chronic pancreatitis or inflammatory bowel disease or other cause of absorptive hyperoxaluria.
Morphological aspect of pure type Ic calcium oxalate monohydrate stones (surface).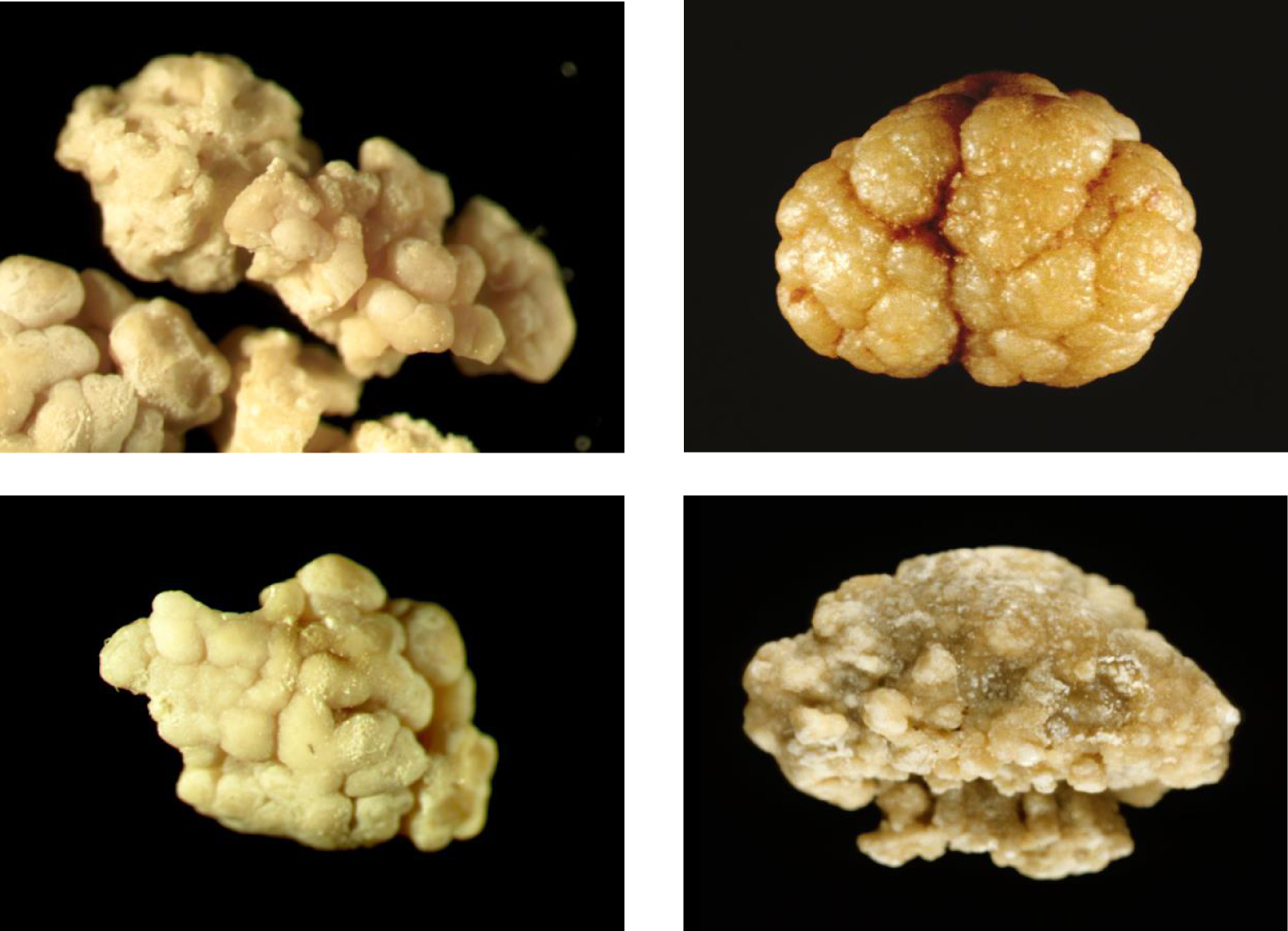
2.2. Methods
2.2.1. Morpho-constitutional analysis of urinary stones
Each calculus was checked for its morphological characteristics using a stereomicroscope to define the morphological type according to the previously reported classification [17, 18] and to select the representative parts of the stone (core, inner layers and surface) for specific infrared analysis. Among calcium oxalate stones, it was shown that type I stones mainly formed of COM are related to hyperoxaluria. By contrast, type II stones are mainly associated to hypercalciuria. Type I may be subdivided in five subtypes, namely Ia, Ib, Ic, Id and Ie [20, 21, 22]. Type Ia stones are essentially related to intermittent hyperoxaluria from dietary origin while type Ic stones are very widely associated with PH and type Ie stones to enteric hyperoxaluria. Type Ic stones, that are strongly suggestive for PH have a particular morphology characterized by a pale, yellowish budding surface and a loose, finely granular and unorganized section with light color, cream to pale yellow-brown as shown in Figures 1 (surface) and 2 (section). Stone morphology is very different from that observed in common type Ia COM stones resulting from dietary habits and/or low diuresis. As shown in Figure 3, type Ia COM stones are characterized by a dark-brown smooth surface with nipples. The stone section is dark-brown in color and made of concentric layers with radial crystallization.
Examples of poorly organized inner section of type Ic stones [23, 24].
