1. Introduction
The use of mesoporous materials such as mesoporous silica in the pharmaceutical domain has gained increasing interest over the last few years due to their ability to stabilize the amorphous form, and to enhance dissolution and oral bioavailability of poorly soluble drugs [1, 2, 3]. Even though most of the methods of loading drug molecules onto mesoporous silica implies the use of organic solvent in which the drug is dissolved [4], recently there has been an increasing number of reports of solvent-free drug loading techniques [3]. These latter are preferable in preventing the use of large quantity of solvents, are less time-consuming due to avoiding the solvent removal step and are safer due to the lack of inherent risk of potentially toxic residual solvents. The applied solvent-free loading methods include simple dry blending [5], classical melt-quenching of the drug in the presence of the carrier [6], microwave-assisted heating [7], hot-melt extrusion [8] supercritical carbon dioxide impregnation [9], milling [6] and cryomilling [10]. Among these, several published works and patents [6, 11, 12, 13, 14, 15] endorse the importance of this processing method for obtaining physically stable amorphous dispersions capable of enhancing drug dissolution. However, the investigation of drug chemical stability upon milling in the presence of mesoporous silica has received only very poor attention, and, even when there was interest, differences in chemical stability were attributed to the higher molecular mobility and reactivity inherent in the amorphous state rather than being evaluated in the relation to the excipient and technology.
The aim of this short communication is to test the capability of Syloid 244 FP to promote a change in the physical nature of Praziquantel, a BCS class 2 anti-helminthic drug [16], upon grinding in mild conditions. At the same time, within the scope of this short paper is an investigation upon the identification and quantification of the degradation products of Praziquantel (PZQ) derived from the mechanochemical activation in the presence of Syloid 244 FP. Our previous studies conducted on the same drug testified to a peculiar propensity to chemical degradation when neat ground in the presence of pharmaceutical excipients [17], while, in an unexpected way, PZQ is not prone to degradation when ground by itself [18, 19, 20]. Further studies also attested that degradation products are dependent upon the type of polymer used during milling [21], and these compounds are different from those documented for PZQ in different stressing conditions [22, 23]. To highlight differences due to the grinding process in the presence of this specific mesoporous excipient both physical and chemical characterizations (DSC, PXRD, ESEM, HPLC-UV, UPLC-MS and 1H NMR) are carried out on the simple physical mixture and on mechanochemically activated samples.
2. Material and methods
2.1. Materials
Praziquantel (PZQ, (11b,RS)-2-(Cyclohexylcarbonyl)-1,2,3,6,7,11b-hexahydro-4-H-(pyrazino[2,1-a]isoquinolin-4-one)) of Ph. Eur. grade was kindly donated by Fatro S.p.a. (Bologna, Italy). Syloid 244 FP Silica (amorphous silicon dioxide meeting the requirements of the European Pharmacopeia for Silica, Colloidal Hydrated) was a kind gift from Alltech Italia Srl (Grace Division) (Sedriano, Italy). In accordance with the technical information provided by the manufacturer (see also online Supplementary material), Syloid 244 FP Silica has the following characteristics: average particle size (via Malvern) 3.1 μm, pore volume (N2) 1.6 cm3/g and specific surface area 300 m2/g. Deuterated chloroform (CDCl3, 99.80% with TMS as standard) was obtained from Euriso-Top, St. Aubin, Cedex. Redistilled dichloromethane (technical grade) was used as solvent. Acetonitrile ultra gradient, HPLC grade was used (J. T. Baker). Water used in this study is purified by a Milli-Q system and 10 mM solution of NH4HCO3 (adjusted to pH 10 with ammonia) was made.
2.2. Methods
2.2.1. Preparation of the physical mixtures and coground samples
The physical mixture (MIX 1) was prepared, for comparison purposes, by mixing PZQ in an agate mortar (3 min) with Syloid 244 FP in a 1:1 weight ratio. Due to the very low density of the excipient, a higher dilution would correspond to a jar with very large predominance of Syloid 244 FP particles. Furthermore the high therapeutic dosage of PZQ requires low dilution by excipients. The coground samples were obtained by milling PZQ and Syloid 244 FP in the same 1:1 drug-to-excipient weight ratio as the physical mixture. A vibrational mill Retsch MM400 (Retsch, Germany), zirconium oxide jars (35 mL) each one with 2 milling media (10 mm diameter) were used. The total amount of powder in the jars was 4.50 mL corresponding to a loading volume in each jar of 15.8% respectively, since the total volume of the milling media was 1.04 mL. The frequency of milling was set at 25 Hz and the time of milling was from 15 (coground 1, COG 1) to 30 min (coground 2, COG 2). These operating conditions were defined during preliminary trials and follow previous experiments [16, 17] carried out in the same apparatus using the same active ingredient. After preparation, the samples were stored in glass vials in a desiccator in the dark at room temperature.
