1. Introduction
Cell surface glycoconjugates (glycoproteins, glycolipids, and glycoRNA) are fundamental in cellular communications through carbohydrate–protein interactions [1, 2, 3]. In addition, glycans can recognize each other, thus allowing multivalent intercellular events. These numerously and variably exposed saccharides comprising glycoproteins, glycolipids, and polysaccharides, such as glycosaminoglycans, are responsible for a plethora of biochemical processes, including signaling, immune function, cell adhesion, and pathogen recognition (Figure 1). Not surprisingly, they are intrinsically involved in cancer vaccines for viral and bacterial infections, and they constitute highly efficient targeting elements for gene and drug delivery, particularly as nanoparticles.
Left panel: typical cell surface carbohydrates exposing multiple copies of saccharides [1, 2, 3, 4, 5, 6]. Right panel: glycoproteins, glycolipids, and glycoRNA are involved in a wide range of biochemical functions.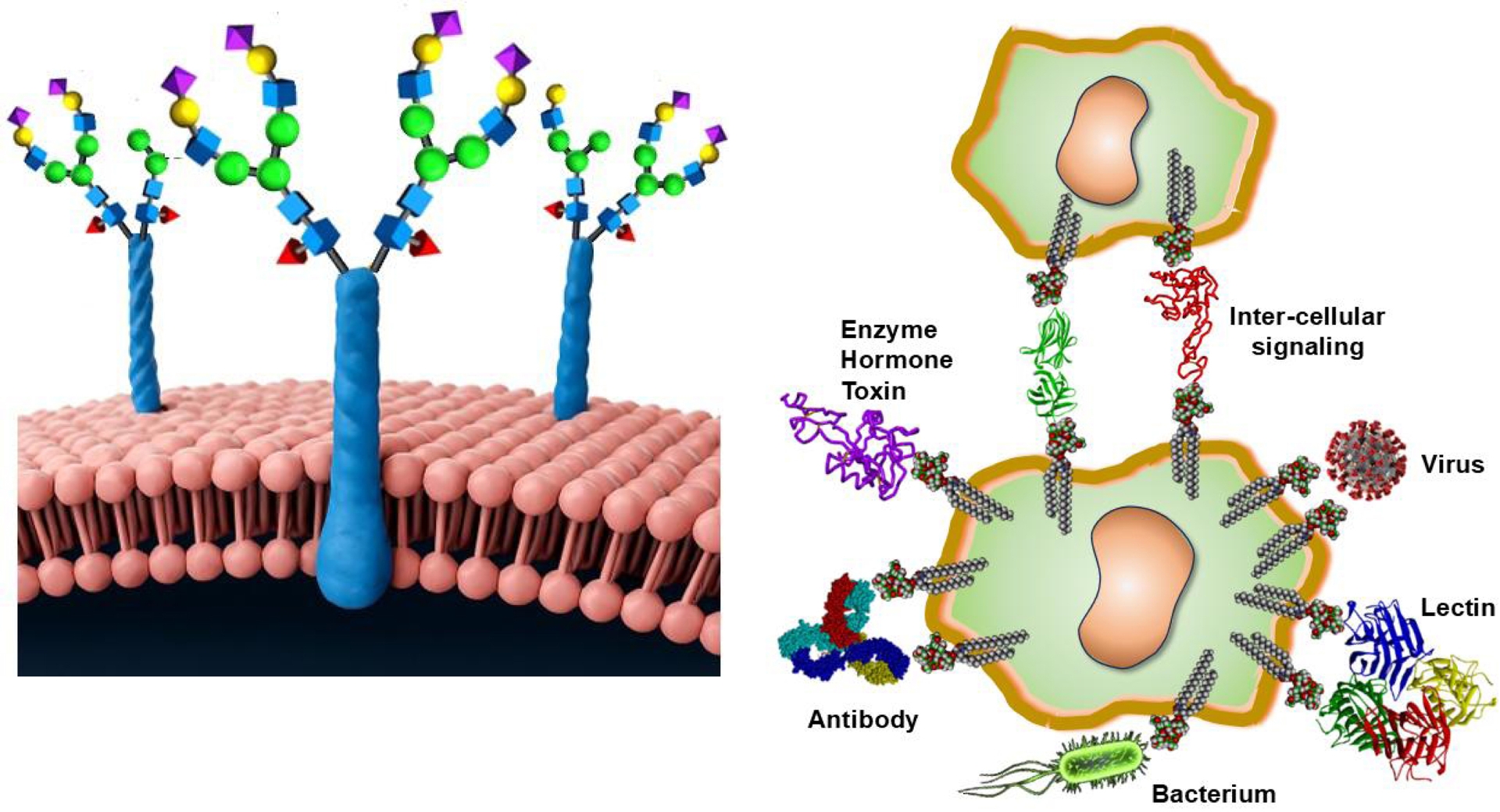
Conspicuously, carbohydrate–protein interactions are multivalent by nature and result in precise and robust binding interactions (avidity, nM range) as opposed to mM affinity when taken individually (affinity) [7, 8]. Therefore, designing artificial carbohydrate ligands capable of imitating cell surface carbohydrates necessitates three major parameters encompassing the following: (1) the exact saccharides (or oligosaccharides) involved, (2) their multivalent presentation, and importantly (3) their relative architectural topologies [9, 10, 11].
For these multiple reasons, we embarked several years ago on the design of glycodendrimers [12, 13, 14, 15, 16, 17, 18], following initial activities in glycopolymers [19, 20, 21, 22]. As opposed to ill-defined polymers, glycodendrimers are a fascinating class of synthetic monodispersed macromolecules that combine the structural precision of dendrimers with the biological functionality of carbohydrates, usually exposed at the dendritic surfaces. The first version of glycopolymers was based on polyacrylamide and resulted in very high molecular weight materials having large polydispersity (Figure 2). These problems were only partly fixed with more modern methods such as the RAFT polymerization method and others [23, 24, 25]. Even though these glycopolymers were tremendously powerful in binding to their cognate receptors and lectins [26, 27], they still lacked the ability to optimize geometrical constraints while minimizing the required number of specific glycosidic residues [28, 29, 30]. Importantly, in our opinion, obtaining precise thermodynamic binding parameters on a per saccharide basis is still an unanswered question. An additional improvement in the field of glycopolymers was achieved with the syntheses of dendronized glycopolymers [31, 32, 33, 34, 35].
The course of evolution of multivalent glyconanoparticles.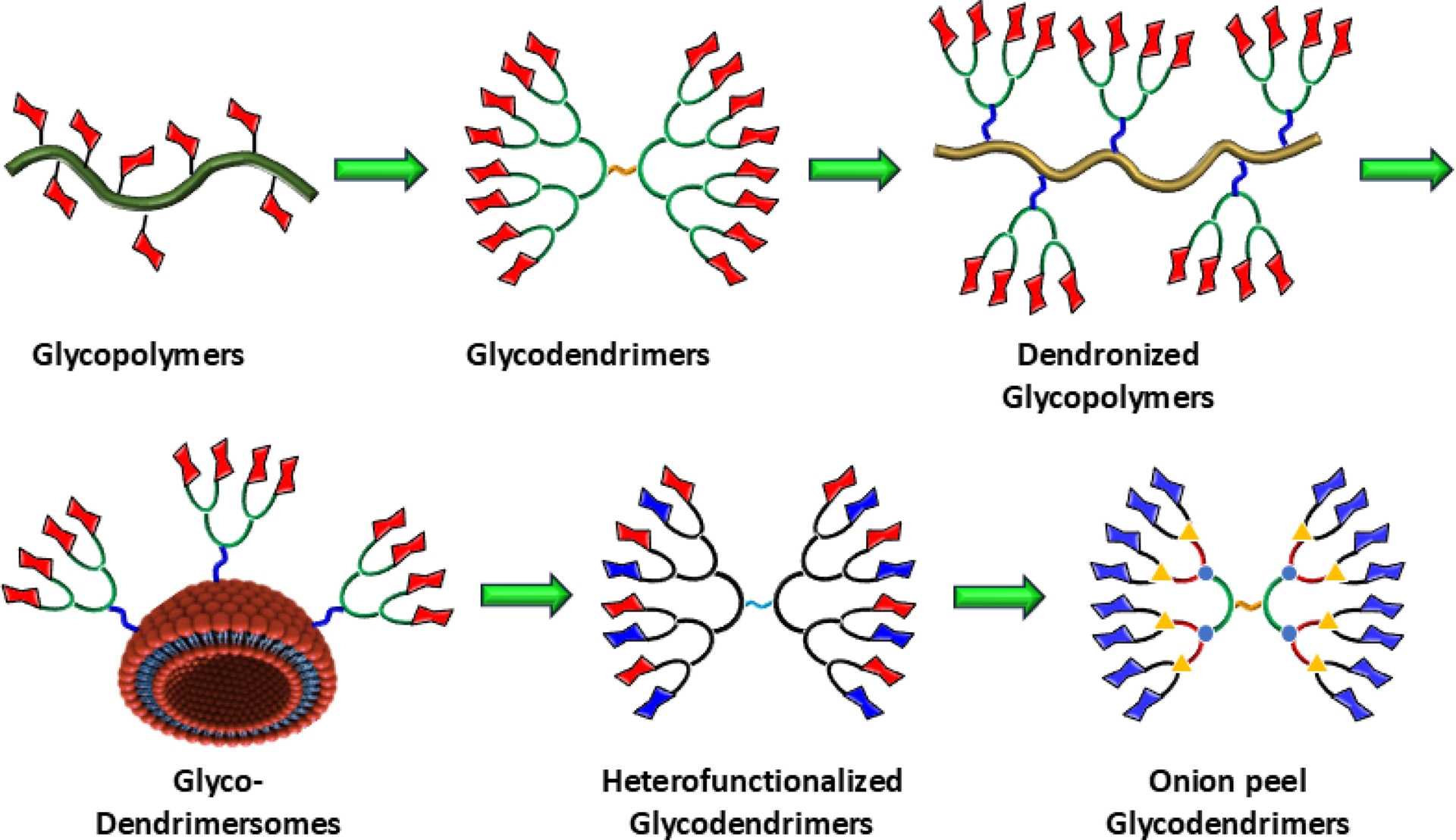
Originally, dendrimers have been designed through rather simplified synthetic strategies involving a multifunctional scaffold (typically two) such as in poly(amidoamine) (PAMAM) dendrimers followed by incremental branching with identical subunits [36]. Their construction initially followed two major paths: divergent and convergent methods. Fortunately, the strategies have rapidly evolved into more sophisticated approaches in order to expedite the synthesis of dendrimers while increasing monodispersity. This was accomplished by accelerated synthetic strategies [31, 37, 38].
As mentioned, these typical convergent, divergent, and accelerated methodologies are commonly created on repetitive synthetic sequences built with identical chemical motifs. Yet, to address the complexity, multivalency, and structural variety of carbohydrate receptors, we embarked on an alternative strategy through which glycodendrimers were built with varied chemical building blocks that offer the required flexibilities and specific targeting abilities [39]. The strategy, designated as “onion peel”, allows the synthesis of “heterolayered” glycodendrimers encompassing a wide range of chemical scaffolds varying in chemical, physical, and biochemical properties [32, 33, 40, 41, 42, 43, 44, 45, 46, 47]. This short review highlights our own work in this area.
2. Synthetic methodologies toward glycodendrimers
Glycodendrimers [48, 49, 50] have been constructed by chemical [38, 51, 52, 53, 54, 55, 56, 57], chemoenzymatic [58, 59], and self-assembling approaches [60]. In one of the later cases, transition metal complexes were first synthesized and appropriately functionalized saccharide dendrons, possessing metal ligands, were allowed to self-assemble through strong coordination/chelation chemistry. In an alternative method, glycolipids, including dendritic glycolipids, are allowed to self-assemble into glycoliposomes or glycodendrimersomes [61, 62].
To further expedite the synthetic developments, highly functionalized scaffolds have also been used to accelerate multivalency [63, 64, 65]. Figure 3 illustrates some of these high valency scaffolds that have been used in the construction of glycodendrimers. For instance, Majoral–Caminade cyclotriphosphazene has been successfully used as the core molecule for the expedient synthesis of highly branched α-D-mannopyranosides [65, 66, 67, 68, 69]. Similarly, suitably functionalized hexaphenylbenzenes were also employed in the rapid synthesis of glycodendrimers having six exposed saccharide residues in the first generation. Analogously, aromatic scaffolds such as calix[n]arenes [70], corannulenes [71], pyrenes, fullerenes, and single-walled carbon nanotubes also constituted high valency building blocks [72]. Interestingly, carbohydrates themselves have also been utilized as multivalent scaffolds. For example, mono-, di- and oligosaccharides represented excellent cases for the accelerated building of chiral glycodendrimers.
Multifunctional building blocks used as core molecules toward accelerated syntheses of glycodendrimers: (A) number in parentheses refers to the potential valency (exposed surface groups) on the chosen scaffolds; (B) interlayer building blocks.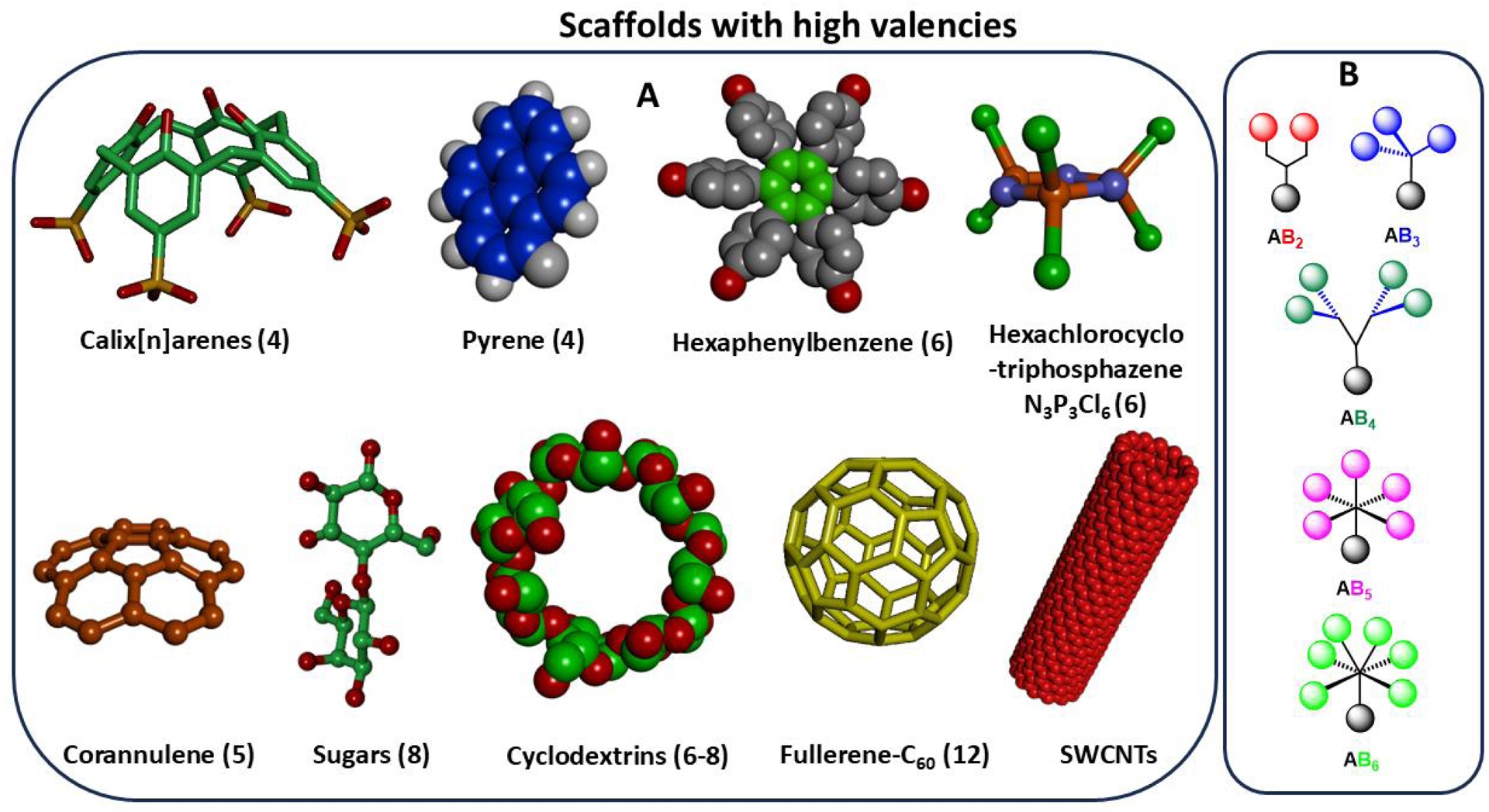
2.1. Choosing high valency scaffolds
As a typical illustration of the high potential and efficiency in the use of multifunctional scaffolds [73], our team has used β-cyclodextrin [74, 75, 76]. Cyclodextrins constitute a family of cyclic oligosaccharides derived from starch by enzymatic treatment. There are three major structures of cyclodextrins (α, β, and γ) forming 6-, 7-, and 8-membered rings through (1-4)-α-D-glucopyranosidic linkages, respectively. For instance, using β-cyclodextrin as a scaffold (1), the hydroxy groups were transformed into peracetylated heptakis iodo β-cyclodextrin (2), which upon treatment with per-O-acetylated 1-thio-α-D-mannose (3) (Cs2CO3, DMF) followed by the removal of O-acetyl protecting groups produced glycodendrimer 4 in high yield (Scheme 1). The expedient synthesis of heptakis α-D-mannopyranosides provided potent glycoclusters capable of rapidly interacting with the homotetrameric leguminous lectin isolated from Canavalia ensiformis (Concanavalin A [ConA]) as demonstrated by their capacity to form stable insoluble cross-linked lattices. In this work, several related families of mannosylated β-cyclodextrin were similarly prepared, some of which had aromatic linkers while others had extended linking arms. The results further demonstrated the general usefulness of carbohydrate-containing aromatic aglycones in the binding of lectins. The use of cyclodextrins as multivalent scaffolds toward the construction of hyperbranched glycodendrimers has been comprehensively detailed in a seminal review by a Spanish group [77, 78].