Examples of surface and inner section of common type Ia COM stones [23].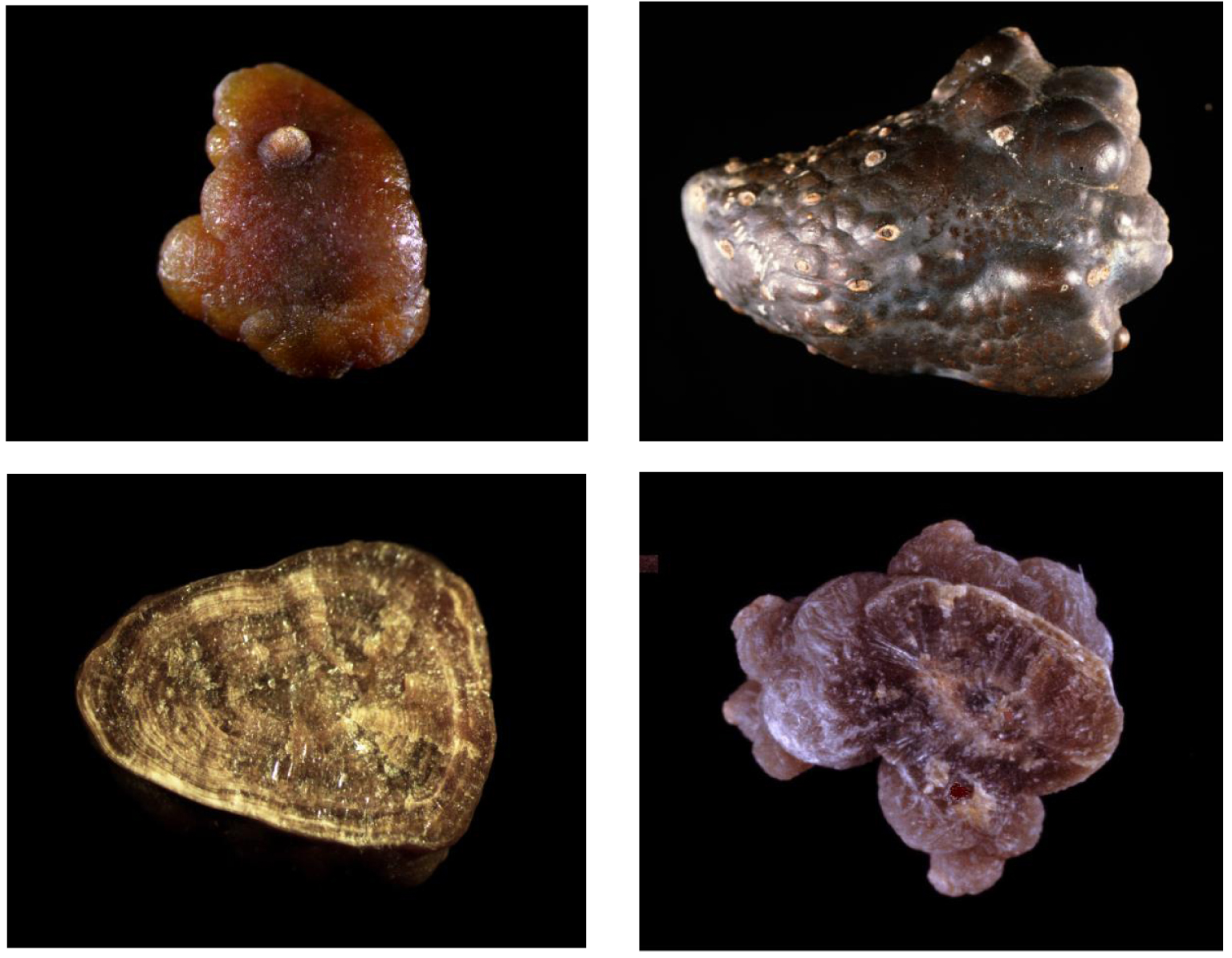
A sequential infrared analysis is the second step of the morpho-constitutional analysis. For this purpose, a fraction of 1–2 mg of each calculus sample selected under the stereomicroscope was pulverized and crushed in 100–200 mg of pure and dry potassium bromide for IR spectroscopy analysis [25]. The mixture was transformed into a transparent pellet of 13 mm in diameter using a press under a pressure of 10 tons/cm2 as previously described [25]. The pellet was then placed in a special holder and inserted into the infrared spectrophotometer for analysis. The instrument used was a Nicolet IS10 Fourier transform infrared spectrometer from Thermo-Nicolet. The infrared spectra were recorded on the wavelength range from 2.5 to 25 μm (4000–400 cm−1) with a resolution of 4 cm−1. Air was used as the reference. The resulting spectra were identified by comparison with available reference spectra [25, 26]. Thereafter, in order to assess the total composition of the stone, each calculus was pulverized in its totality and a fraction of the powder was again used to prepare a pellet to be analyzed.
2.2.2. Crystalluria studies
For half of the patients with type Ic stones (n = 43), at least one and often several urine samples were available for metabolic evaluation and crystalluria analysis. We performed crystalluria studies to confirm the diagnosis and for the follow-up of the patients. Thus, we examined 173 crystalluria in fresh first-voided morning urine samples, according to a uniform protocol described elsewhere [27]. In short, urine samples brought to the laboratory within 2 hours of voiding were kept at room temperature and were rapidly processed. Urine-specific gravity and pH were measured. Undiluted urine was then homogenized by gentle shaking and turning over (neither centrifuged nor filtered) and immediately placed in a Malassez cell (CML, Nemours, France) containing 10 mm3, then examined by light microscopy using a polarizing microscope (Nikon, Champigny-sur-Marne, France). The entire cell was examined at ×200 magnification to localize crystals and aggregates, then at ×400 magnification (high power field). All crystals and aggregates were counted on the entire cell. The results were expressed as number of crystals by mm3.
2.2.3. Metabolic evaluation
Because it is very difficult to collect reliable 24 h-urine samples in children for biochemistry measurements, we determined the concentrations of urine solutes in the first-voided morning urine samples. In every sample, urinary concentrations of calcium, magnesium, phosphate, sodium, urate, urea, proteins and creatinine were measured. All parameters were determined on a Roche Cobas 8000 analyzer. Oxalate concentration was measured enzymatically with oxalate oxydase at 590 nm (Oxalate kit reference: 2401, SOBIODA, France). Citrate concentration was enzymatically determined by UV spectroscopy with citrate lyase at 340 nm.