2.2.2. Physical characterization of the physical mixtures and coground samples and drug recovery
The changes of PZQ physical properties caused by grinding procedure in the presence of Syloid 244 FP was monitored by DSC, ESEM and PXRD. Each sample (MIX 1, COG 1 and COG 2 and raw materials) was analyzed using a Mettler DSC TA 4000 (Greifensee, Switzerland) connected to a calorimetric cell Mettler DSC20 and using STARe software version 9.30 for data analysis. The samples, each containing about 2 mg of accurately weighed PZQ and placed in 40 μl aluminum perforated crucibles, were heated at a scanning rate of 10 °C/min in a temperature range between 25 and 200 °C, under air atmosphere. The same samples were also analyzed by powder X-ray diffraction using a Bruker AXS D5005 X-ray diffractometer with Ni-filtered Cu K𝛼 radiation (𝜆 = 1.5418 Å). The preparation of the samples consisted in pressing about 20–30 mg of powder over a glass slide to have a flat surface. The data were collected in a 2𝜃 range of 3°–35°, 0.5° every 2 s. Additionally, the samples were observed using a ESEM Quanta 200 under low vacuum (minor/equal to 1.5 Torr), using an electron beam of 20 kV, with 5000× and 20,000× magnifications.
The content of PZQ after milling was assayed by means of a reverse phase HPLC-UV method [24], reported in literature and already applied in previous experiments [17, 18, 19].
2.2.3. UPLC-MS conditions
The UPLC-MS analysis was performed on the Acquity UPLC coupled with SQD mass spectrometer. An Acquity UPLC BEH C18 (50 mm × 2.1 mm inner diameter, 1.7 μm packing diameter) column was used for chromatography. Column temperature was kept at 40 °C. Acetonitrile and 10 mM aqueous solution of NH4HCO3 (adjusted to pH 10 with ammonia) were used as solvents. Injection volume was 2 μL. Flow rate was kept at 0.9 mL⋅min−1. Ionization mode for the mass spectrometer was alternate scan positive and negative electro-spray (ES+/ES−).
2.2.4. NMR spectroscopy
All 1H NMR spectra were recorded on a Bruker Avance 600 spectrometer equipped with a 14 T superconducting magnet and two 5 mm probes. Analyzed samples were dissolved in deuterated chloroform (CDCl3) using tetramethylsilane as reference. Measurements were performed at 300 K using a simple pulse–acquire sequence.
3. Results
PZQ was neat ground with Syloid for 15 and 30 min at 25 Hz and samples were immediately analyzed by DSC. After only 15 min grinding a dramatic reduction in PZQ enthalpy was noticed (Figure 1), passing from about 107 J/g to 6 J/g (COG 1). A completely noncrystalline product was obtained after 30 min (COG 2). Looking at the raw components, DSC curve of the excipient only manifested a slight endothermal desorption at temperatures lower than 90 °C, whereas the DSC trace of PZQ is characterized by the classical melting peak at about 141.94 °C [17], followed by an exothermic degradation with a maximum at about 185 °C. These thermal features can be both recognized in the physical mixture (MIX 1), together with the (barely visible) dehydration endotherm of the excipient. Interestingly, the dramatic reduction of the melting enthalpy could not be attributed to the dilution effect of the excipient, but to the noncrystalline nature of the drug, as testified by the DSC curves of the MIX 1 having the same composition. Also, a pronounced exothermic transition (at about 167–170 °C) was noticed both in the physical mixture and in the coground samples (COG 1 and COG 2), slightly downshifted and remarkably intensified in comparison to that of the pure API.