Using a highly functionalized scaffold such as β-cyclodextrin harboring seven primary hydroxyl groups, glycodendrimers with several copies of carbohydrate ligands (here α-D-mannopyranoside) can be rapidly constructed.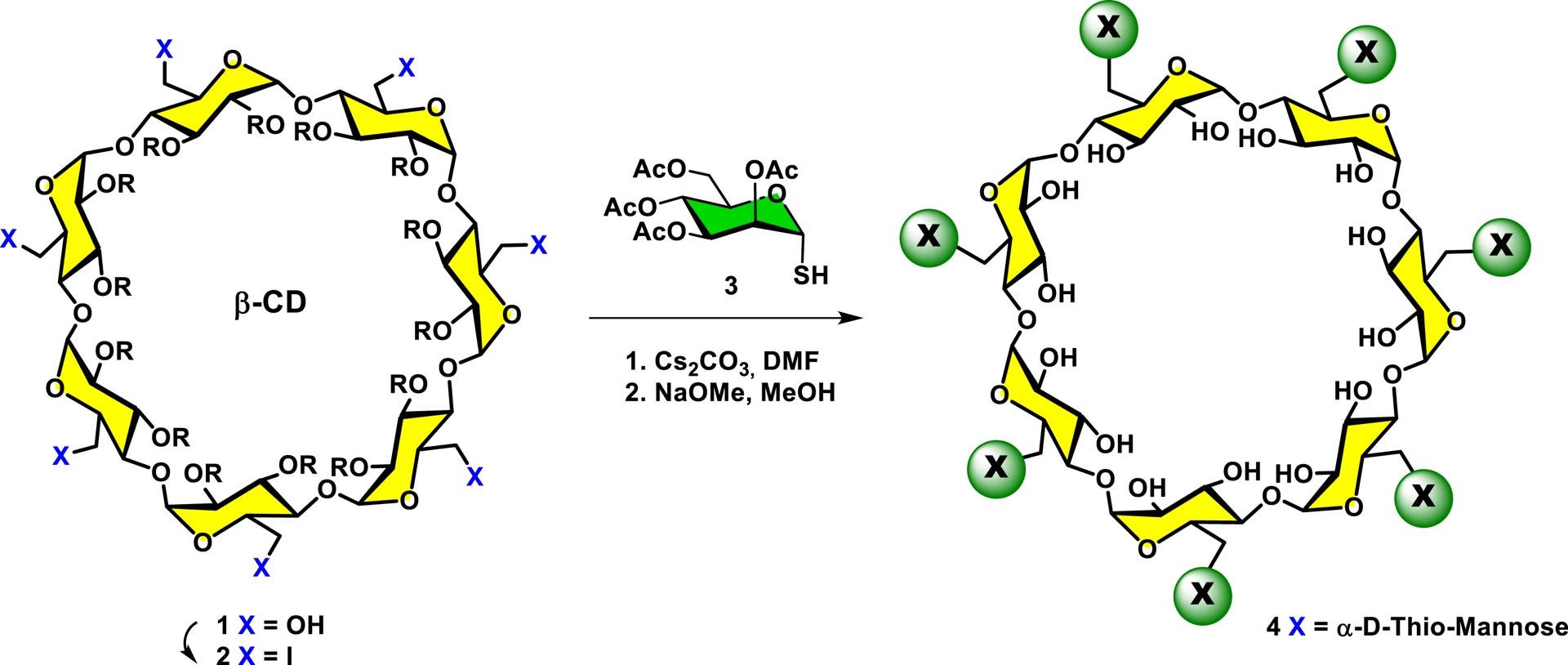
Several additional examples using high valency scaffolds for the construction of glycodendrimers have been reported since their early discovery in 1993 [12] (Figure 3). As mentioned, examples included calix[n]arenes [16, 70, 79], corannulenes [71, 80, 81], fullerenes [82], and hexaphenylbenzene [83]. The scope of using vastly functionalized aromatic scaffolds has been included in a review article by authors [71]. Figure 3 illustrates some of the most significant samples. Our group has been the first to use calix[4]arene as a tetrameric scaffold for the build-up of glycodendrimers. For instance, compound 5 harboring four sialic acid residues, an important carbohydrate in viral infections, particularly involving flu viruses, was synthesized using a neuraminidase resistant α-thio-sialic acid precursor [26, 84] (Figure 4). An additional noteworthy application of glyco-calix[4]arene, exposing lactoside residues (not shown), was identified, and it demonstrated anti-trypanosomal activity comparable to the drug benznidazole in in vitro anti-parasite assays [79]. Notably, analogous constructs encompassing the tumor-associated carbohydrate antigen, Thomsen nouveau (TN; α-D-GalNAc) [85], were found to adsorb on polystyrene microtiter plates through lipophilic tert-butyl tails. This glycodendrimer was found to be ten times more potent than its monomeric counterparts toward the binding of a model leguminous lectin Vicia villosa agglutinin in a competitive enzyme-linked lectin assay against asialoglycoprotein [16, 70].
Typical examples of glycodendrimers built on high-valency-containing scaffolds.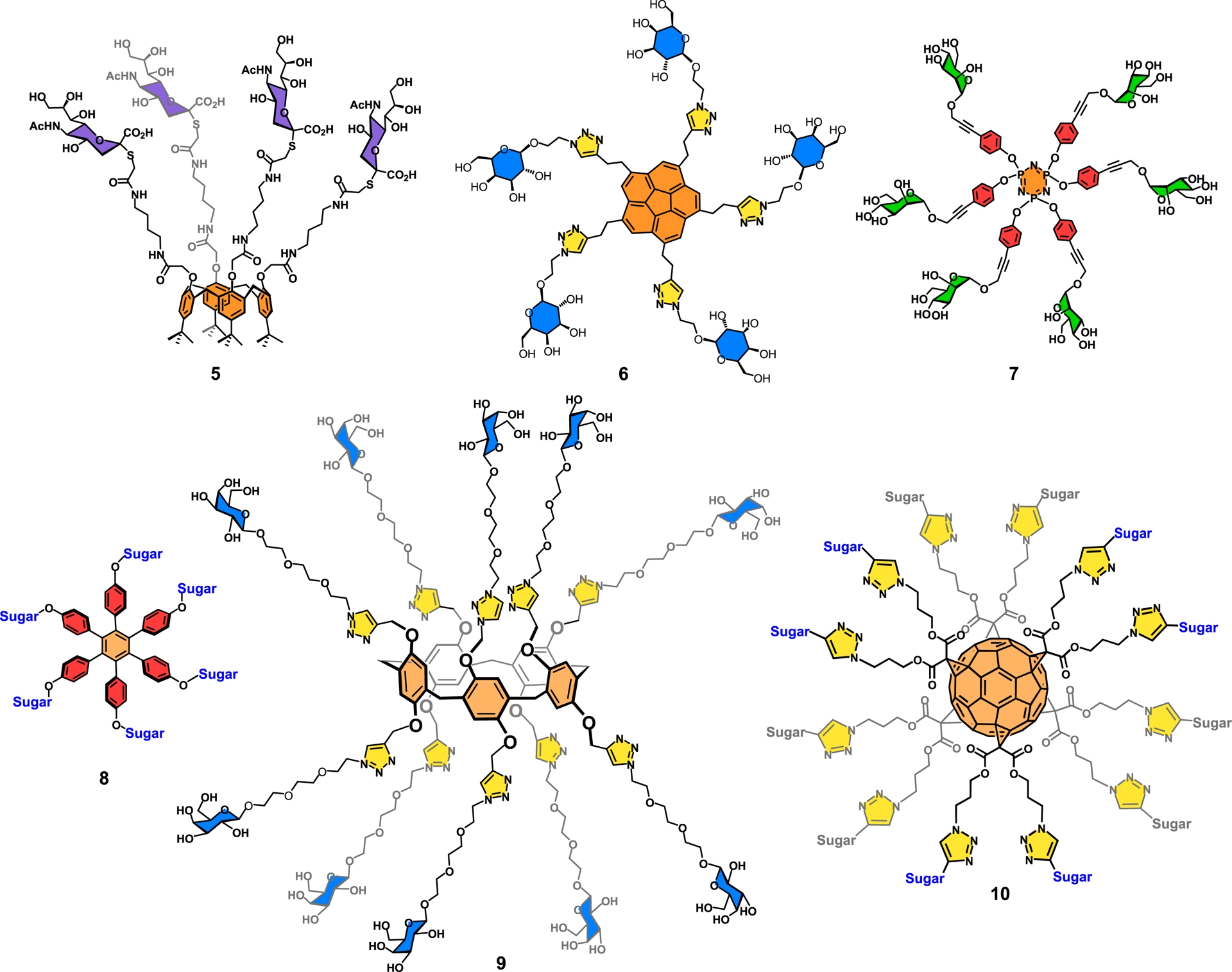
The Siegel group synthesized an interesting aromatic glycodendrimer based on corannulene, which harbored five β-D-galactopyranoside moieties (6) [80] (Figure 4). The pentakis bioconjugate was prepared using the increasingly successful copper catalyzed azide–alkyne cycloaddition reaction (CuAAC; click chemistry). Our own group was deeply involved in the design of a uropathogenic inhibitor based on Escherichia coli FimH infection [86]. Toward this goal, a wide range of α-D-mannopyranoside dendrimers were successfully produced, among which is hexakis mannoside 7 built on a cyclotriphosphazene core [44, 45, 66]. It is to be noted that the cyclotriphosphazene scaffold exposes three of its six appendages on top of the molecules while the other three are in the opposite downward directions [32, 44]. This geometry is in sharp contrast when hexaphenylbenzenes are used as the central building block. A glycodendrimer such as 8 constitutes a case where the carbohydrate residues are planarly exposed [83].
Steadily increasing numbers of biologically valuable carbohydrates presented as surface groups have been aptly exemplified with the use of pillar[5]arenes (9) having ten exposed galactoside residues [87]. The same group pioneered the practice of fullerenes such as 10 and having twelve saccharides on the surface (Figure 3) [82]. Both cases further illustrate the usefulness of click chemistry in the expedient synthesis of glycoclusters [88, 89, 90]. Remarkably, both compounds 9 and 10 were applied as potent ligands in the fight against bacterial infections (Figures 3 and 4).
2.2. Onion-peel strategy
As opposed to conventional dendrimer syntheses that use low valency scaffolds and identical building blocks in repetitive sequences requiring a large number of steps to achieve high surface group densities, the onion-peel strategy starts with highly functionalized scaffolds such as that shown in Figure 2 and including dipentaerythritol 19 (Scheme 3). By using different building blocks at each layer, a much wider chemical space can be reached, thus enabling improved control over chemical, physical, and biochemical properties in addition to providing high surface group density in much fewer steps. Figure 5 presents the onion-peel strategy. Clearly, not only the methodology offers a wide range of chemical possibilities but also it offers a rapid build-up and reduced number of steps to create a higher number of surface groups.
Onion-peel strategy for the construction of high-density surface groups possessing different building blocks at each layer.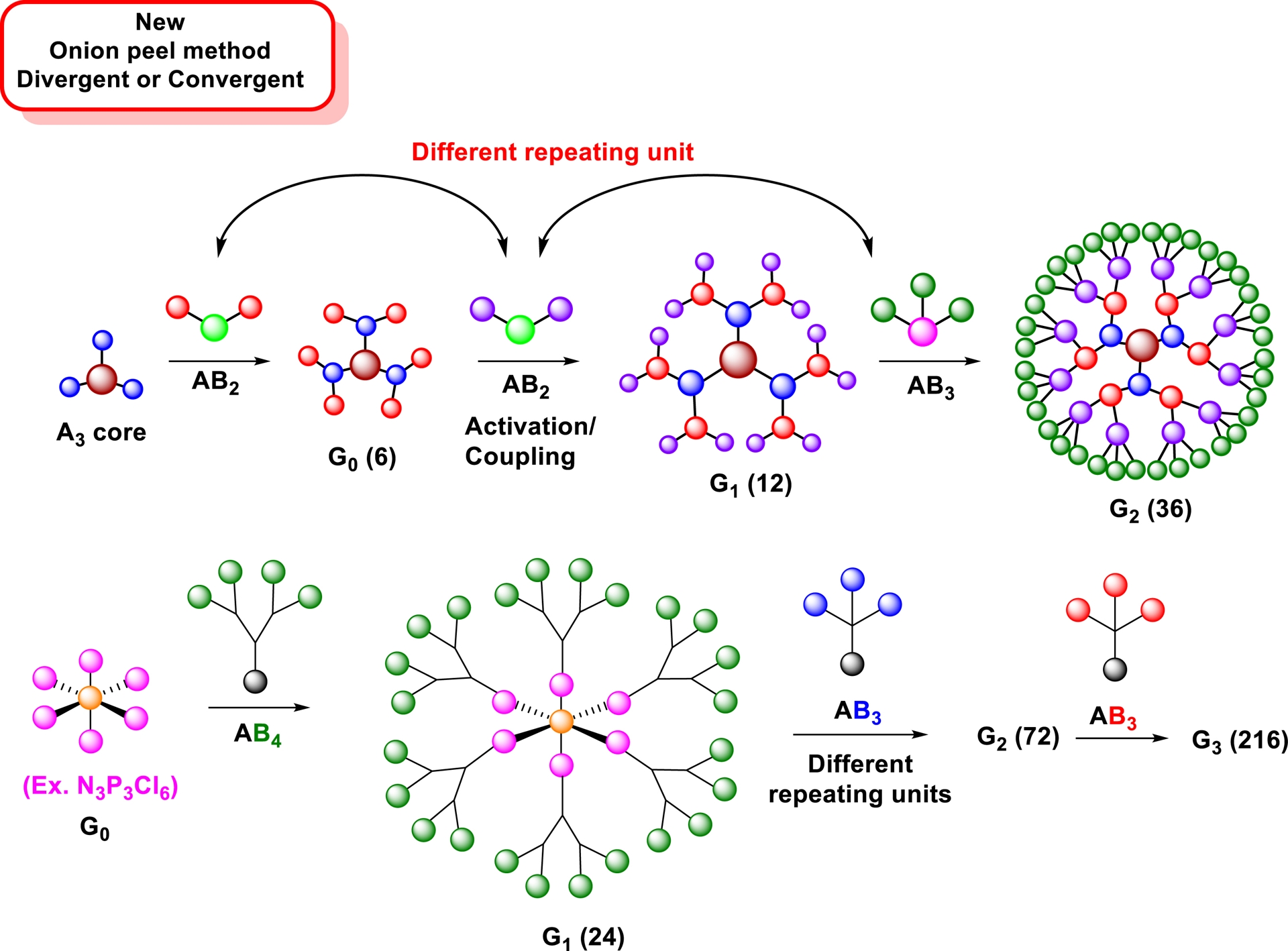
As a typical example, taking trivalent scaffolds (trimesic acid, tris(2-aminoethyl)amine, cyanuric acid) and AB2 type interlayer residues (3,4-, 3,5-dihydroxybenzoic acids), glycodendrimers with 6, 12, and 36 residues can be obtained in generations G0, G1, and G2, respectively [41, 91]. This is in sharp contrast when PAMAM dendrimers are employed, which provide only 4, 8, and 16 surface bearing moieties in identical generations.