Lithogenic risk thresholds for oxalate, citrate, magnesium, urate, phosphate and calcium were defined as follows [28, 29, 30]: oxalate > 0.3 mmol/l, citrate < 1 mmol/l, calcium > 3.8 mmol/l, magnesium < 2 mmol/l, urate > 2 mmol/l at pH < 5.5 and >3.5 mmol/l at pH > 6, phosphate > 13 mmol/l at pH > 7.5 and >24 mmol/l at pH < 6.5.
In order to assess the excretion of oxalate, we calculated the ratio of oxalate concentration to creatinine concentration, which is considered a strong marker for heavy hyperoxaluria of genetic origin [31, 32]. The reference values used to interpret the oxalate/creatinine ratio (mmol/mmol) by age group are: ⩽6 months: <0.36; 7–12 months: <0.23; 1–2 years: <0.18; 3–<5 years: <0.10; 5–<12 years: <0.08; ⩾12 years: <0.07 [31].
Data are presented as means ± SEM. Intergroup comparisons used Student t-test, or analysis of variance (ANOVA) for continuous variables, and the 𝜒2 and Fisher’s exact tests for categorical variables. A p value less than 0.05 was considered statistically significant. Statistical procedures used the SPSS.23 software.
3. Results
3.1. Localization of calculi
The anatomical localization was specified for 55 stones (64.7% of cases). The stones were found within the kidney in 84% of cases, in the ureter in 8% and in the bladder in 8%. No statistical difference was found between girls and boys regarding stone location.
3.2. Infrared analysis
The results of stone analysis by Fourier transform infrared (FTIR) spectroscopy are summarized in Table 1. The results are expressed as the main component for each stone. Type Ic morphology being reported as pathognomonic of PH [19, 33, 34], it deserved special attention including at the infrared spectrum level where some differences could be observed by comparison with common COM spectra .
Frequency of main components identified in Moroccan children stones
| Main component | Girls (n = 195) | Boys (n = 419) | Overall (n = 614) |
|---|---|---|---|
| COM | 107 (54.9) | 141 (33.7) | 248 (40.4%) |
| COD | 22 (11.3) | 43 (10.3) | 65 (10.6%) |
| Carbapatite | 15 (7.7) | 45 (10.7) | 60 (9.8%) |
| Brushite | 1 (0.5) | 1 (0.2) | 2 (0.3%) |
| Struvite | 18 (9.2) | 96 (22.9) | 114 (18.6%) |
| Uric acid | 3 (1.5) | 15 (3.6) | 18 (2.9%) |
| Ammonium urate | 17 (8.7) | 41 (9.8) | 58 (9.4%) |
| Cystine | 5 (2.6) | 28 (6.7) | 33 (5.4%) |
| Xanthine | 2 (1.0) | 2 (0.5) | 4 (0.7%) |
| Drugs | 3 (1.5) | 4 (1.0) | 7 (1.1%) |
| Others (proteins, lipids,…) | 2 (1.0) | 3 (0.7) | 5 (0.8%) |
COM was the most frequent main component of stones, found in 248 calculi (40.4%), followed by MAP (114 stones, 18.6%) and COD (65 stones, 10.6%). However, only 110 stones contained more than 95% COM. Among the 85 Moroccan children who had type Ic calculi, 83 formed stones containing more than 95% COM. They accounted for 75.5% of cases. One stone with Ic morphology actually had a mixed morphology Ic + IIa and contained 82% COM and 18% COD. The last one contained 90% COM and 10% carbapatite.
The molecular and crystalline identification of different areas of the type Ic COM stones analyzed by FTIR spectroscopy showed a chemical composition similar to that of most urinary calculi, i.e., mainly consisting of calcium oxalate monohydrate (COM, whewellite) which was the main component of all stones. COM is characterized by its bands on the infrared spectrum at 1619 cm−1, 1316 cm−1 and 780 cm−1 (Figure 4). Of note, by contrast with synthetic whewellite, the infrared spectrum of all urinary stones exhibits an additional broad vibration about 1100 cm−1 (black arrow in Figure 4A), the intensity of which being close to the absorption of the minor peak of whewellite at 886 cm−1. That vibration does not belong to whewellite and results from the 𝜈3 vibration of the PO4 group present in organic material such as uropontin which is known to have a strong affinity for several crystalline faces (100/121) of COM [35]. It has been shown that this organic phosphate may be converted into apatite after pyrolysis of COM stones [36].