DSC curves of the coground (COG 1 and COG 2) compared to the physical mixture (MIX 1) and the raw materials.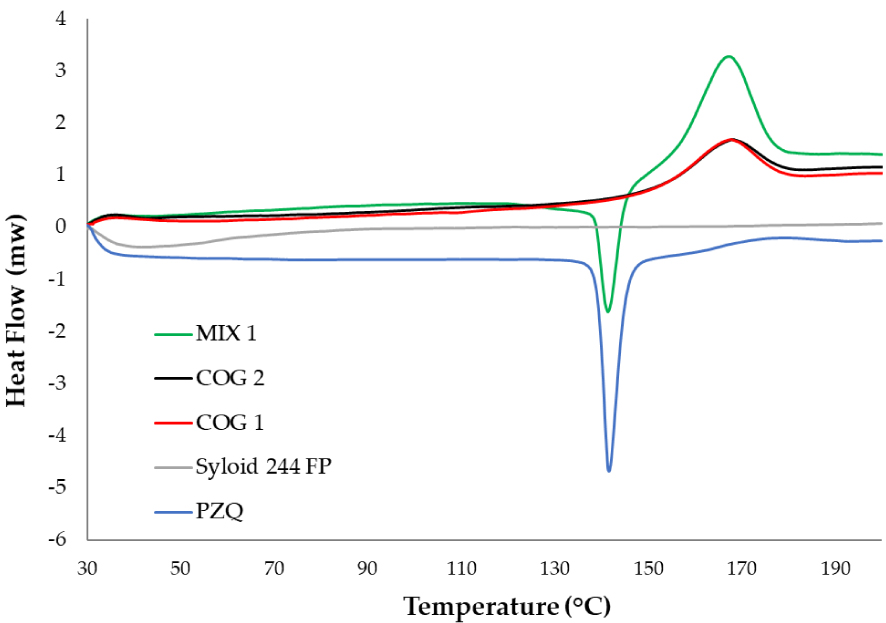
ESEM pictures of the pure excipient, 1:1 physical mixture (MIX 1) including PZQ in the upper frame, 15 min coground (COG 1) and 30 min coground (COG 2) (5000× magnifications).
Regarding the morphology of the samples, Figure 2 reports the images collected by ESEM for the physical mixture, raw Syloid 244 FP and coground samples. In the MIX 1 the raw materials appeared intimately mixed, but both PZQ and the excipient are still visible, each one with its characteristic shape. In particular, raw PZQ particles appeared with the typical elongated prismatic morphology, previously observed [25, 26] (see elliptic frame in Figure 2), while Syloid 244 FP particles showed grains with different particle sizes (round frame of Figure 2). Conversely, both in the 15 min and in the 30 min coground samples, very small filamentous particles of praziquantel were observed between the Syloid 244 FP particles, as well as the loss of the crystalline structure of praziquantel. The ESEM pictures of these noncrystalline dispersions of PZQ/Syloid 244 FP very closely resemble previously reported photomicrographs of amorphous binary coground of PZQ/crospovidone [17] and PZQ/montmorillonite [27]. ESEM analysis thus attested that the drug has completely lost its original crystal habit after only 15 min of grinding, corroborating previous DSC findings.
PXRD pattern of the 15 and 30 min coground (COG 1 and COG 2) compared to the physical mixture (MIX 1) and the raw materials.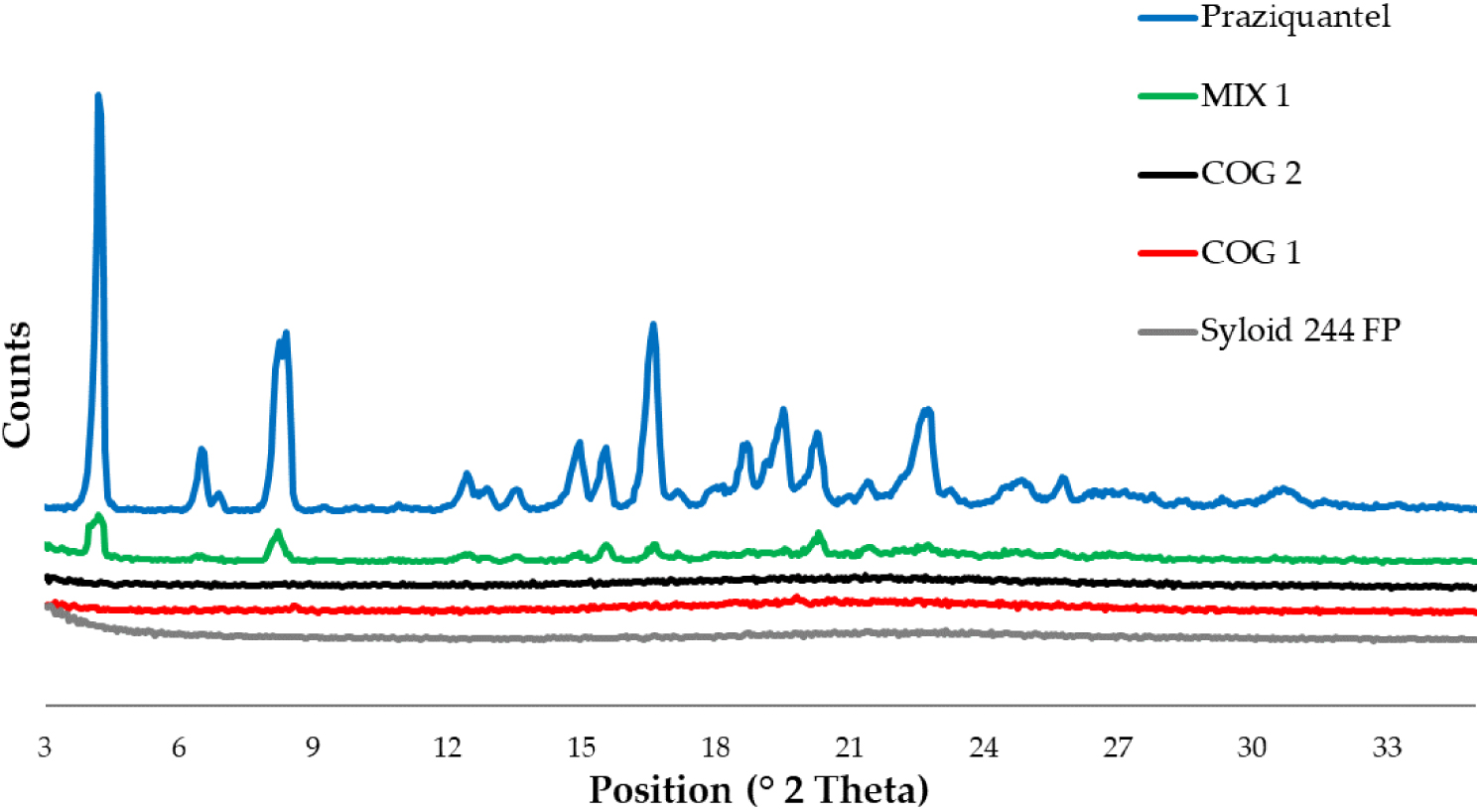
The high de-structuration of PZQ was confirmed by the PXRD analyses (Figure 3). Starting PZQ pattern is characterized by a very intense peak at 4.01 of 2𝜃, followed by reflections at 6.32, 8.00–8.19, 14.73, 15.74, 16.40, 18.48, 19.30, 20.07 and 22.57° of 2𝜃, in accordance with previous findings [19, 20]. Syloid 244 FP showed a halo pattern without any sign