Analogously, if a hexachlorocyclotriphosphazene (N3P3Cl6) core is used in combination with alternating AB4 and AB3 interlayer building blocks, glycodendrimers having 24, 72, and 216 residues can be obtained as early as generations G1, G2, and G3, respectively [32, 43, 44, 92].
As an illustration of this approach, the synthesis of a glycodendrimer having 36 LacNAc disaccharides as surface groups was published (Scheme 2). For this purpose, a hexachlorocyclotriphosphazene (N3P3Cl6) core was initially chosen, which was treated with monopropargylated benzene-1,4-diol (hydroquinone) 11 (Cs2CO3, THF, 66 °C, 18 h) to provide the known hexapropargylated cyclotriphosphazene 12 as a crystalline adduct in 83% yield [32, 44, 93]. The next step involved a thiol–yne photocatalyzed addition of 3-mercaptopropanoic acid (13) to form dendrimer 14 having twelve carboxylic acids as surface groups in 76% yield. Trispropargylated pentaerythritol (15) serving as an AB3 wedge was subsequently anchored via carbodiimide-mediated esterification to achieve the construction of hypercore 16 in good yield (72%). This sequence of reactions corresponds to 6 × 2 × 3 (for 36 groups) illustrating an A6 core, followed by an equivalent AB2 interlayer, and finally an AB3 building block. The final step necessitated the use of a LacNAc azide (17), which upon CuAAC coupling produced targeted glycodendrimer 18 in 76% yield. The strategy allowed the use of an unprotected glycoside derivative. Spectroscopic analyses (NMR, MS) were all consistent with the anticipated structure. A low-resolution mass spectral value was obtained by the MALDI-TOF technique ([M+Na]+ adducts) for which compound 18 exhibited a molecular weight of m/z 19774.8 for a calculated value of m/z 19779.9.
Typical example of a fast growing onion-peel glycodendrimer using high-density scaffold (A6) together with different interlayer building blocks (AB2 and AB3) that resulted in a dendrimer having 36 N-acetyllactosamine residues as surface groups [32].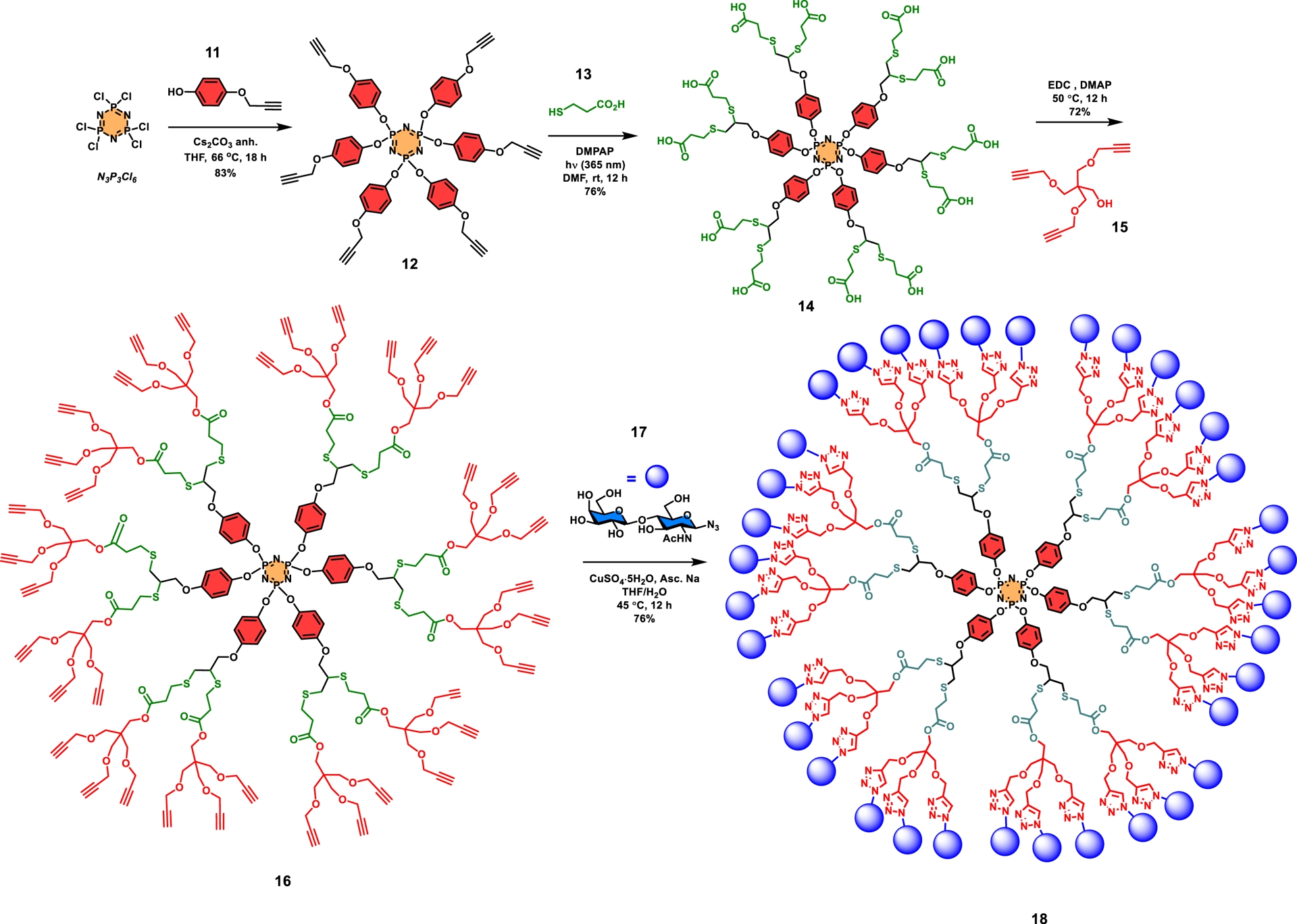
Dipentaerythritol has been underused as a high-density scaffold with its six primary hydroxyl functionalities (A6 core), which when combined with an AB3 interlayer moiety formed a glycodendrimer having 36 surface residues in five consecutive steps. This example further illustrates the possibility of adopting both divergent and convergent approaches using the onion-peel concept [40].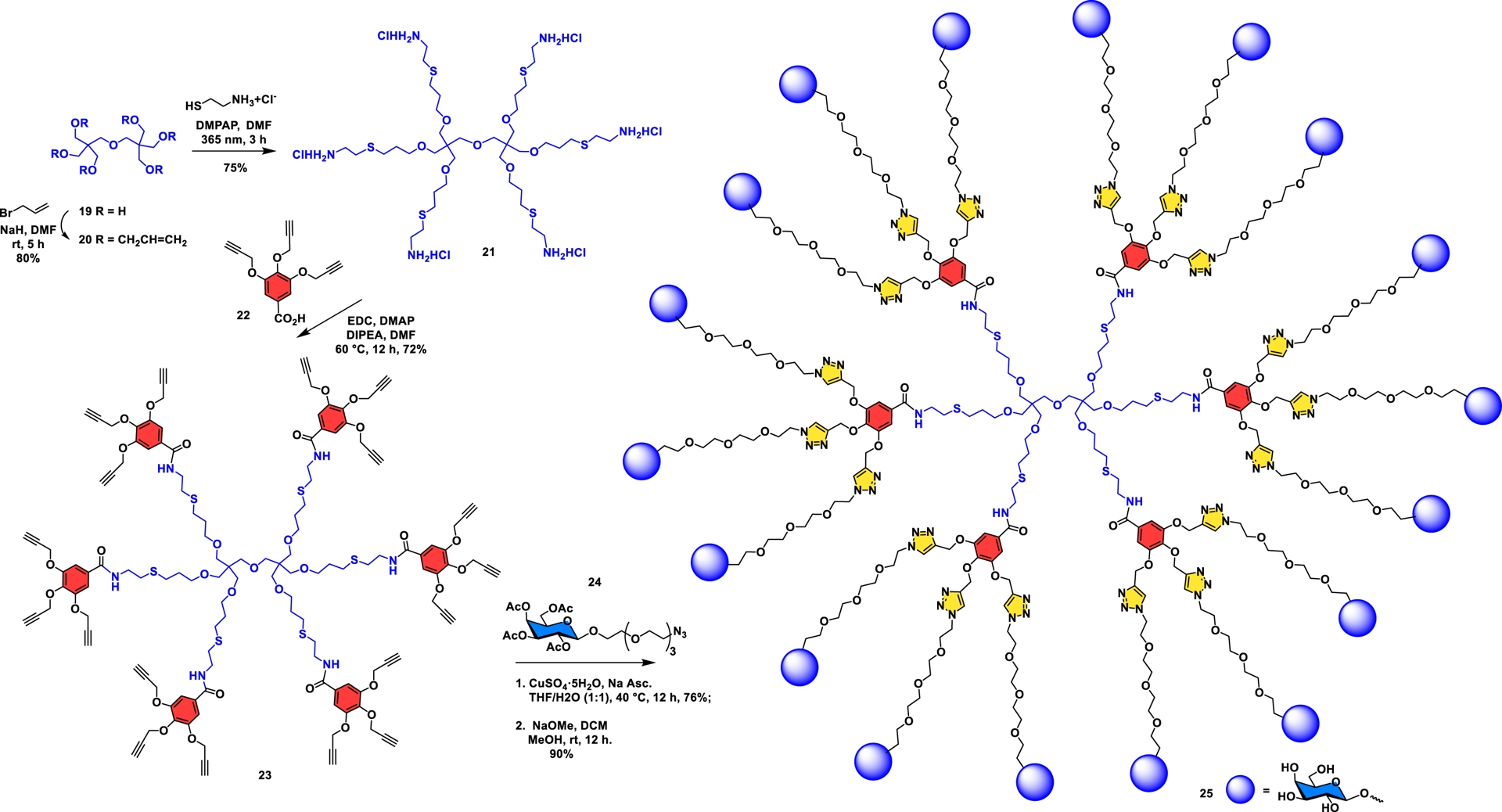
Surface plasmon resonance (SPR) studies were conducted to assess the protein-binding abilities of 18 relative to glycodendrimer congeners possessing fewer LacNAc surface groups. For this purpose, the LacNAc-specific leguminous lectin (ECA) from Erythrina cristagalli was used. In these studies, the lectin was immobilized onto a CM5 gold sensor surface. Solutions with various concentrations of LacNAc-functionalized dendrimers were passed over the surface-bound lectin, and significant interactions were determined for each glycodendrimer and compared to monovalent standard LacNAc (17). Determination of the kinetic parameters relative to the glycodendrimer–lectin interactions were fitted by using a 1:1 Langmuir model. In general, multivalent compounds exhibited high nanomolar affinities with the dimeric ECA. All glycodendrimers exhibited high relative potencies when compared to monomeric 17. In one case, a maximum of 216-fold enhancement in global affinity was observed for the best candidate harboring only 24 LacNAc groups. This work demonstrated once more that highly dense glycodendrimers do not necessarily outperform those with lower carbohydrate residues. Our overall experience in this field indicated that not only the number of carbohydrate residues exposed as surface groups represents beneficial key factors but also their inter-residue accessibility (steric hindrance), the linker’s length, the structure of their aglycones, and obviously the multivalency of the required protein receptors.
In this work, the relative binding affinities of closely related glycodendrimers were evaluated by competitive SPR using the β-D-galactoside specific bacterial lectin from the gram-negative bacteria Pseudomonas aeruginosa. This protein constitutes a virulence factor and is involved in the pathogenesis of the bacteria in cystic fibrosis patients. The SPR experiments clearly demonstrated that all glycodendrimers exhibited much higher binding affinity compared to their corresponding monovalent derivatives due to the “multivalent or glycoside cluster effect”. The incorporation of a tetraethylene glycol linker to the galactoside aglyconic portion allowed a twofold enhancement of the affinity for the lectin by providing better accessibility to the deep binding pocket of the lectin. Galactodendrimer 25 provided one of the best ligands known to date with an IC50 value of 37 nM. This result unambiguously highlights the key role of linkers in interactions with lectin, with a counterbalanced entropic cost due to their flexibility. Additionally, tridimensional distribution of terminal and optimized galactosides crucially contributed to high potencies since a substantial improvement (32-fold) was observed for each ligand in 25 when compared to a monomeric reference.
2.3. Chemical ligation and orthogonal chemistry
This objective may be accomplished through the design and synthesis of heterofunctionalized macromolecules with precise molecular architecture and the integration of multiple bioactive moieties. In our view, recent progress in orthogonal chemoselective ligation methodologies positions these strategies among the most effective for constructing such biomacromolecular systems in a controlled and reproducible manner—a prerequisite for ensuring consistent biological outcomes [94, 95, 96].
Due to their exceptional reactivity and stability, suitably functionalized carbohydrates represent key molecular building blocks in this context. Alongside, glycans and other dendrimer scaffolds can be modified with compatible functional groups such as alkenes, alkynes, thiols, and azides. These approaches, for instance, may enable the development of synthetic multiantigenic vaccines incorporating diverse tumor-associated carbohydrate and peptide antigens [3]. Moreover, the high modularity and broad compatibility of orthogonal ligation techniques with carbohydrates, peptides, and nucleic acids could facilitate the creation of novel biomolecular assemblies with unprecedented biological functions and therapeutic potential.
The use of glycosides as both a high-density core and as interlayer residues has also been a valuable concept in the construction of dendrimers in general and of glycodendrimers in particular [97]. The example that follows combines several principles presented in this review. By including a carbohydrate moiety in the design, a key concept has been added through which chirality can be included, hence providing an additional architectural parameter (Scheme 4) [40].