Infrared spectra of pure calcium oxalate monohydrate (COM) stones: (A) spectrum of common biological whewellite; (B) spectrum of whewellite found in a number (not all) type Ic stones; (C) detailed part of the spectrum shown in Figure 4A; (D) detailed part of the spectrum shown in Figure 4B (note the weak absorbance of the 1100 cm−1 area by comparison with Figure 4C).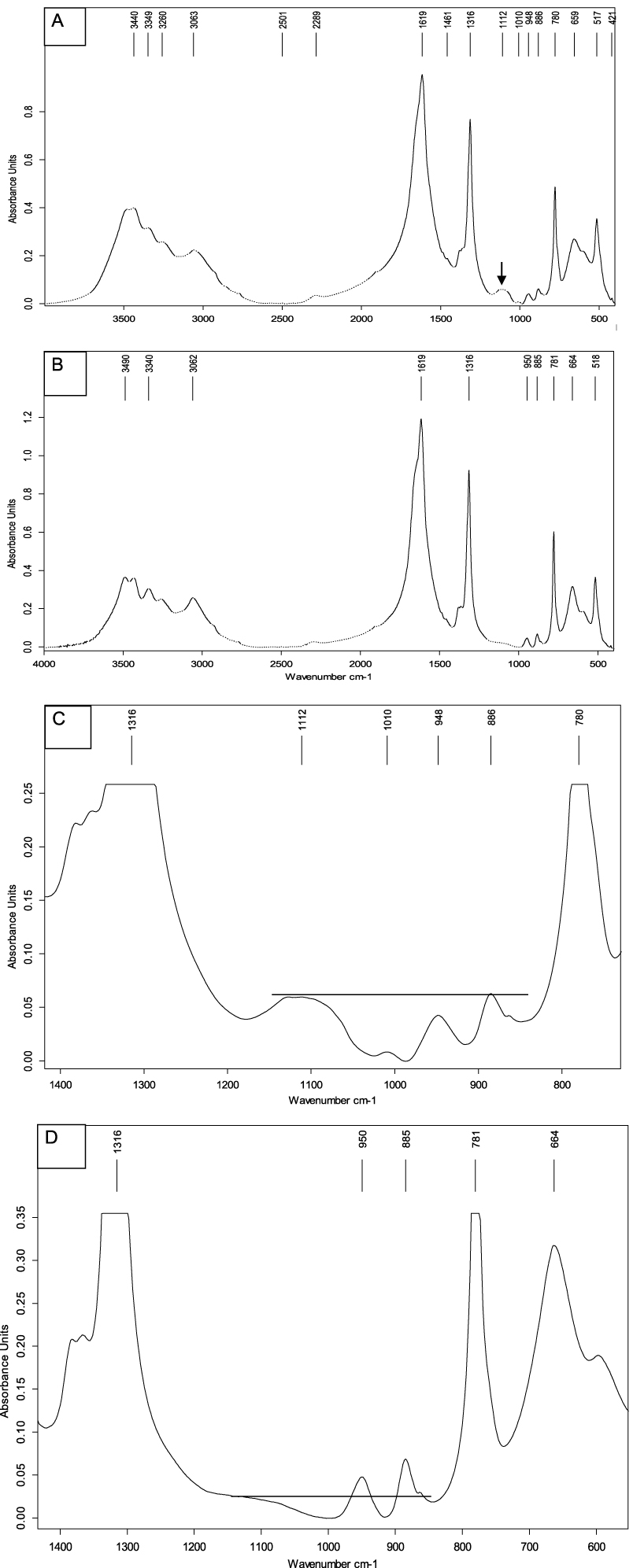
The comparison between type Ia and type Ic stones regarding the ratio of peak heights 1100/949 cm−1 is particularly interesting from a clinical point of view. For type Ia stones, the ratio was >1 in 72.3% of cases, around 1 in 17% of cases and slightly decreased below 1 but greater than 0.7 in only 10.7% of cases. By contrast, in type Ic stones, the ratio was >1 in only 3.6% of cases, about 1 in 7.1% of cases and below 1 in 89.3% of cases. Of note, among type Ic stones with a decreased ratio, 80% of stones had a ratio below 0.7.
3.3. Demographic profile of type Ic stone formers
The mean age (±SD) of the patients was 7.09 ± 3.7 years. The patient’s age ranged from 1 to 17 years among boys with an average age of 7.5 ± 3.8 years and from 1 to 15 years in girls with an average age of 6.45 ± 3.5 years. Figure 5 shows the patient’s age at the first stone episode. Of note, 28.2% of stones were observed before the age of 5, 47.1% in patients aged from 5 to 10 years and 24.7% above the age of 10 years. The mean male to female ratio for the whole series was 1.66.
Distribution of type Ic morphology according to patient’s age.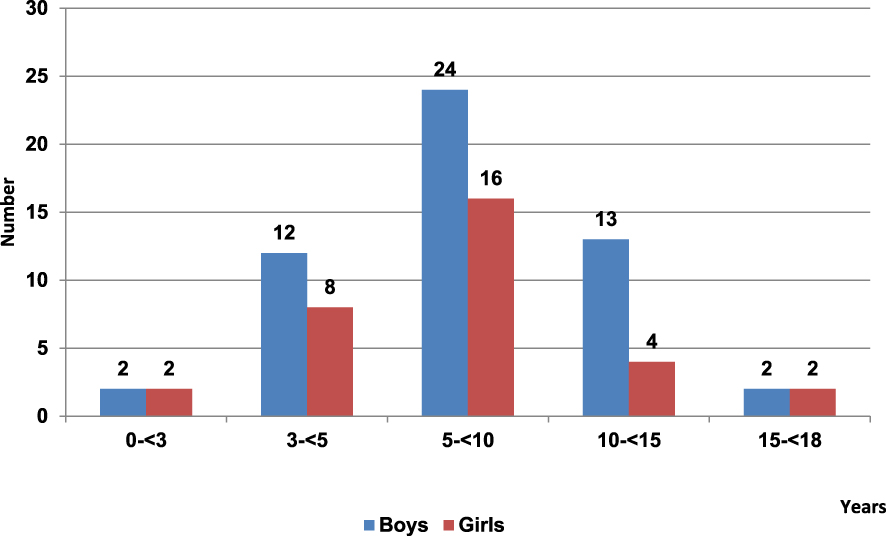
3.4. Crystalluria
Among those children with COM stones of type Ic morphology, 43 patients provided spot urine for examining biochemistry and crystalluria. In all cases, abundant crystalluria mainly composed of typical COM oval crystals with depressed core (Figure 6) was observed. Usually, voluminous aggregates of COM crystals are also observed (Figure 7). The average number of crystals was 151 ± 12 /mm3 and the global crystal volume for COM was 14,632 ± 1225 μm3/mm3 as calculated according to the previously published formula [37]: N × L3 × 0.19 (where N is the number of crystals/mm3 and L is the mean size of the crystals). By contrast, in urine samples that have hyperoxaluria (oxalate concentration above 0.3 mmol/l) for various reasons in the absence of primary hyperoxaluria, the average number of whewellite crystals was 6 /mm3 and the corresponding crystal volume was 350 μm3/mm3 (p < 0.0001). These results, like stone morphology, highly suggested primary hyperoxaluria in this cohort of patients [7, 19, 33, 34, 37, 38, 39].