of crystallinity and the physical mixture evidenced the main peaks of PZQ in a partially broad spectrum due to the effect of amorphous silica. The coground samples were characterized by a halo pattern with the complete disappearance of the PZQ signals, as well as the typical very intense reflection at 4° of 2𝜃. No new solid forms were formed since no evidence of new peaks was noticed.
These results highlight the extraordinary efficacy of this excipient in promoting a noncrystalline solid by neat grinding. Other pharmaceutical ingredients (e.g. crospovidone, povidone, copovidone) previously co-milled with the same drug in analogous conditions required longer times or more stressful processes (i.e. cryogrinding) [17, 18] to reach a comparable crystallinity degree reduction. In the light of previous experiments where the presence of the excipients in the coground systems favored both lattice de-structuration and chemical degradation of the drug upon milling, we deemed it mandatory to verify drug recovery in this special case where complete noncrystalline state is reached so quickly. Since this excipient is able to transfer the mechanical energy to the active powder in such an efficient manner, we wondered if it would also be responsible for drug degradation.
HPLC results demonstrated that in the case of Syloid 244 FP also the co-milling process at room temperature induced a partial degradation of the API showing an effective reduction in drug recovery. In particular, the COG 1 and COG 2 had a mean PZQ content of 96.12% ± 0.35 and 96.08% ± 0.51 (mean ± S.D., n = 3), respectively, whereas in the simple physical mixture (MIX 1) drug recovery was higher but still evidenced a certain amount of degraded drug (98.48% ± 0.48), also in accordance with the exothermal event recorded at the DSC. It is likely that the particles of this excipient, having well-known abrasive properties and characterized by an intrinsic hardness, act as milling media toward the drug, in addition to the mill balls.