Accelerated glycodendrimer synthesis through: highly functional core, glucoside as chiral interlayer building blocks, and orthogonal ligation.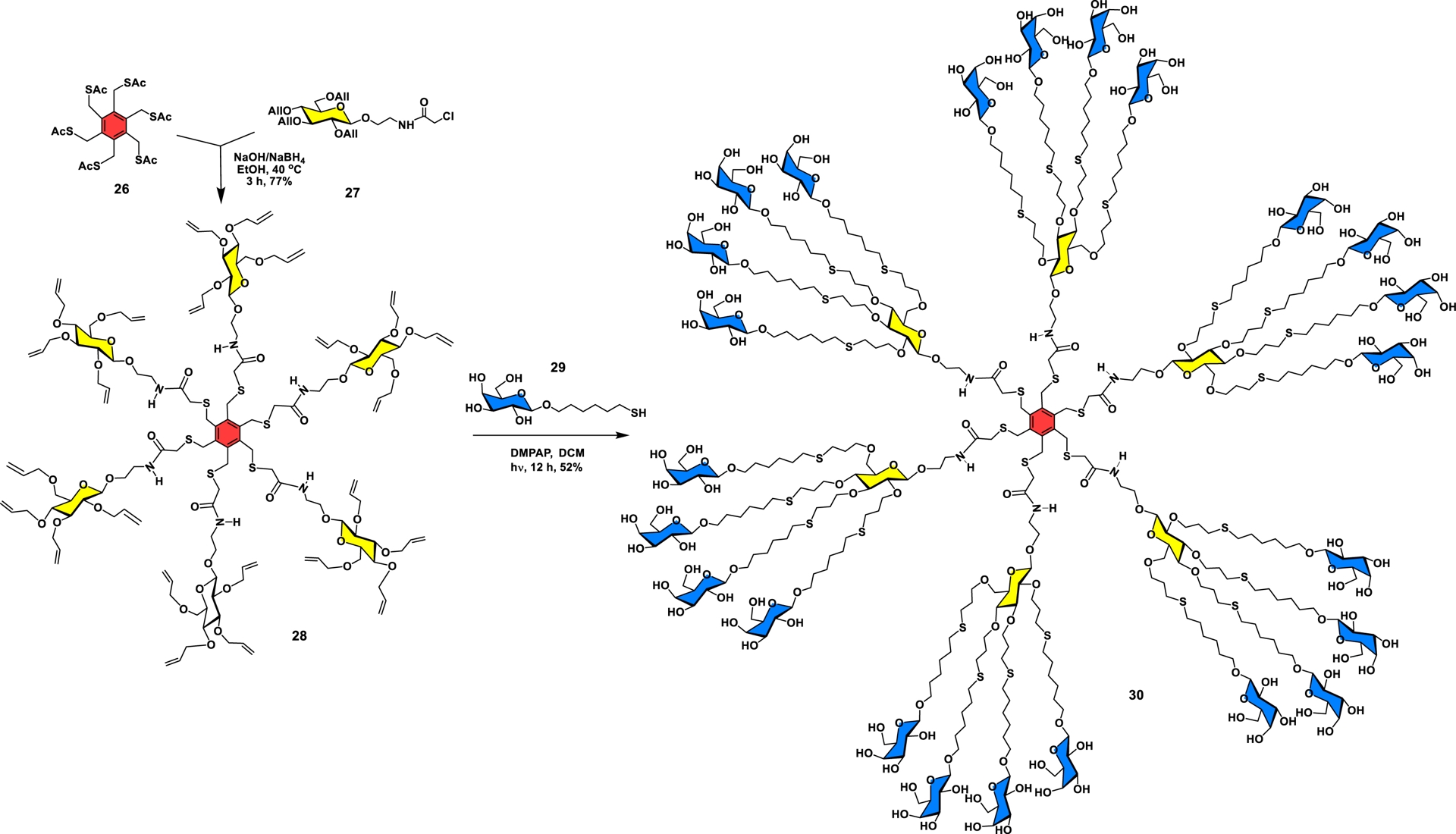
In the present example, an orthogonal coupling strategy was developed by combining thiol–ene and SN2 reactions, which was subsequently applied for the accelerated synthesis of multifunctional dendrimers using carbohydrate as both building blocks and surface groups. In this work, we first established the orthogonality of a thiol–ene against an SN2 reaction. Toward this goal, a glucoside-based AB4 monomer (27), possessing four allyl appendages and a N-(2-(glucosyloxy)ethyl)-2-chloroacetamide group as the aglycone, was used as an interlayer building block (Scheme 4). The ensuing nucleophilic substitution was executed with hexakis(thioacetoxymethyl)benzene (26) (A6 core) under reducing conditions to avoid disulfide formation to provide intermediate 28 in 77% yield [40]. Notably, dendrimer 28 is a chiral entity encompassing 24 surface groups that was next successfully treated with thiogalactoside 29 under photocatalyzed thiol–ene reaction to form glycodendrimer 30 (52%). Using SPR studies, the β-D-galactopyranoside-coated dendrimer exhibited nM binding affinity against the bacterial LecA lectin from P. aeruginosa.
In our endeavors to create glycodendrimers with high biological values, we initially focused our efforts on constructing multivalent architectures with just a single carbohydrate moiety at the surface. This was deemed necessary in order to target only one protein receptor at a time with the highest possible specificity and affinity. However, there are situations for which different carbohydrate residues are necessary to block a precise biological function. This is particularly the case with bacterial lectins expressing two or more carbohydrate-binding proteins such as those found in P. aeruginosa [98]. For this scenario, we synthesized Janus-type glycodendrimers containing for instance both α-L-fucopyranoside on one end and β-D-galactopyranoside at the opposite face (Figure 6). With the next glycodendrimer generation, as mentioned, an onion-peel strategy was employed for a subsequent modulation of biophysical properties.
Janus glycodendrimer with two different saccharide moieties on each side of the “faces” (left) and “onion-peel” glycodendrimer having alternating and different glycoside residues (right).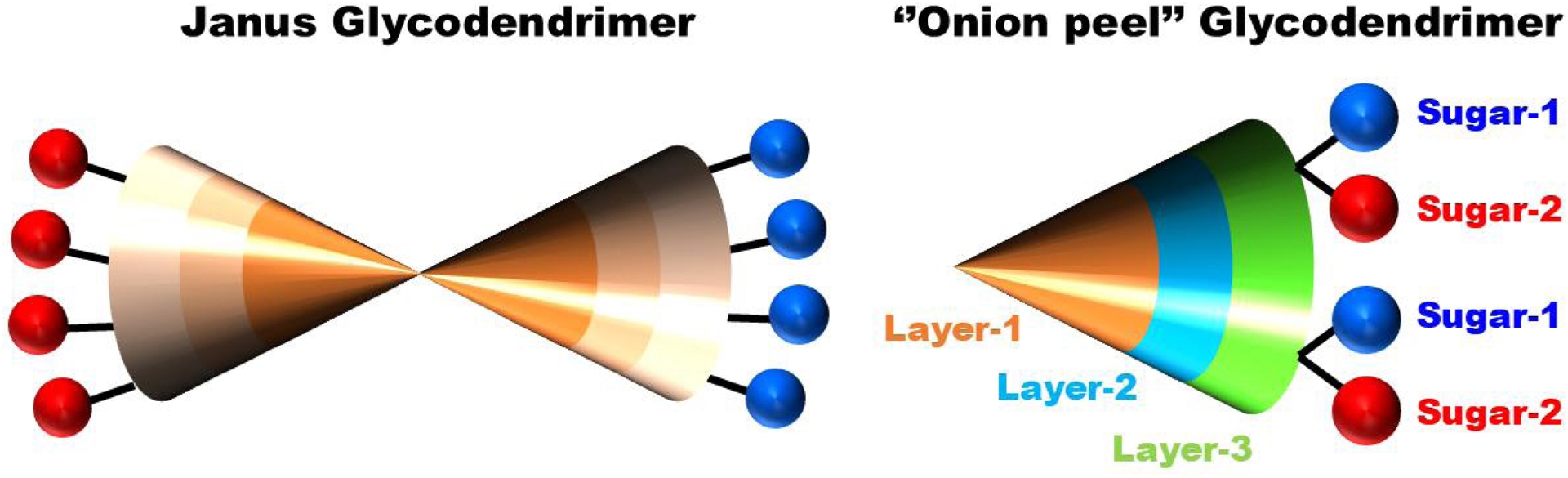
An additional variant was further included through which both the Janus dendrimer and the onion-peel concept were combined together with optimized saccharide aglycones to provide specificities toward each targeted lectin (Figure 6) [42, 43]. Hence, the implementation of the orthogonal ligation strategy was also incorporated into our design. For this purpose, a photolytic thiol–ene reaction was combined with click chemistry (Schemes 5 and 6).
Building an orthogonally functionalized s-triazine triol scaffold to be used for the divergent synthesis of an heterolayered onion-peel glycodendrimer.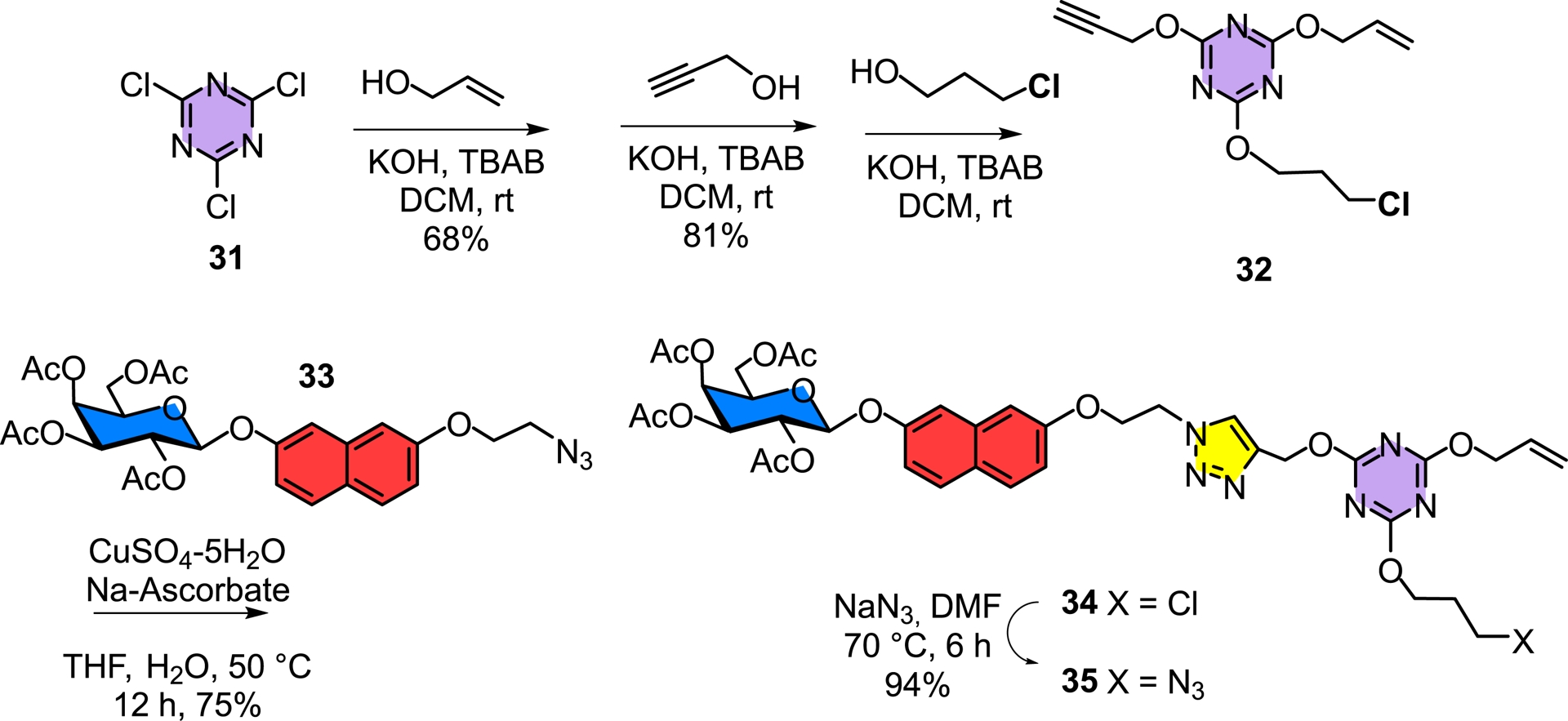
Synthesis of a heterobifunctional glycodendron harboring both galactose and mannose residues together with orthogonal azide at the focal point.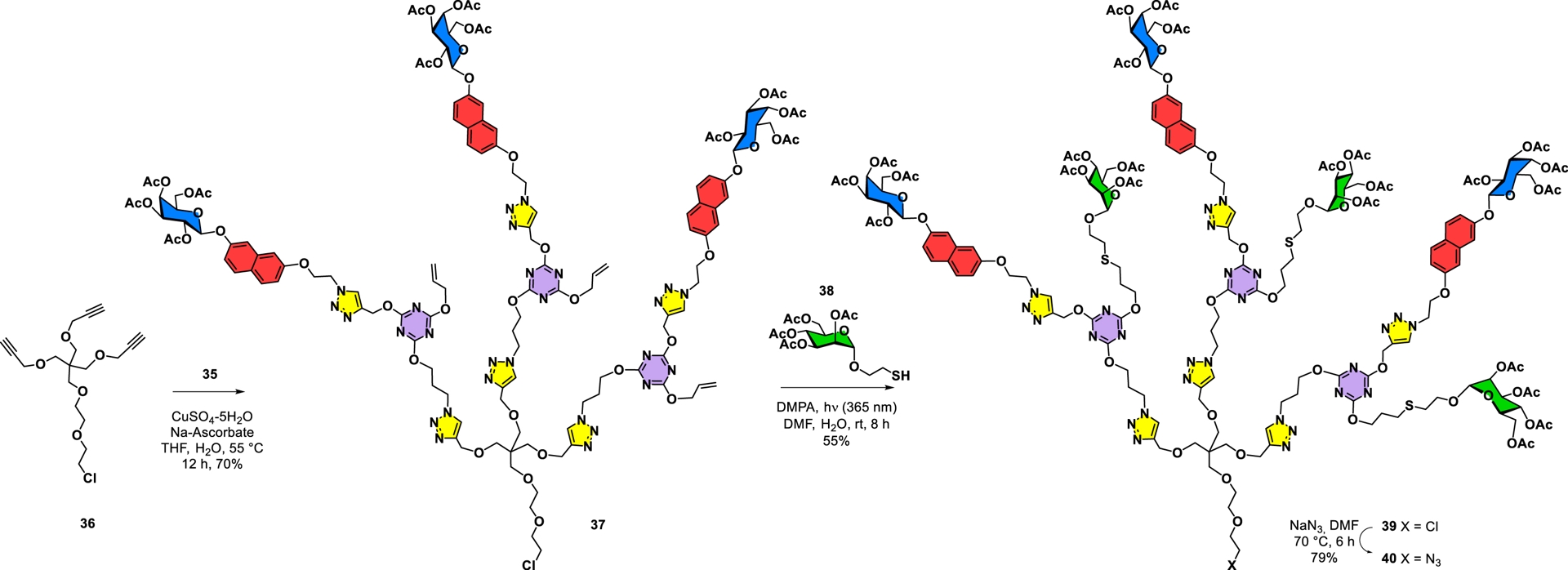
Finally, the target glycodendrimer was cleanly built on the hexakispropargylated cyclotriphosphazene core 12 using powerful click chemistry to yield 41 (59%) (Scheme 7). It is to be noted that these complex peracetylated compounds could be readily purified by standard silica gel column chromatography. Conventional transesterification under Zemplén conditions (K2CO3, MeOH) provided glycodendrimer 42 (90%) containing 36 alternating carbohydrate surface groups, of which 18 were β-D-galactopyranosides and a further 18 were α-D-mannopyranosides. This case constitutes an ideal example corresponding to a 6 × 3 × 2 methodology (A6 core, AB3 interlayer, and AB2 interlayer).
Convergent building of an alternating heterobifunctional glycodendrimer harboring a total of 36 galactoside and mannoside residues using a cyclotriphosphazene core.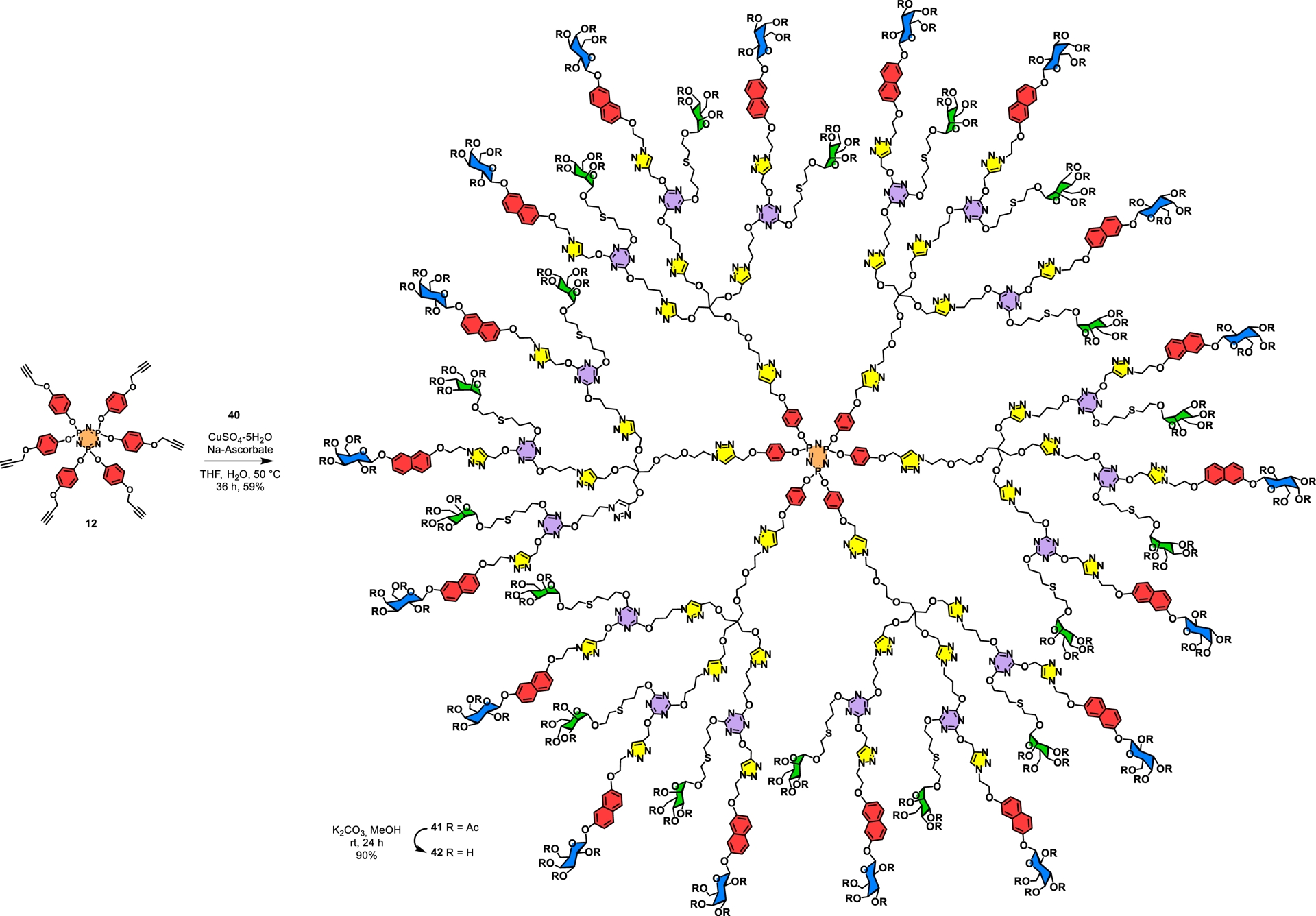
The biochemical functionality of the aforementioned hybrid glycodendrimer (42) was evaluated using kinetic studies through DLS. Consequently, 42 (∼3–5 nm) readily formed large cross-linked aggregates in the presence of the homotetrameric mannose-specific leguminous lectin ConA (∼8 nm) from C. ensiformis (jack bean), resulting in glycodendrimer–protein cross-linked aggregates of ∼3500 nm within 2 min [9]. At the same time, the galactose-specific homotetrameric lectin LecA from P. aeruginosa also formed large cross-linked lattices (∼5000 nm) within 4 min, thus demonstrating the accessibility of either glycoside with larger protein receptors.