Oval crystals of whewellite with a depressed center. (A) Polarized light; (B) white light.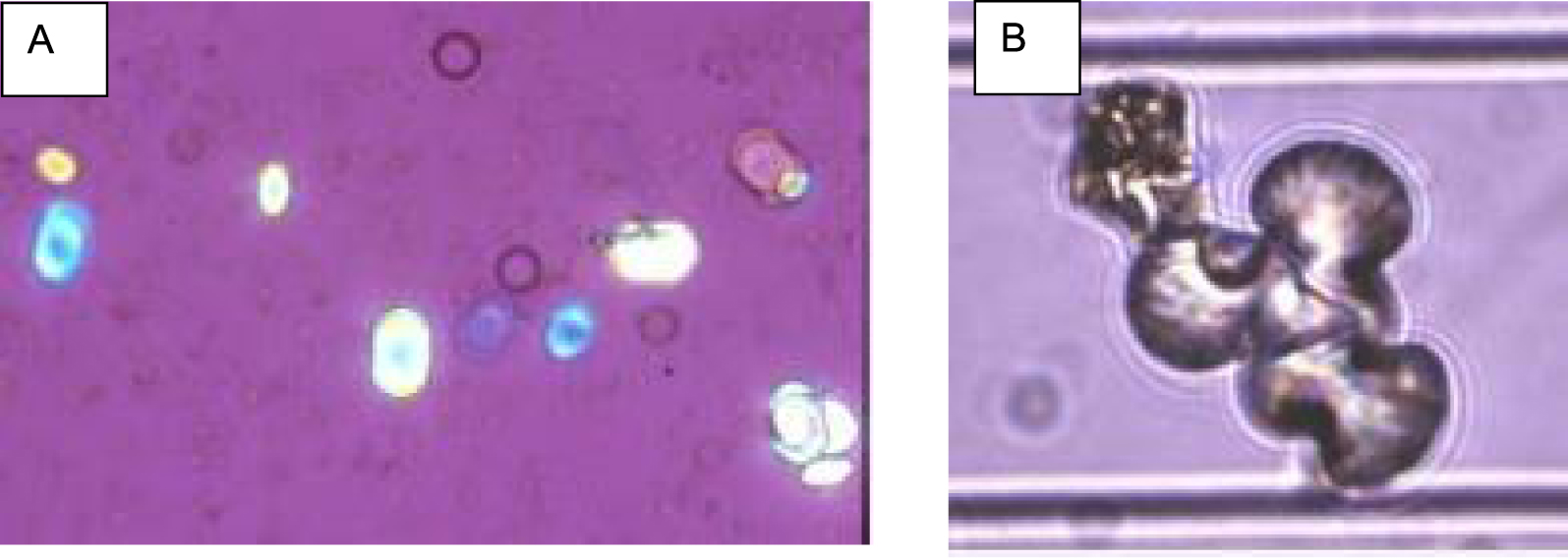
Voluminous aggregates of whewellite crystals. (A) Polarized light; (B) white light.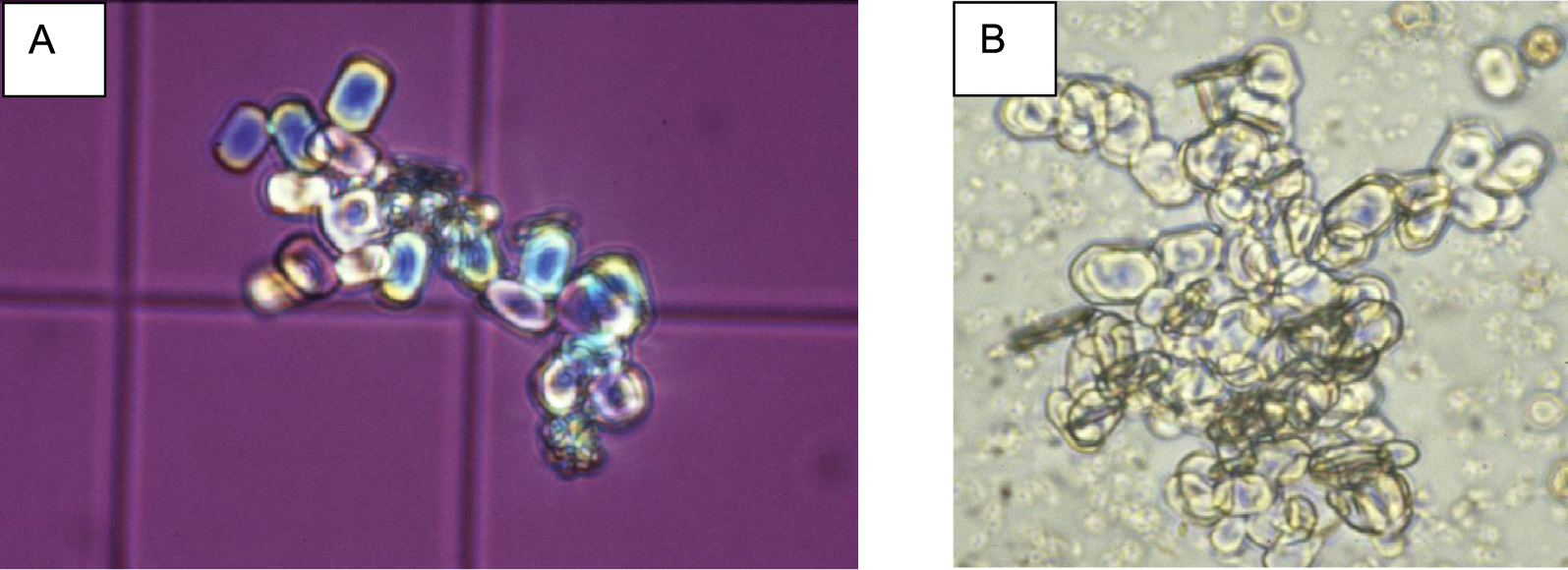
3.5. Biochemical examination of first morning urine samples
The different urinary parameters evaluated in type Ic COM stone formers and in the control group are summarized in Table 2. Of note, no significant difference between boys and girls was found for any parameter, neither in the patients with type Ic COM stone nor in the control group.