Chromatogram of the starting sample and mass spectrum of the signal at 1.074 min which belongs to the molecule of PZQ.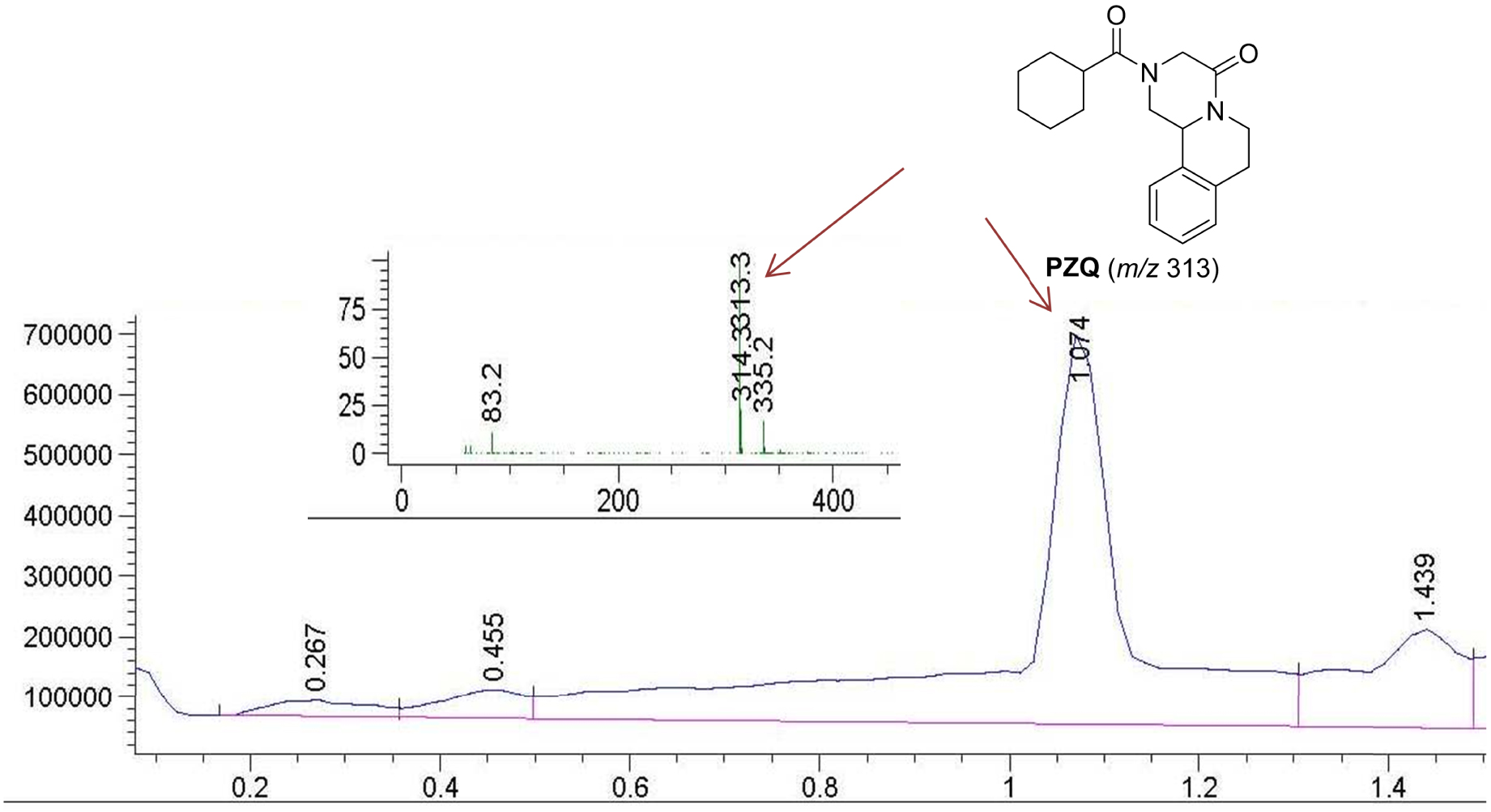
Further analyses were aimed at investigating the degradation products of PZQ derived from the mechanochemical activation with Syloid 244 FP. Analysis of the samples was done on a UPLC-MS system and coupled with NMR analyses. Analysis of the samples was started on the UPLC-MS system and chromatograms were recorded. Mass spectra were obtained also by UPLC-MS system for the compounds present in the samples MIX 1, COG 1 and COG 2. From these spectra, besides that for the molecule of PZQ, only one other new relevant peak was visible (except in sample MIX 1) that was not present in the starting PZQ. By detailed analysis of these chromatograms and the accompanying mass spectra we have come to the conclusion that there is a same new peak present in variable ratios in samples COG 1 and COG 2. In general, the samples COG 1 and COG 2 appear to be alike in having the same additional large peak for one new degradation product (DP). From these data, one can conclude that PZQ undergoes only one degradation pathway. It is very important to say that the starting sample PZQ was not totally pure (Figure 4), so the identified minor signals from PZQ sample were not taken into consideration as new signals in the analysis but as impurities in the starting material.