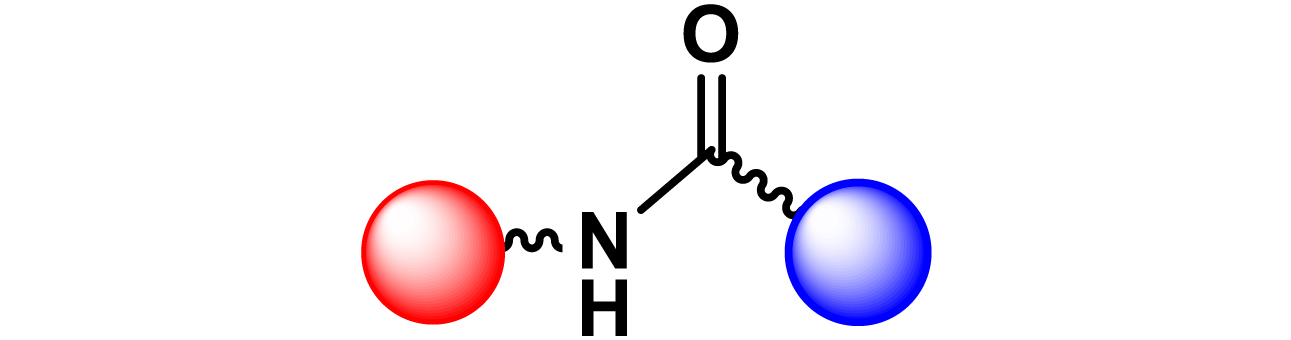
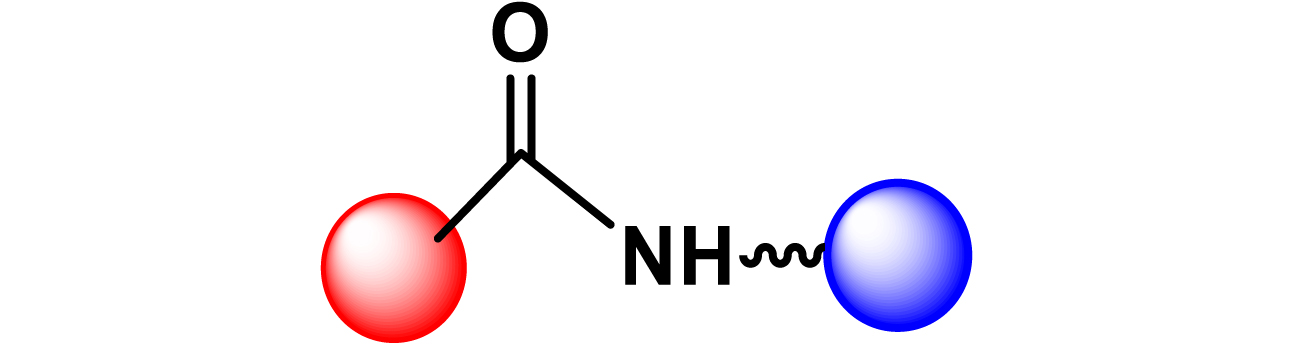
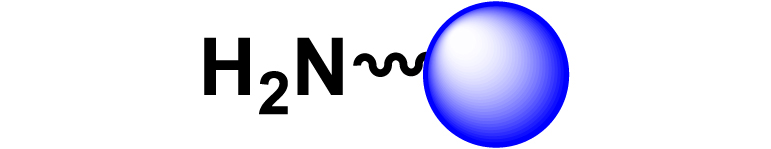
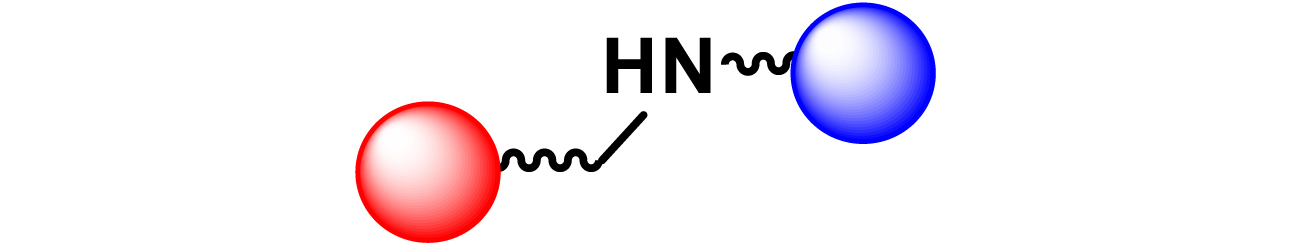
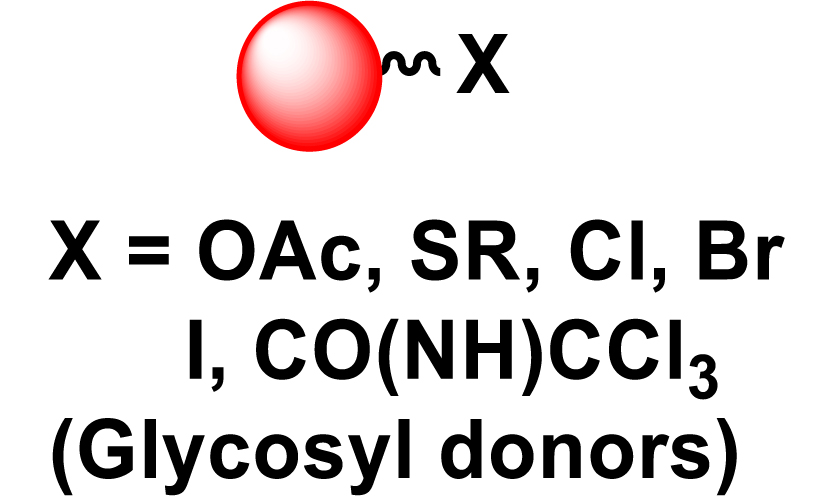
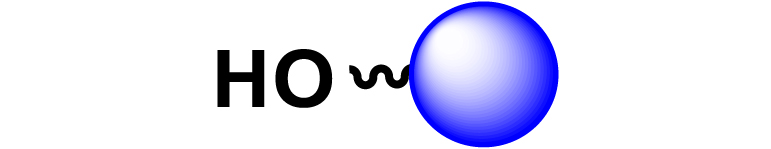
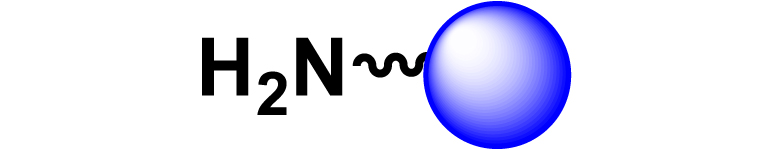
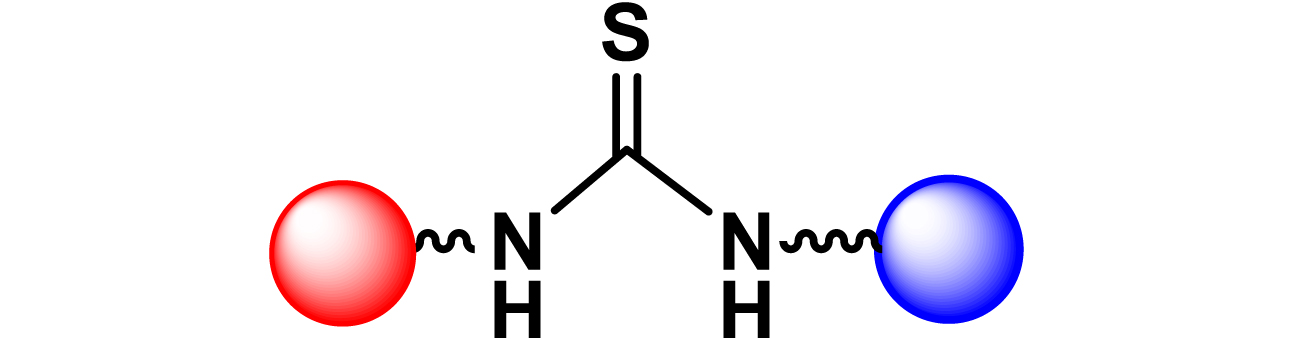
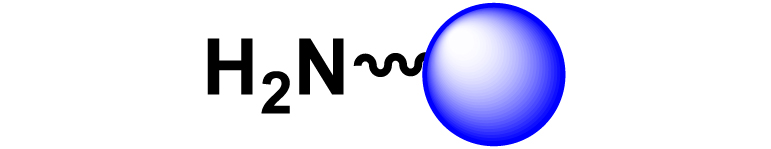
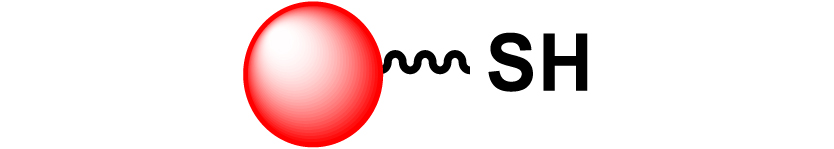
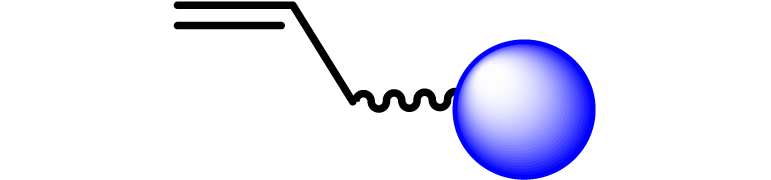
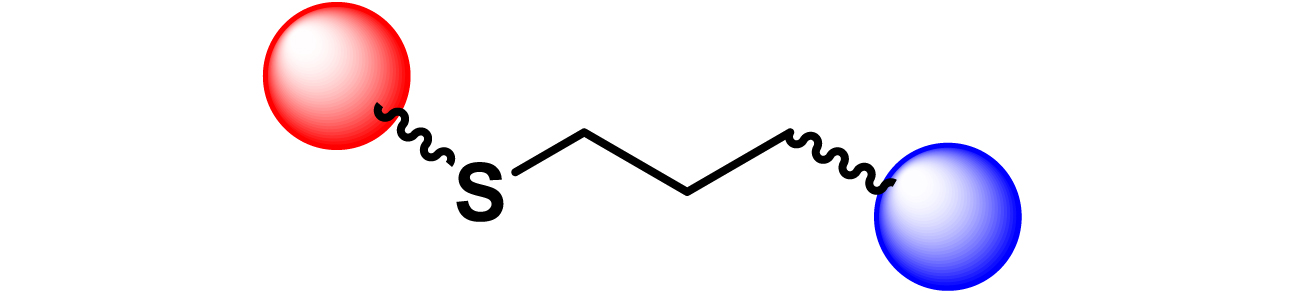
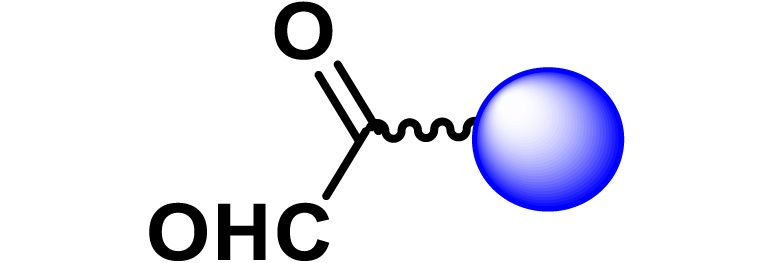
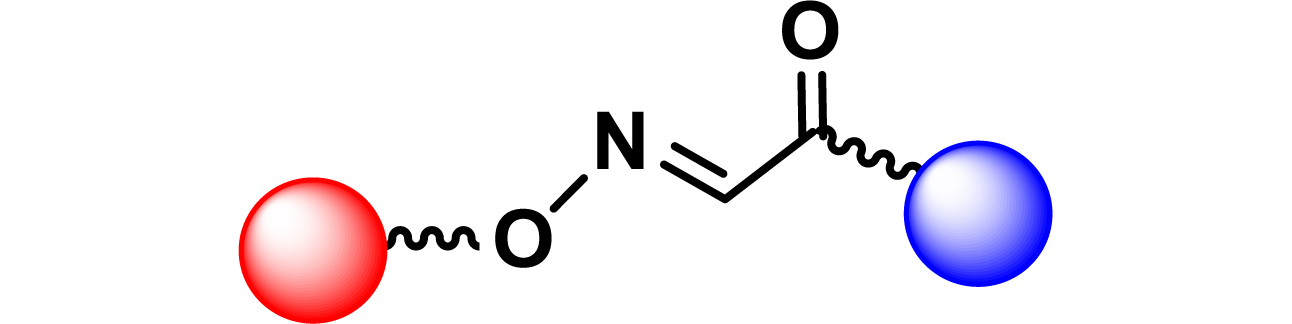
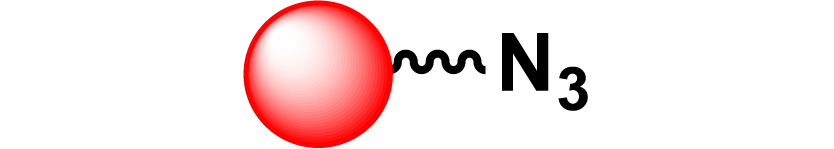
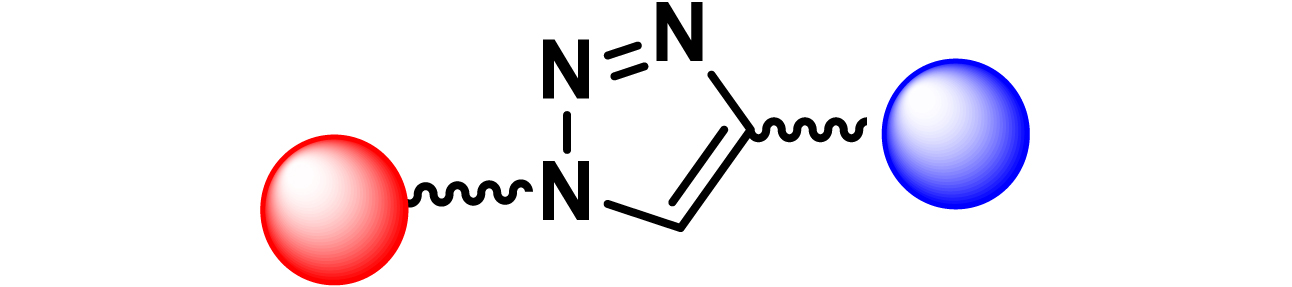
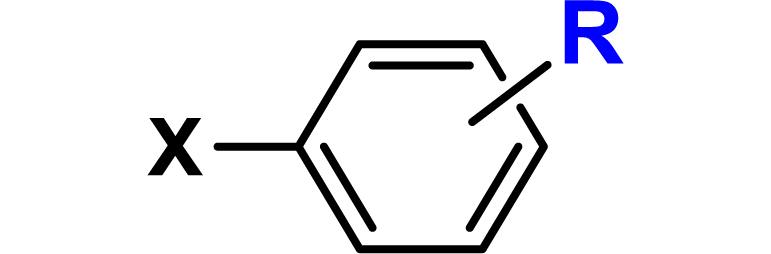
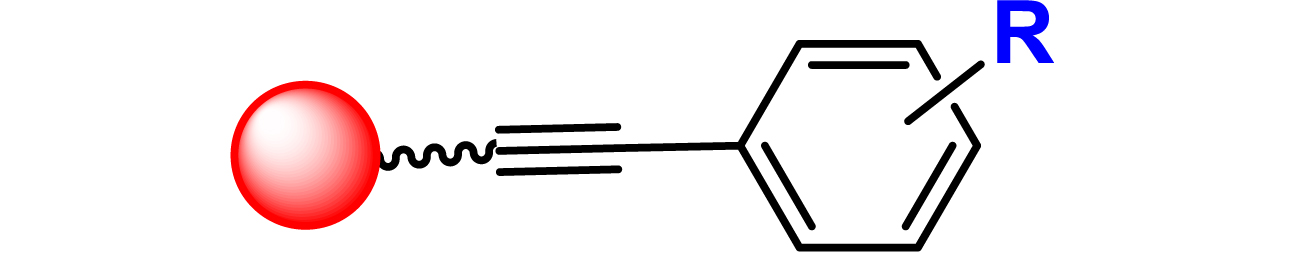
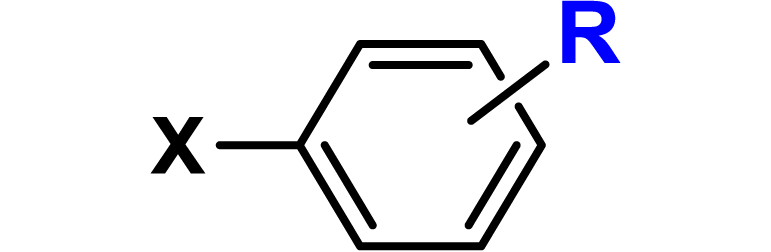
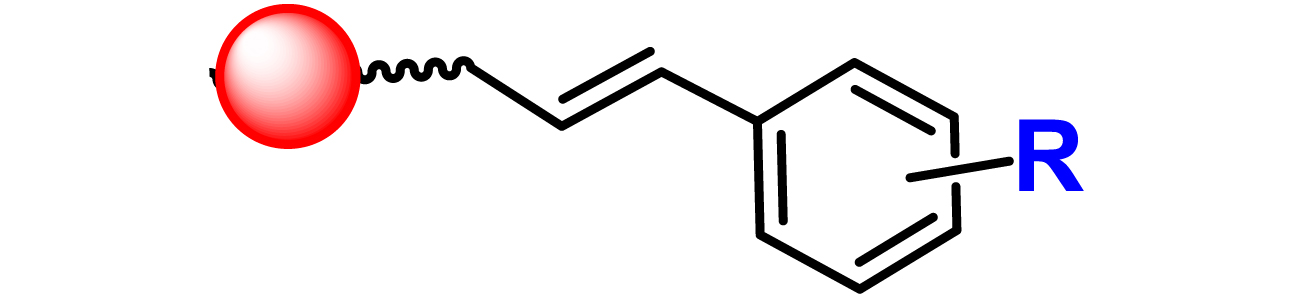
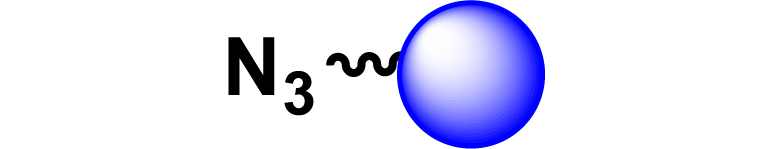
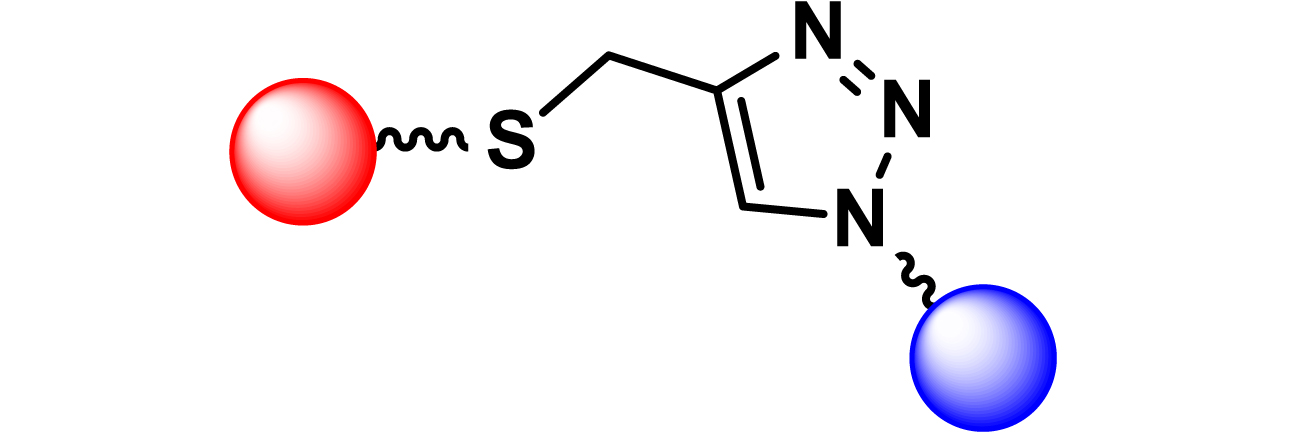
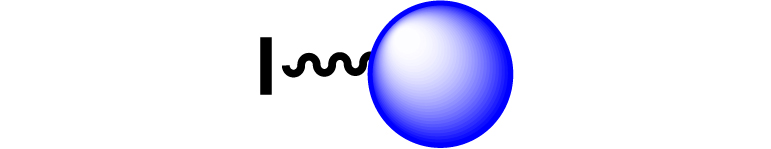
Given the high potential of glycodendrimers in biological settings and the complexity of carbohydrate entities with their multiple and varied functional groups together with the necessity for reliable and selective ligation chemistry, we illustrate the most common of these glycoconjugation partners in Table 1. As can be seen, the carbohydrate moieties usually employed in glycoconjugate synthesis such as amines, acids, azides, and alkynes represent the most frequently encountered examples that are traditionally utilized.