Our data showed that hyperoxaluria was the most common abnormality and was coexisting with a dramatically elevated urinary oxalate-to-creatinine ratio in all the children with type Ic stones as previously reported in PH patients [1, 31]. The mean value for oxalate concentration in type Ic COM stone formers was 1.01 ± 0.042 mmol/l (vs 0.136 ± 0.029 mmol/l in the control group, p < 0.0001). All but one urine samples exhibited oxaluria >0.3 mmol/l, i.e. above the threshold value of oxalate for the crystallization risk of calcium oxalate [40]. Of note, the oxalate to creatinine ratio was high (0.1 mmol/mmol) for this patient. In the control group, 83 samples (90.2%) had a concentration level of oxalate below 0.3 mmol/l (p < 0.0001). Very high level of the oxalate/creatinine ratio was observed in type Ic COM stone formers: 0.26 ± 0.012 vs 0.03 ± 0.010 in the control group, p < 0.0001 suggesting the diagnosis of PH in the absence of other cause of hyperoxaluria. Hypocitraturia was coexisting in 66.7% of the samples and low calcium concentration <1 mmol/l was observed in 56.7% of the cases. These findings were similar in the control group: 64.1% of the patients had citrate concentration in urine below 1 mmol/l and 55 subjects (59.8%) had urine calcium concentration below 1 mmol/l. By contrast, only 4.8% of urine samples from type Ic COM stone formers presented with hypomagnesuria (Mg concentration < 2 mmol/l) while that criterion was observed in 44.6% of the subjects in the control group. Of note, proteinuria was slightly increased in type Ic COM stone formers (0.24 ± 0.04 g/l vs 0.15 ± 0.03 g/l in CG, p = 0.002), perhaps as a marker of some degree of kidney damage.
Urinary biochemical evaluation in first-morning urine samples of children with type Ic COM stones and in the control group
| Type Ic COM stone formers | Control group | |||||
|---|---|---|---|---|---|---|
| Number | 43 | 92 | ||||
| Mean ± SEM | Median | Mean ± SEM | Median | P value Ic stones vs CG | ||
| Age | 7.09 ± 0.74 | 6.0 | 8.36 ± 0.51 | 8.5 | NS | |
| pH | 6.07 ± 0.11 | 5.90 | 6.04 ± 0.08 | 5.93 | NS | |
| Specific gravity | 1012.8 ± 1.0 | 1013 | 1010.6 ± 0.7 | 1010 | NS | |
| Creatinine (mmol/l) | 5.11 ± 0.62 | 4.25 | 4.78 ± 0.43 | 3.74 | NS | |
| Calcium (mmol/l) | 1.03 ± 0.29 | 0.73 | 1.63 ± 0.2 | 0.68 | NS | |
| Phosphate (mmol/l) | 14.6 ± 1.5 | 11.9 | 13.1 ± 1.0 | 10.9 | NS | |
| Sodium (mmol/l) | 63.9 ± 3.6 | 53 | 32.2 ± 2.4 | 32 | <0.0001 | |
| Magnesium (mmol/l) | 4.94 ± 0.46 | 3.94 | 3.03 ± 0.32 | 2.53 | NS | |
| Urate (mmol/l) | 2.06± 0.22 | 1.56 | 1.70 ± 0.15 | 1.22 | 0.02 | |
| Oxalate (mmol/l) | 1.010 ± 0.042 | 0.9 | 0.136 ± 0.029 | 0.1 | <0.0001 | |
| Citrate (mmol/l) | 0.70 ± 0.12 | 0.39 | 0.89 ± 0.08 | 0.57 | NS | |
| Proteins (g/l) | 0.24 ± 0.04 | 0.14 | 0.15 ± 0.03 | 0.09 | 0.002 | |
| Urea (mmol/l) | 205.9 ±18.8 | 190.7 | 186.9 ± 12.8 | 162.0 | NS | |
| Ratio calcium/creatinine (mmol/mmol) | 0.21± 0.06 | 0.17 | 0.42 ± 0.04 | 0.20 | NS | |
| Ratio oxalate/creatinine (mmol/mmol) | 0.27 ± 0.012 | 0.26 | 0.03 ± 0.010 | 0.03 | <0.0001 | |
Figure 8 shows the correlation between urinary oxalate concentration and oxalate/creatinine ratio with a distinct distribution of the two groups.