Sample MIX 1 gave the same analysis as the starting PZQ, so in the physical mixture of PZQ and the Syloid (1:1 by weight) the conditions do not affect or degrade the active pharmaceutical compound. This is confirmed by the chromatogram from the UPLC-MS, where the signal for [M + H]+ at mass 313 is at 1.074 min and matches that of PZQ. The sample COG 1 shows the presence of a new compound DP accompanied by the starting PZQ and this is clearly seen in the chromatogram and also in the MS spectrum of the signal which appeared at 0.998 min with molecular ion m/z 345 (Figure 5) for 32 mass units higher than that for the starting PZQ molecule (Scheme 1). The fragmentation in the mass spectrum of DP indicates the loss of oxygen on getting the fragment with m/z 329 and the base peak with m/z 311 for an additional 18 units lower, indicating the loss of water. The interesting point is that the COG 1 sample seems to be somewhat more degraded than the COG 2 sample.
Chromatogram of the sample COG 1 and mass spectrum of the signal which appeared at 0.998 min.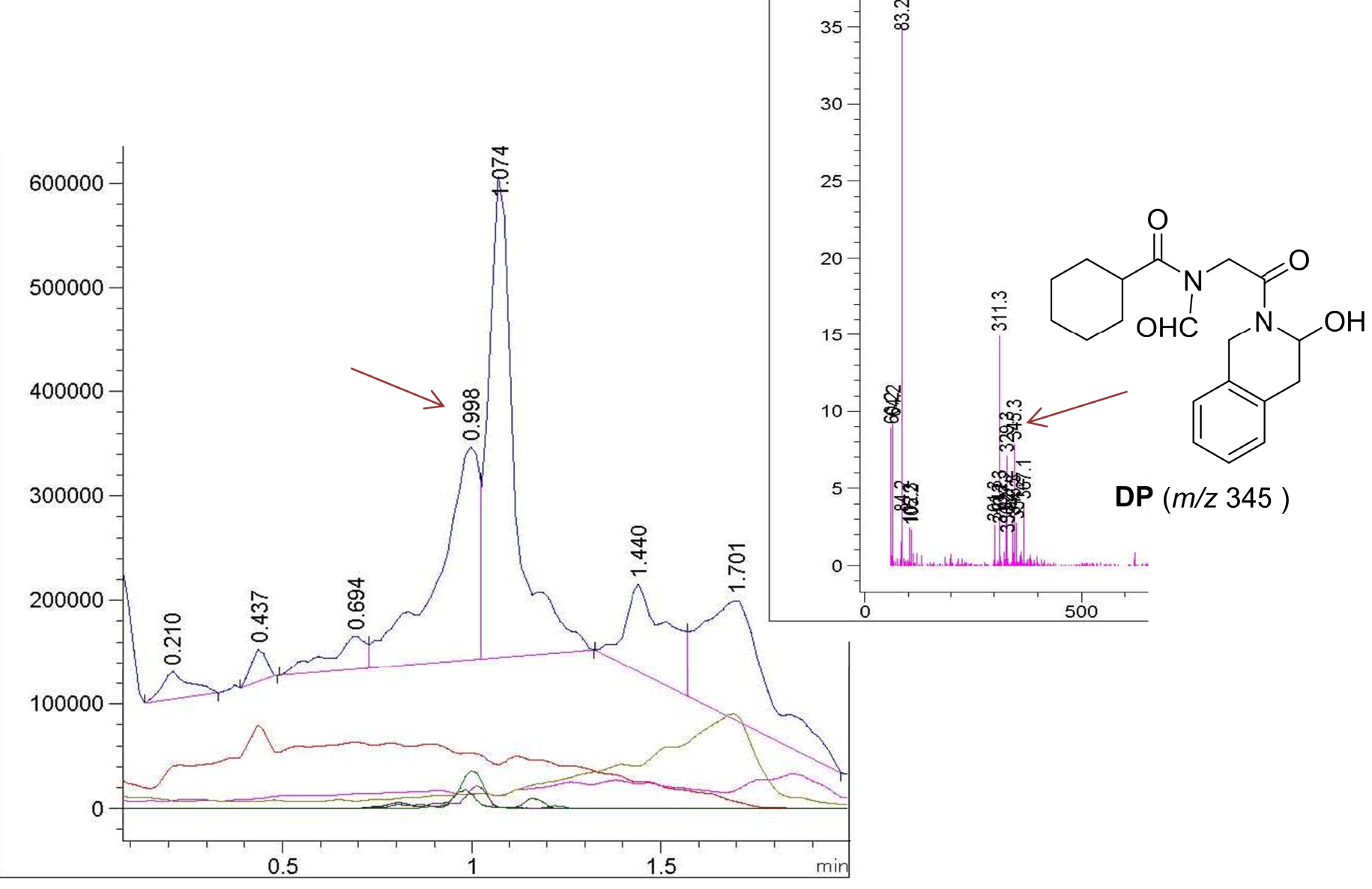
The only detectable degradation product DP (molecular ion m/z 345) after grinding with Syloid in the vibrational mill.