Most common ligation chemistry used in the synthesis of glycodendrimers
| Entry | Carbohydrate functionality | Scaffold functionality | Ligation | Refs. |
|---|---|---|---|---|
| 1 |
 |
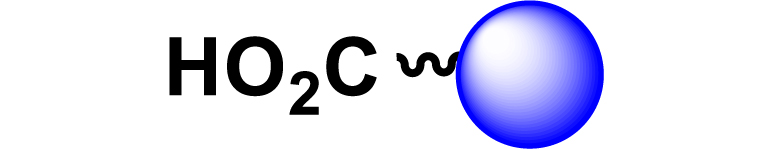 |
 |
[99, 100] |
| 2 |
 |
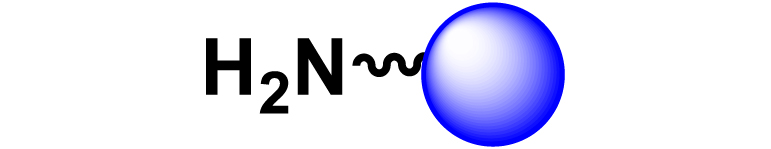 |
 |
[99, 101] |
| 3 |
 |
 |
 |
[102, 103, 104] |
| 4 |
 |
 |
 |
[105] |
| 5 |
 |
 |
 |
[106, 107, 108] |
| 6 |
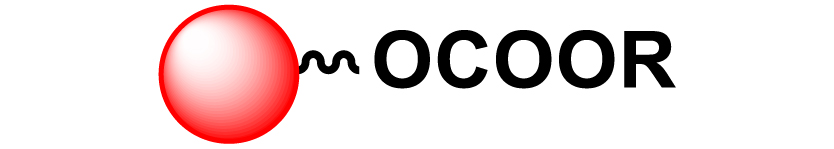 |
 |
 |
[109, 110, 111] |
| 7 |
 |
 |
 |
[72, 97, 112] |
| 8 |
 |
 |
 |
[3, 113, 114] |
| 9 |
 |
 |
 |
[32, 115, 116] |
| 10 |
 |
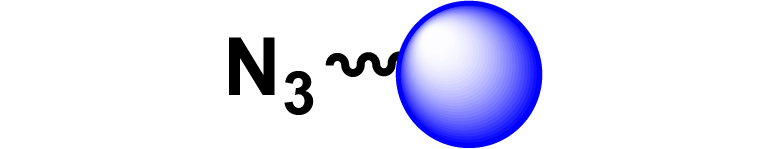 |
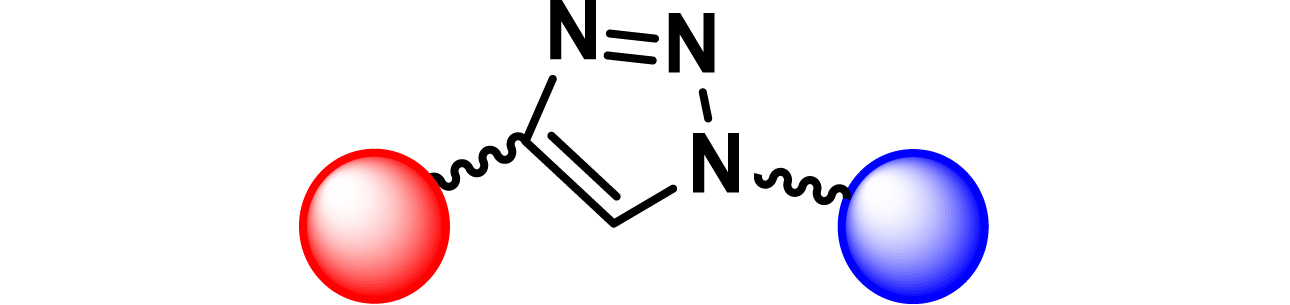 |
[32, 115, 116] |
| 11 |
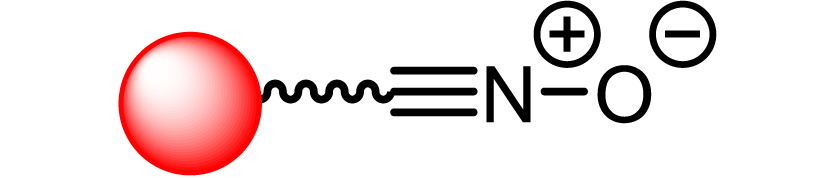 |
 |
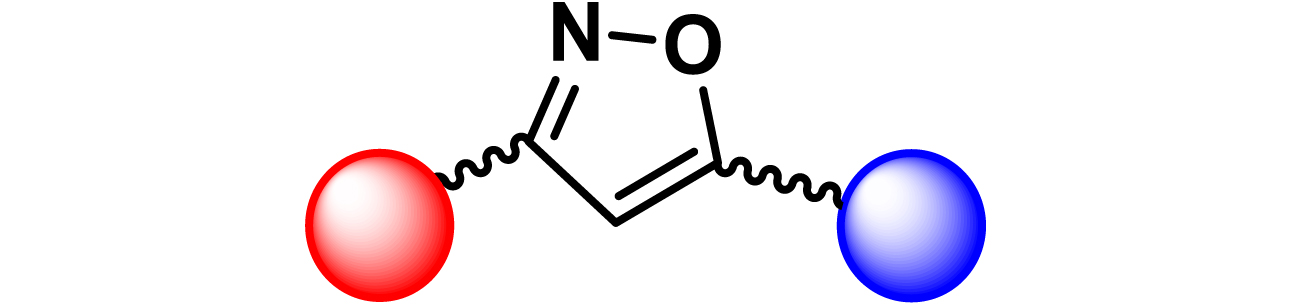 |
[117] |
| 12 |
 |
 |
 |
[118] |
| 13 |
 |
 |
 |
[119] |
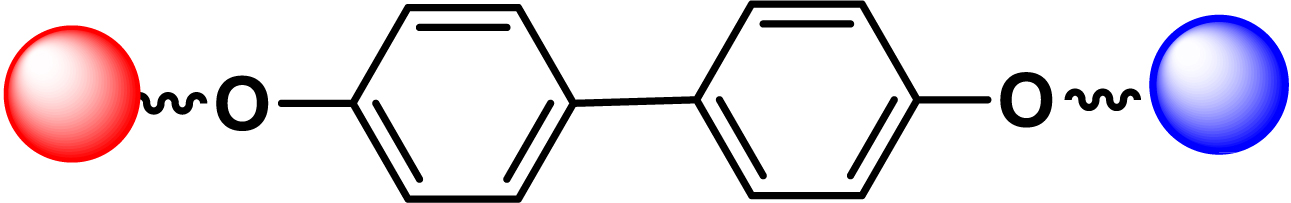
| 14 |
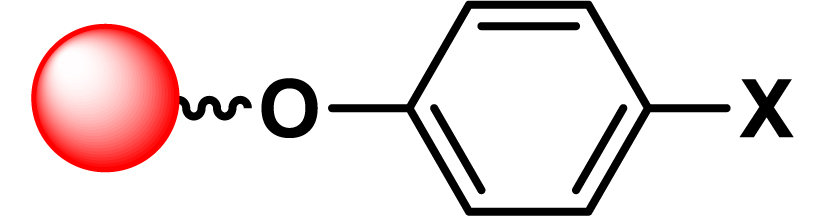 |
 |
 |
[120, 121] |
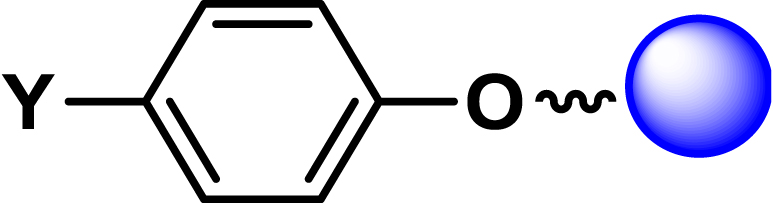
| 15 |
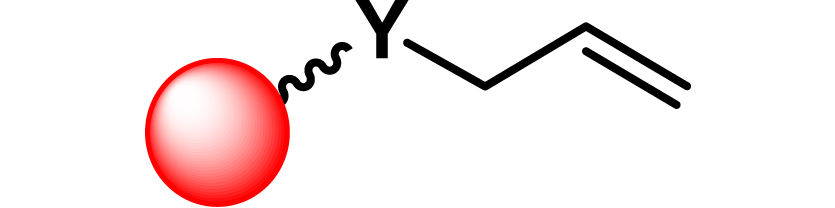 |
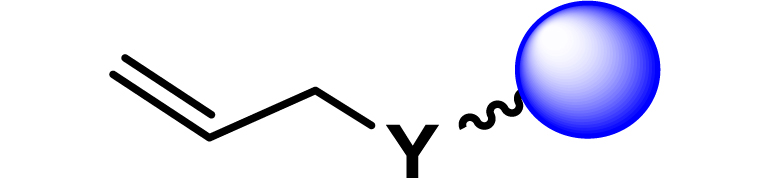 |
 |
[122, 123] |
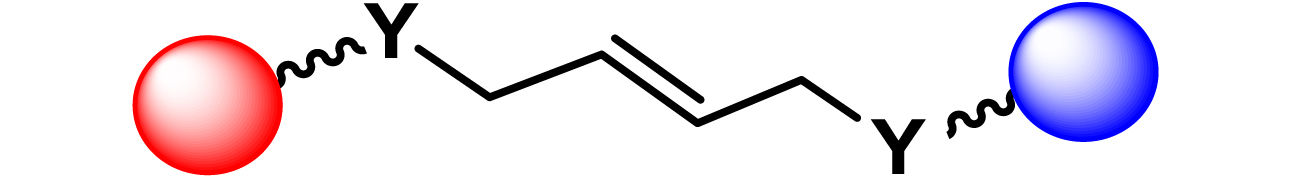
| 16 |
 |
 |
[124, 125, 126] | |
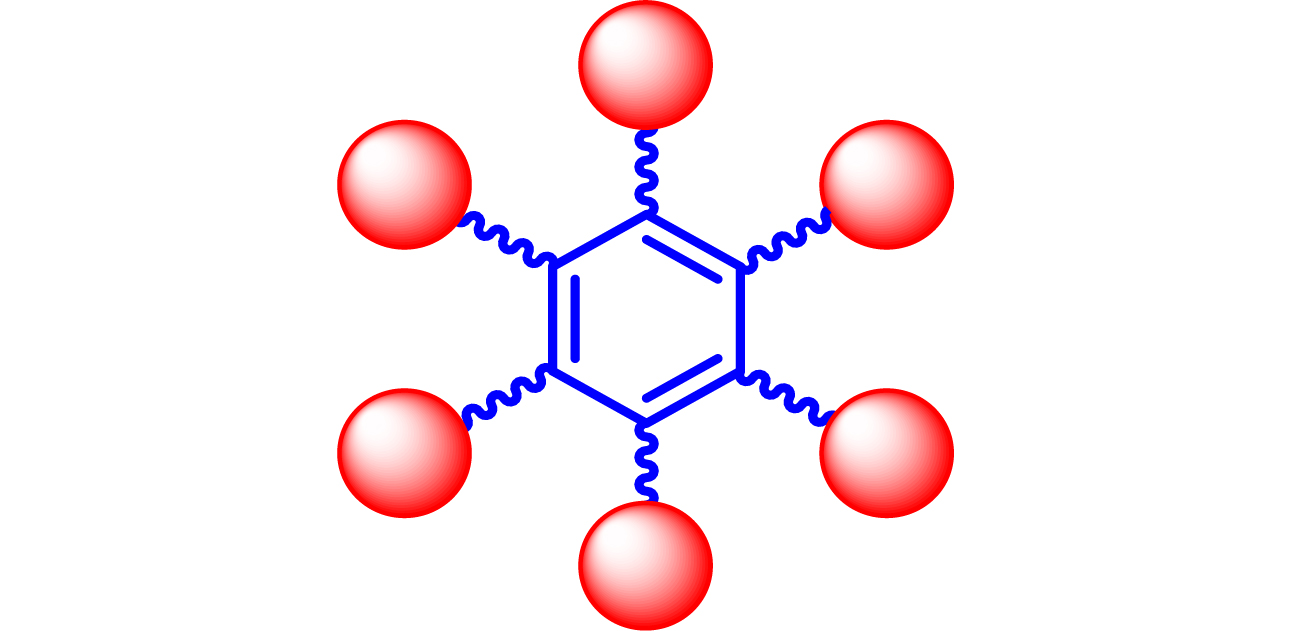
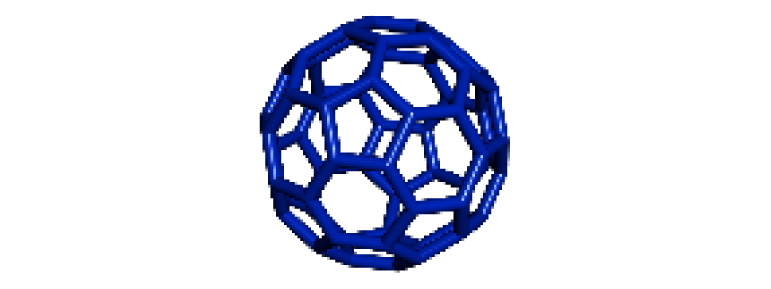
| 17 |
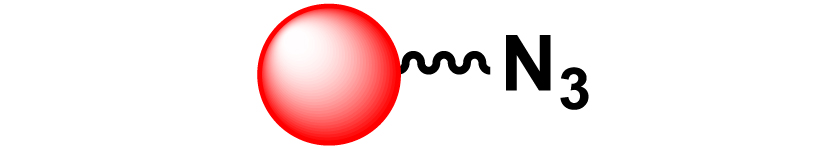 |
 |