Relationship between oxalate and oxalate/creatinine ratio in spot morning urines of type Ic stone formers and control groups. Blue circles = control group; red circles = type Ic stone formers.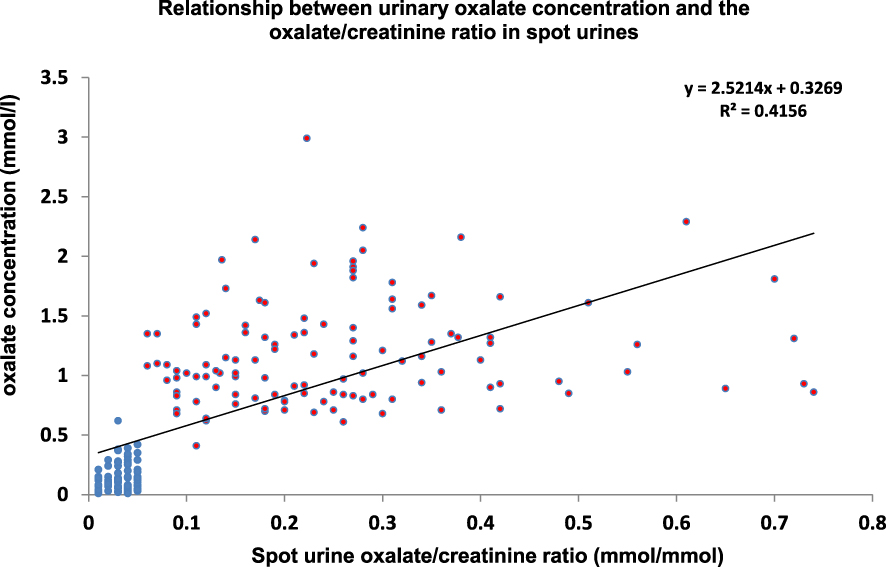
3.6. Influence of age
As seen above, the clinical significance of the oxalate to creatinine ratio is changing with patient’s age. It is also a common finding for other urinary solutes of interest for stone disease like calcium or uric acid. The explanation is that, at birth, tubular immaturity is common and may affect either proximal or distal tubular cells or both, modifying secretion and reabsorption of these substances. It is the reason why the oxalate to creatinine ratio may be high at birth, for example about 0.1 mmol/mmol without any genetic disease. In normal subjects, the ratio should decrease rapidly over time to 0.03 mmol/mmol. If we subdivide our series of patients in age classes, we can observe the change in oxalate to creatinine ratio (Figure 9). Age classes were defined as follows: class 1: <1 year; class 2: 1–2 years; class 3: 3–<5 years; class 4: 5–<12 years; class 5: ⩾12–<18 years. Because class 1 and class 2 included only 2 patients either in the control group or in the type Ic stone group, these age classes were not shown on the figure below. The highest value of the oxalate to creatinine ratio was observed in the age class 3 with a median value at 0.33 for type Ic stone patients and ten times lower at 0.04 for the control group (p < 0.0001). As expected, the ratio decreased with age in both groups, by about 50% in the age class 4 for PH patients, and by about 25% in the control group. Then, the ratio slightly decreased in class 5 for both groups.
Evolution of the oxalate/creatinine ratio (mmol/mmol) according to patient’s age. CG = control group; Ic stones = type Ic COM stone formers; Age classes: 3 = 3–<5 years; 4 = 5–<12 years; 5 = 12–<18 years.
4. Discussion
PH is the most severe lithogenic disease associated with highly recurrent stone formation and gradual deterioration of kidney function. Thus, it is of prime importance to identify early this disease because treatment with vitamin B6 is able to reduce effectively oxalate excretion in about 25% of PH1 patients, which may stop or delay the impairment of kidney function [41, 42, 43]. Moreover, very promising therapy based on siRNA treatments able to reduce the oxalate synthesis by the liver is now available [44, 45]. Actually, diagnostic delay is still responsible for ESRD in at least one third of the patients. Several explanations for such delayed diagnosis may be proposed: non typical course of the disease, common crystalline composition of the stones, inaccessibility to oxalate testing, false results of the dosages as a consequence of inadequate pre-analytical conditions, high cost of genetic studies, and so on. However, almost all of the patients who suffer PH throughout the world had recurrent stones and these stones can be analyzed. Unfortunately, an accurate identification of the crystalline compounds based on physical methods such as infrared or Raman spectroscopy or X-ray diffraction is not sufficient to guide the diagnosis because they only identify COM which is the most common component of urinary stones. Fortunately, the study of stone morphology, although not much used around the world, could be of great help, as shown by this study. It may help the diagnosis instantly and is very inexpensive and not so time-consuming by comparison to other methods such as infrared spectroscopy alone when considering the additional benefit for the etiological orientation of stone disease. Morphological classification of the stones constitutes the preliminary work to the genetic study that will confirm the diagnosis of primary hyperoxaluria, the type of PH (PH1, PH2, and PH3) and the characterization of the genes’ mutations that may be relevant for the medical management of the disease.
Several works emphasized the link between stone morphology and the cause of hyperoxaluria [19, 33, 46]. They highlight the interest of performing a morphological classification of the stones in addition to stone composition. This is in agreement with the results observed in a study involving 124 patients affected with PH1 and where most calculi exhibited a peculiar morphology with COM composition identified by FTIR and scanning electron microscopy (SEM) [34].
In case of inherited lithogenic diseases, like PH, the main clinical interest of morphological analysis of the stones by optical examination is to orient immediately the diagnosis to severe diseases, especially when diagnosis by other methods is not easy to perform.
In our series of 614 stones from children, we found that COM was the main component in 248 cases (40.4%) confirming our previous observations [47] while calcium oxalate dihydrate accounted only for 10.6% of cases. It is well known that hyperoxaluria favors COM (as opposed to calcium oxalate dihydrate) which is favored by hypercalciuria [28, 40]. Thus, our results suggest that hyperoxaluria is more prevalent than hypercalciuria in the formation of urinary calculi in Moroccan children. Of interest is the morphology of stones containing 90% or more of COM (n = 123, i.e., 20% of all stones and 49.6% of stones mainly composed of COM). We found that type Ic stones accounted for 83 cases (13.5% of the series and 67.5% of stones containing at least 90% COM). Two stones had a mixed morphology, Ic + IIa in one case, Ic + IVa in the other. They contained 20% of COD and 12% of carbapatite respectively. It was shown that COM stones with a type Ic morphology are most strongly associated with primary hyperoxaluria [18, 19, 33]. In our series of stones, which today include 614 samples from 419 boys and 195 girls, type Ic morphology was observed in 85 calculi (13.8%). Infrared analysis of these stones revealed a high proportion (89.3%) of stones with a decreased ratio of peak heights 1100/949 cm−1 by comparison to common type Ia stones. From a clinical point of view, the band at 1100 cm−1, which corresponds to organic material deposited on the crystals can be considered as a marker of slow and/or intermittent growth of the whewellite stone. By contrast, when whewellite crystals are abundant and rapidly formed and aggregated, as in primary hyperoxaluria, such organic material is often less incorporated to the crystal aggregates and we observe that the relative intensity of the peak at 1100 cm−1 is then significantly decreased as shown in Figure 4D. In our experience, this finding is in agreement with a rapid crystallization of whewellite due to heavy hyperoxaluria.