A detailed analysis of the 1H NMR spectra of the samples gave more insight into that newly formed peak, not present in raw PZQ.
Comparison of the parts of 1H NMR spectra of PZQ (a), MIX 1 (b), COG 1 (c) and COG 2 (d).
On the basis of all obtained information and comparison of the crucial parts of the 1H NMR spectra of PZQ (a), MIX 1 (b), COG 1 (c) and COG 2 (d) (Figure 6) the plausible structure was identified as the main impurity in both coground samples COG 1 and COG 2 after grinding with Syloid 244 FP in the vibrational mill as a result of ring opening and subsequent oxidation reactions giving the only transformation structure DP possessing the formyl and hydroxyl groups in its structure (Scheme 1). From the present study and according to the 1H NMR spectrum of the COG 1 sample (Figure 7), the DP product shows a characteristic new signal for the formyl group with the chemical shift at 10.1 ppm and characteristic new signals between 5.5 and 2.5 ppm with very similar coupling constants as for proton signals in the PZQ sample confirming that part of the DP structure remains the same as it originally was in the starting PZQ.
Aliphatic (upper) and aromatic (lower) parts of 1H NMR spectra of COG 1 sample with the highlighted new signals and the proposed structure of the plausible degradation product DP.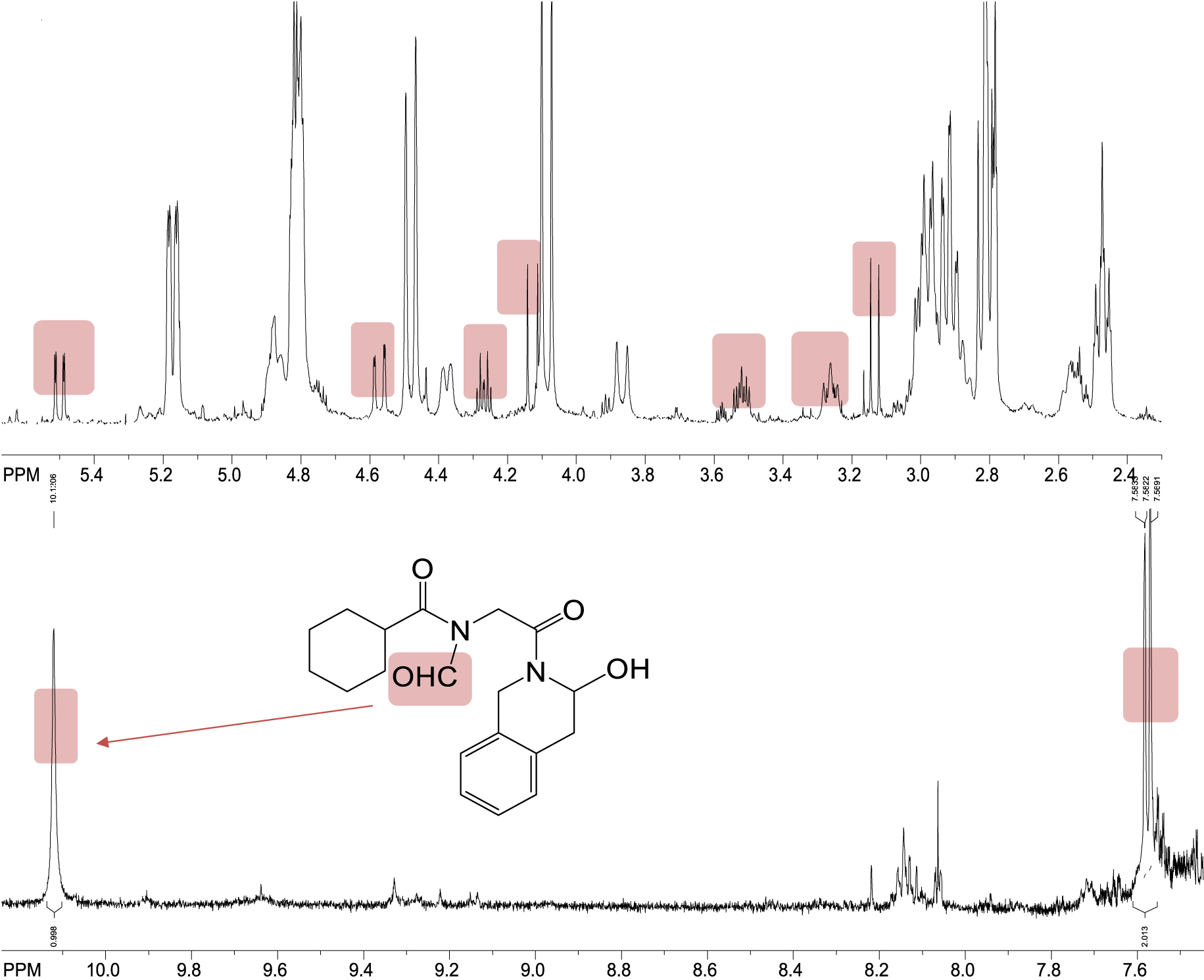
The plausible structure of DP neither belongs to the type of degradation products reported by Čizmić and coworkers [22] nor to those we have seen before in our research [18]. By NMR and MS analyses the different mechanism of degradation with Syloid was found in comparison to povidone and crospovidone [18]. The detected number of new signals in NMR spectra, the integrals, coupling constants and presence of formyl group and the mass and fragmentation in MS spectra clearly show that the proposed structure of DP is the only possible and reasonable one that can satisfy all the data obtained by nuclear magnetic resonance spectroscopy and mass spectrometry experiments. The chemistry leading to such a derivative is definitely plausible; there was a breaking of the single bond between two C atoms of the six-membered ring followed by further oxidation to the formyl and hydroxy functional groups which tend to lose oxygen and water giving very