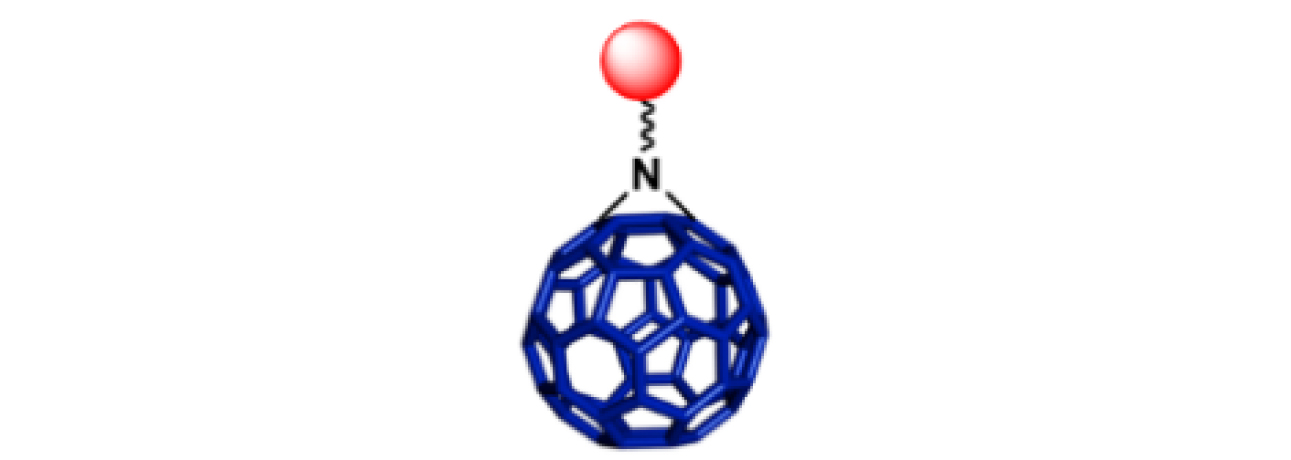 |
[127, 128] |
| 18 |
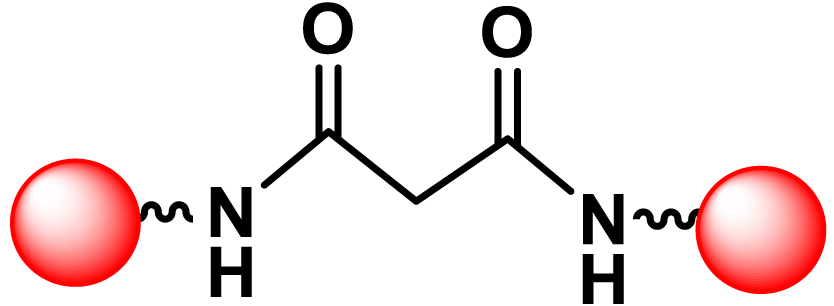 |
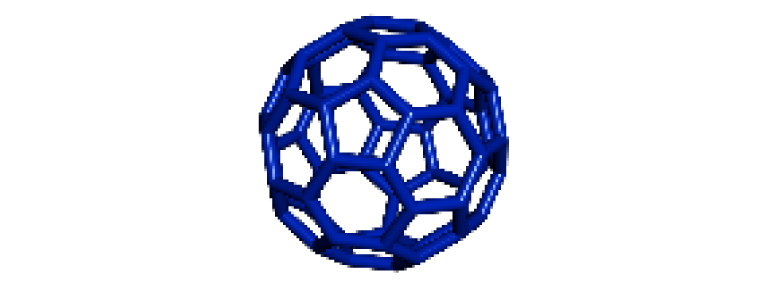 |
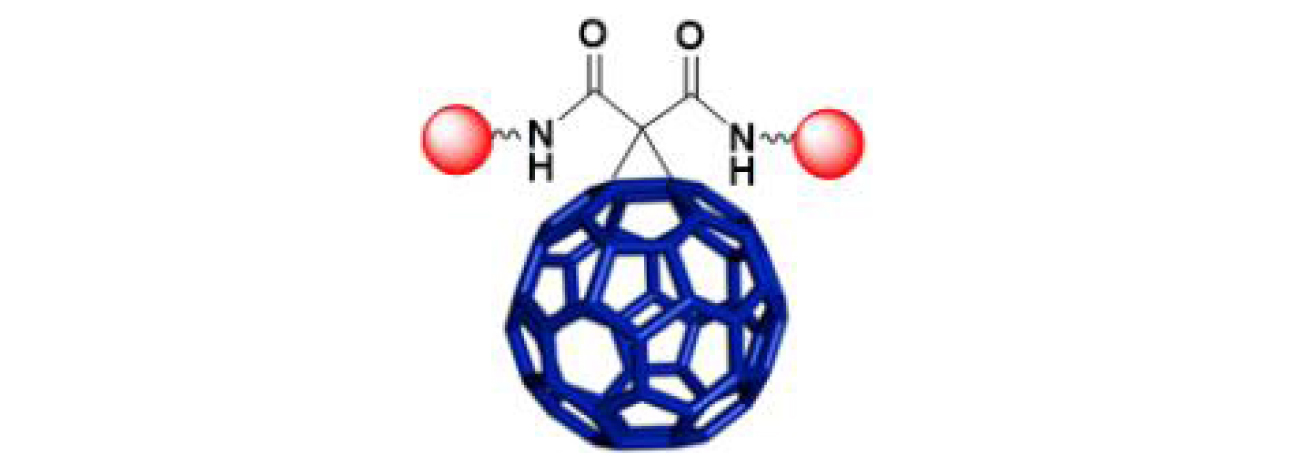 |
[129] |
| 19 |
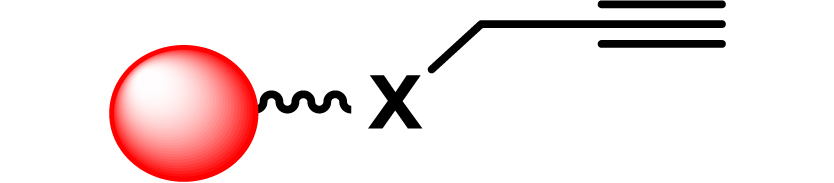 |
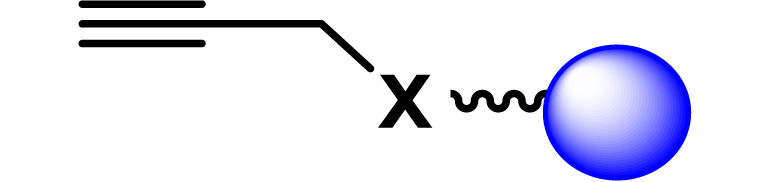 |
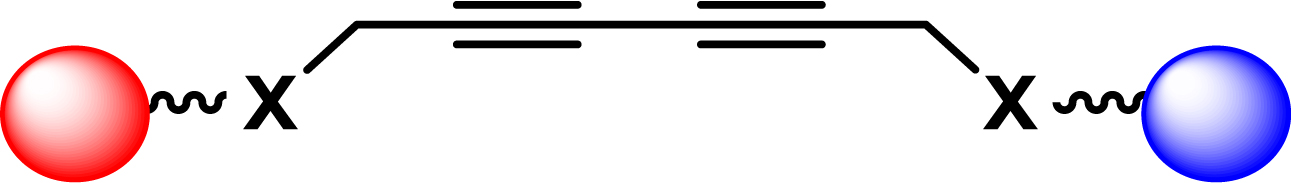 |
[130] |
| 20 |
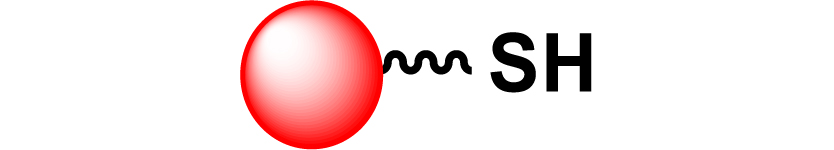 |
 |
 |
[91, 131] |
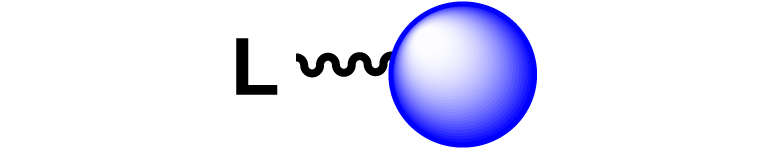
| 21 |
 |
 |
 |
[32, 132] |
| 22 |
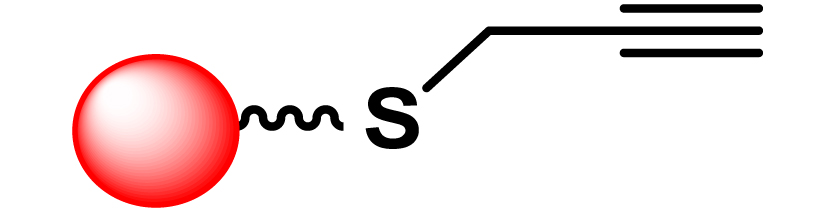 |
 |
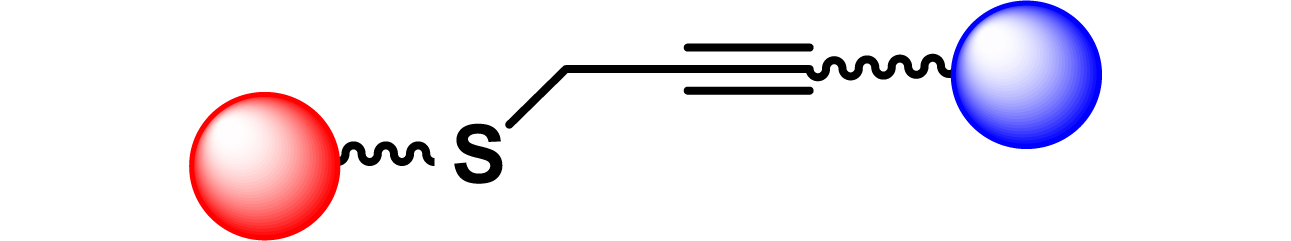 |
[72, 133] |
| 23 |
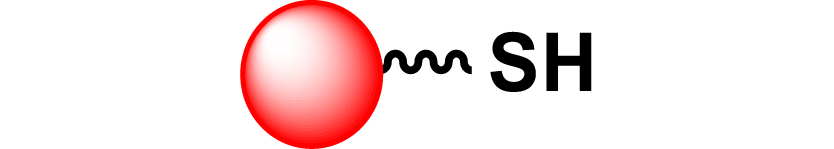 |
 |
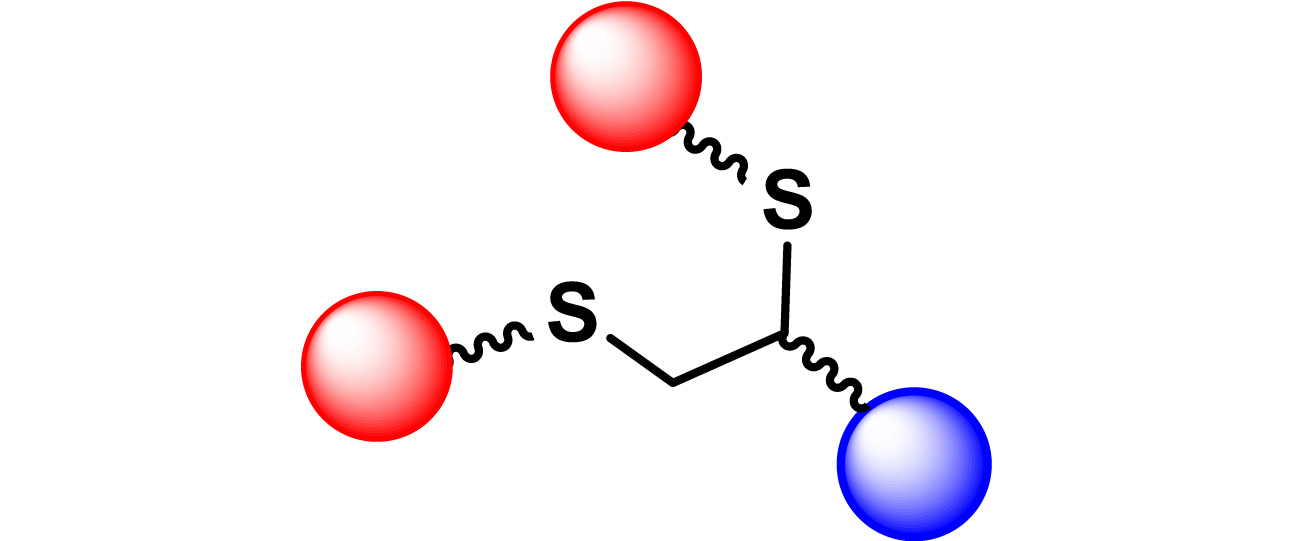 |
[115] |
3. Self-assembling glycodendrimers
Self-assembling is an important phenomenon in biological systems; for example, the formation of cell membranes and metal-based chelation (metalloproteins) represent truly inspiring examples of supramolecular chemistry [134]. Therefore, its implementation in the field of glycodendrimers can greatly reduce the number of steps necessary for the construction of well-defined multivalent architectures having nanometer size. In this section, we illustrate the evolution of our thought process from the initial description of a randomly disposed glycopolymer strategy to describing case studies with transition-metal-templated self-assembly and glycodendrimersomes.