Biological data were available for a subset of 43 patients with type Ic stones. All of these patients had a high oxalate to creatinine ratio for their age class. These data suggest that a high percentage of COM stone formers in Moroccan children may have PH. These data seem in agreement with other reports: due to the high degree of consanguinity [48], the prevalence of homozygous primary hyperoxaluria was found significantly higher in North Africa than in the industrialized countries [49].
Moreover, crystalluria of 43 children with type Ic COM stones showed many COM crystals (Whewellite) with an oval or dumb-bell shape, commonly associated with high levels of urine oxalate [50]. These results indicate a very active calcium oxalate crystallization. This is in agreement with other reports [39]. The global crystal volume of COM was very high (14,632 ± 1225 μm3/mm3). Of note, the average number of COM crystals found in our series of patients was below the threshold published by Cochat et al. The explanation is that recommendations published in Nephrology, Dialysis and Transplantation intended to alert the reader on the very suggestive criteria of the disease to encourage them to undertake further investigations [7]. In our experience, about 70% of patients who have urine samples that contain more than 200 COM crystals per mm3 suffer from primary hyperoxaluria. However, according to our data, a threshold of 150 COM crystals per mm3 could be a better value to systematically look for primary hyperoxaluria.
In addition to the abundance of whewellite crystalluria observed in all these urines, the regular biological monitoring of these children has revealed significant abnormalities, particularly hyperoxaluria with a very high oxalate/creatinine ratio for the whole series: mean ± SEM = 0.27 ± 0.012 mmol/mmol, range: 0.1 to 0.7 mmol/mmol, vs 0.03 ± 0.01 mmol/mmol in children who do not suffer from primary hyperoxaluria (p < 0.0001). Thus, we can confirm that type Ic morphology for urinary stones is a very interesting marker to orient easily physicians toward primary hyperoxaluria, which still need further analysis to be confirmed. Of note, among our 85 children with type Ic stones, 14 have had a genetic analysis. In all cases, mutations of the gene AGXT responsible for PH1 have been identified.
We have also noted low levels of urinary calcium in our series. This finding has been reported by others authors, specifying this decrease in PH1 and PH2 and not in PH3 where the level of urinary calcium may vary and in some instances is quite high [8, 51]. Among the other parameters studied, magnesium measurement was performed in our study because of its protective role against urolithiasis. We found very few cases of hypomagnesuria by comparison with the control group (4.8% vs 44.6%, p < 0.001). This could be explained by conservative treatment of these children based on a supplementation with pyridoxine and magnesium (MAGNE-B6®) [43]. In contrast, hypocitraturia as a lithogenic risk factor was observed in our series with a significant proportion (72.1%) whereas citrate is known to prevent crystallization of calcium salts [17]. Our observation of hypocitraturia in paediatric stone-formers has previously been described by several authors in children stone formers [52, 53, 54]. Despite the low proportion of urinary calcium above 3.8 mmol/l, the high occurrence of hypocitraturia, although similar to that observed in the control group, could be an argument for supplementation with potassium and/or magnesium citrate.
Regarding calcium oxalate crystal phase, a controversy has been raised by some authors who found COD crystals in stones from PH patients [55]. This finding is not surprising because crystalline phases of calcium oxalate first depend on the calcium to oxalate ratio and may be influenced by various other urine parameters such as ionic strength and the citrate content for example. Contrary to the claims of these authors, whewellite found in stones from PH patients does not come from a crystalline conversion of weddellite, which is a common finding in the case of stones related to dietary conditions. In the case of PH, COM crystals are directly formed from the high content of oxalate in urine as previously reported [34, 40] and confirmed by the crystalluria studies of the present work. All the patients who present with PH have COM crystals in their urine. Moreover, COM is often the unique crystal phase observed in such patients. However, in PH patients who have, in addition to hyperoxaluria, normal or increased urinary calcium, sometimes COD crystals may be observed in association with COM.
This study has at least three limitations: first, genetic diagnosis was only available for a small number of patients. Second, only half of the patients with a type Ic stone provided a urine sample for crystalluria study and biochemical evaluation. Third, our control group is not perfect because only a small number of urine samples from other types I stone formers was available for biochemical comparisons. Nevertheless, all samples from patients with type Ic stones show heavy COM crystalluria, heavy hyperoxaluria and very high oxalate to creatinine ratio, all markers of primary hyperoxaluria.
5. Conclusion
This study confirms the clinical interest of morphological classification associated with the identification of crystalline components by physico-chemical methods. It highlights correlation between the type Ic morphology of COM stones in Moroccan stone children, the presence of COM crystalluria, the high concentration of oxalate in urine as well as the high values of the oxalate/creatinine ratio usually accepted as a strong marker of primary hyperoxaluria in the absence of bowel diseases. Because measurement of oxalate in biological fluids is not easily available and due to its scarcity, primary hyperoxaluria is often misdiagnosed or diagnosis is delayed, leading to an impairment of kidney function. Nevertheless, early diagnosis is crucial, and can be implemented by a morpho-constitutional analysis of the first calculus identified in the child. Type Ic morphology is a very useful tool for the detection of the disease while stone composition is not suggestive.
Genetic diagnosis, now more easily available, is mandatory to identify potential mutations invested in the process and help in the clinical management of the patients.
Conflicts of interest
Authors have no conflict of interest to declare.




 CC-BY 4.0
CC-BY 4.0