characteristic recognizable signals in the NMR and mass spectrum. It is an important fact that degradation product DP using Syloid is quite different and more functionalized than those previously found with povidone and crospovidone [18] due to the nature of mesoporous silica as excipient and its potential for interactions with the molecules of the drug. In the present case ring opening has happened on only one six-membered ring by cleavage of the C–C bond giving the precondition for further oxidation processes resulting in highly functionalized DP. We can offer an explanation of plausible interaction of PZQ with Syloid 244 FP originating this special degradation process under the experimental conditions presented. As is known, the silica surface is covered by silanol groups that represent places of interaction and absorption, able to create both weak or covalent interactions in contact with functionalized substrate that are fundamental to immobilizing the drug. The H-bond donor nature of the silanol group can favor the formation of links when in contact with H-bond acceptor atoms. In this particular case, the H-bonds between two silanol groups and two carbonyl groups of PZQ probably affect the tension of exact ring thereby generating the driving force for the specific transformation to the specific DP. One can conclude that mesoporous silica affects the breaking of the stronger C–C bond than povidone or crospovidone affecting the C–N bond cleavage [18].
4. Conclusions
Syloid 244 FP has demonstrated an uncommon ability of promoting a change in physical state upon grinding toward Praziquantel, obtaining its almost complete noncrystalline state already after 15 min of process, as confirmed by PXRD, DSC and ESEM analyses. Alongside the de-structuring of the crystalline latex, however, a reduced drug recovery could be noted already after 15 min of grinding. In-depth analyses (UPLC to NMR) made it possible to recognize the appearance of a specific degradation product (DP, Scheme 1). The same degradation product is found even at longer grinding times (30′). This article therefore confirms the susceptibility to degradation of Praziquantel when neat co-milled with excipients and confirms that this degradation product is specific to the excipient used.



 CC-BY 4.0
CC-BY 4.0