3.1. Transition-metal-templated self-assembly: glyco-metallo-dendrimers
The concept of self-assembly—where molecules spontaneously organize into structured arrangements—has revolutionized the way we design functional materials. Among the most versatile tools in this domain are transition metals, which serve as dynamic templates guiding the formation of complex architectures. Transition-metal-templated self-assembly leverages the coordination chemistry of metals like iron, copper, palladium, and ruthenium to direct the spatial arrangement of ligands and molecular building blocks [135, 136, 137, 138]. In essence, transition-metal-templated self-assembly is not just a synthetic strategy—it is a design philosophy that blends chemistry, physics, and engineering to build order from chaos. The metal-templated self-assembly strategy has also been effectively extended into the field of glycodendrimers by combining dynamic combinatorial chemistry [139, 140].
In the following example, a 2,2′-bipyridine-4,4′-dicarboxylic acid core was selected as a Cu-based assembly. A bivalent glycodendron composed of N-acetyl-D-galactosamine (GalNAc) (43) was assembled using classic amide bonding to form a tetravalent subunit for the coordination with copper sulfate (Scheme 8). The self-assembly was simply achieved with only two equivalents of 43—one equivalent of CuSO4⋅5H2O in water at 23 °C for two days—to deliver the metal-based symmetrical self-assembled glycodendrimer (44) as a blue-purple colored powder after lyophilization. The readily formed octavalent GalNAc cluster 44 showed high inhibitory properties in the binding of asialoglycophorin to the leguminous lectin V. villosa with a 259-fold higher potency in comparison to its monomeric counterpart [141].
Transition metal used in the self-assembly process toward the construction of glycodendrimers [141].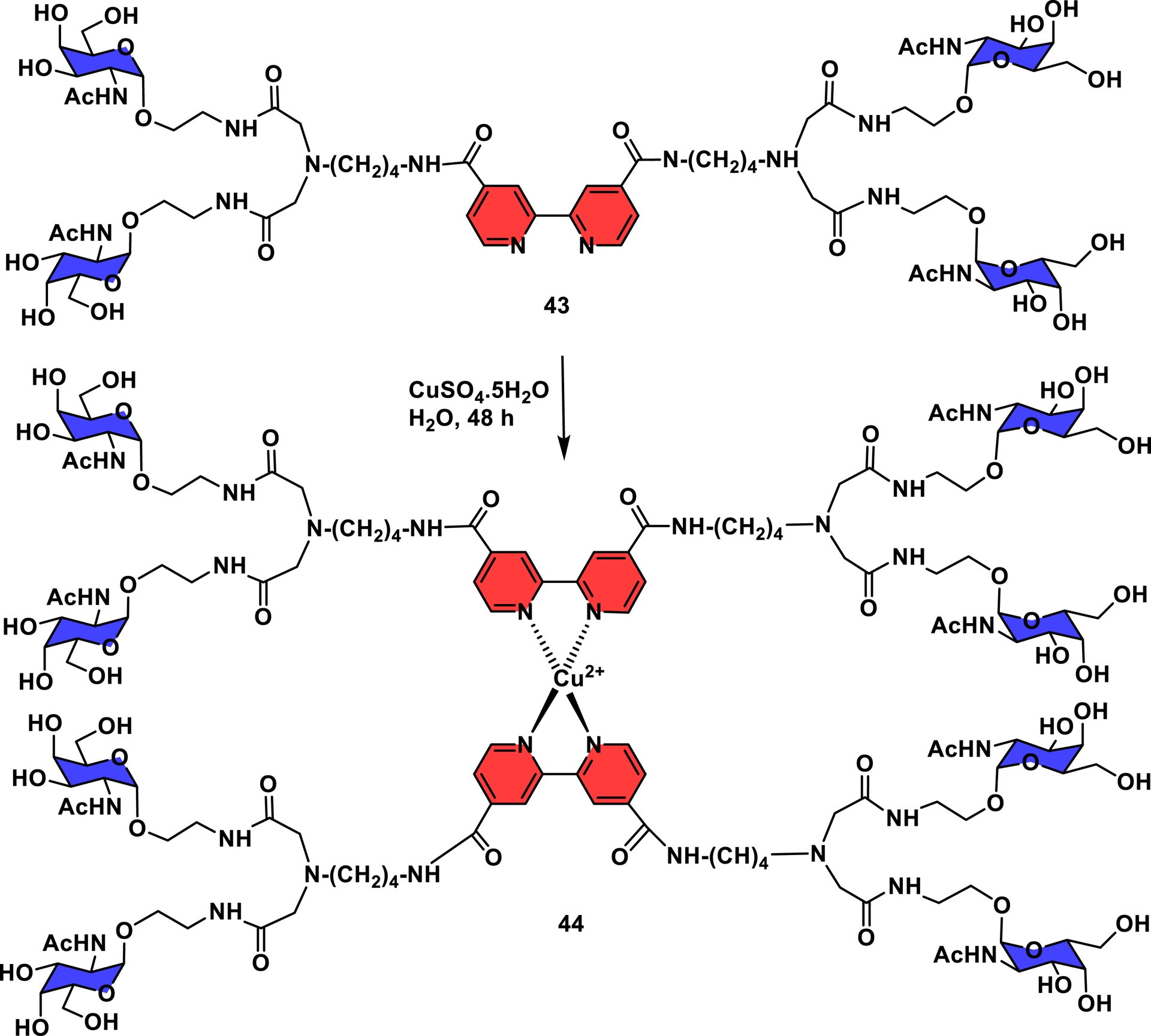
3.2. Lipid-templated self-assembly: glycodendrimersomes
Glycolipids [142], amphiphilic molecules composed of a hydrophilic carbohydrate head group and a hydrophobic lipid tail, exhibit remarkable self-assembly behavior in aqueous environments due to their dual chemical nature. This intrinsic amphiphilicity drives the spontaneous formation of organized supramolecular structures such as micelles, vesicles, and fibrous networks depending on molecular geometry, concentration, and environmental conditions. The self-assembly process is governed by noncovalent interactions, including hydrogen bonding, van der Waals forces, and hydrophobic effects, which collectively stabilize the resulting nanostructures.
Microbial glycolipids are particularly notable for their ability to form reversible, stimulus-responsive assemblies. These systems can transition between morphologies—for example, from spherical micelles to elongated fibers—based on external cues, making them highly versatile for biomedical and industrial applications. Additionally, amphiphilic glycolipid glycoconjugates have been employed to encapsulate hydrophobic drugs and dyes, enhancing solubility, stability, and targeted delivery.
The biocompatibility, biodegradability, and sustainable synthesis of glycolipids via enzymatic or chemoenzymatic routes from renewable feedstocks further underscore their potential in green chemistry and nanomedicine. Their ability to self-organize into functional architectures without the need for harsh processing conditions positions glycolipids as promising candidates for next-generation soft materials and bioactive platforms.
In collaboration with the Percec group, we synthesized seven libraries containing 51 self-assembling amphiphilic Janus glycodendrimers composed of monosaccharides D-mannose and D-galactose and the disaccharide D-lactose denoted “glycodendrimersomes” using different hydrophobic aromatic cores and saccharide-based hydrophilic head groups having different linkers (Figure 7) [143, 144, 145]. Scheme 9 illustrates a typical example for the construction of these biologically relevant amphiphilic nanosized (∼100 nm) materials using click chemistry.
Principle used in the construction of self-assembling glycodendrimersomes from lipophilic tails and glycodendrons [143, 144, 145].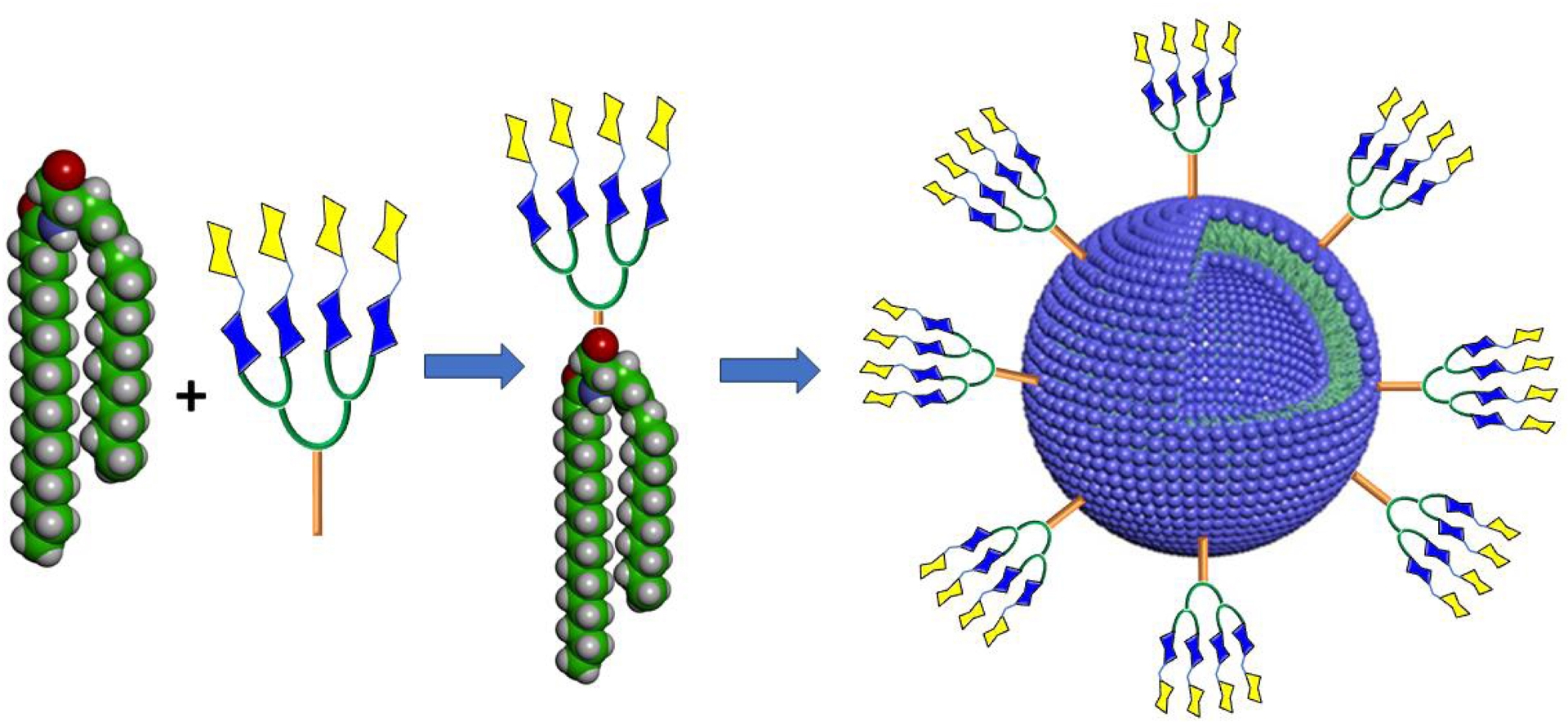
Typical example used in the syntheses of the first glycodendrimersomes [143, 144, 145].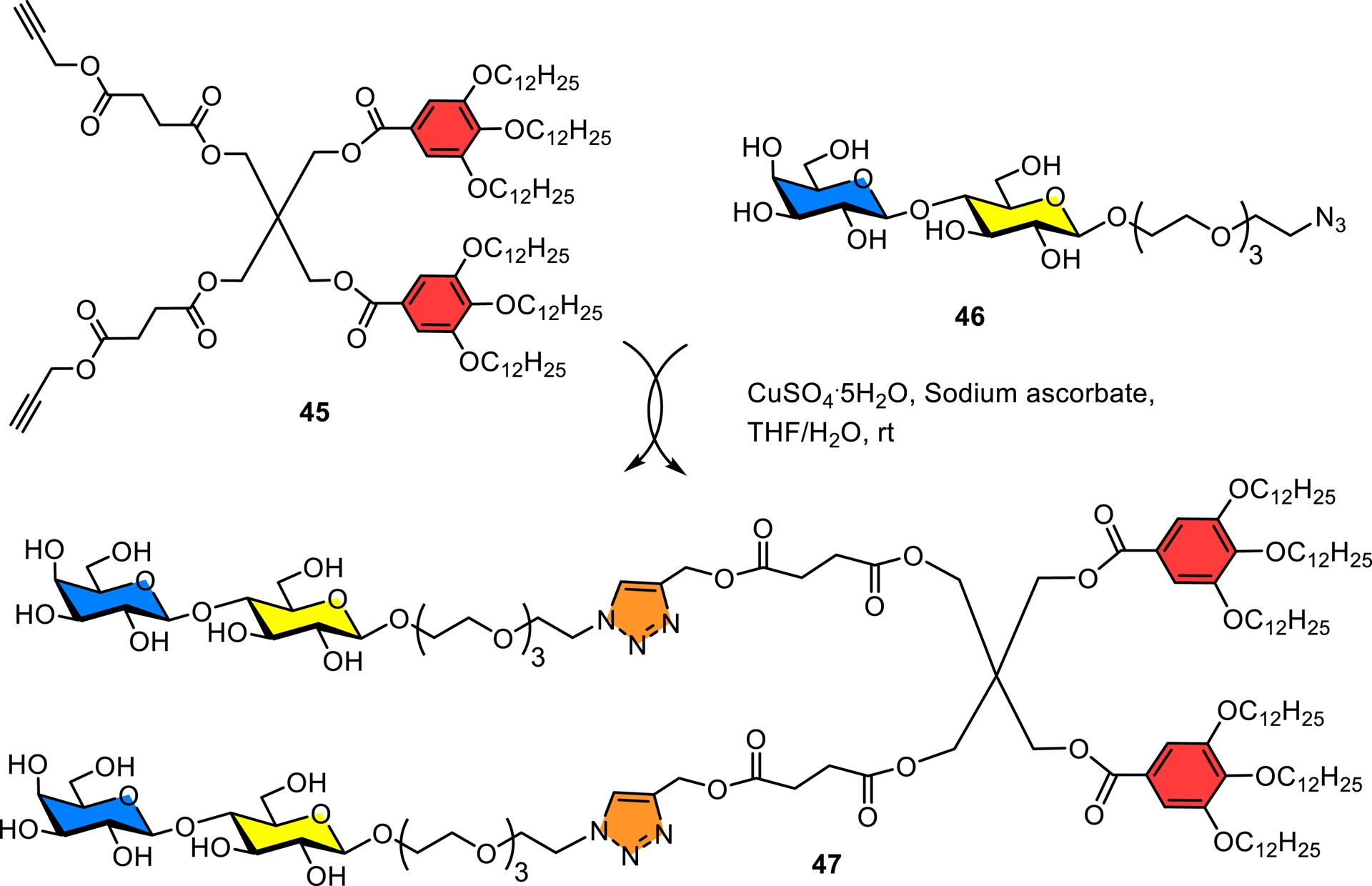
These self-assembled glycodendrimersomes were stable over time in water and in buffer, exhibited narrow molecular-weight distribution, and displayed dimensions that are programmable by the concentration of the solution from which they are injected. This study expanded the molecular principles leading to single-type soft glycodendrimersomes assembled from amphiphilic Janus glycodendrimers. The multivalency of glycodendrimersomes with different sizes and their ligand bioactivity were demonstrated by selective agglutination assays with a diversity of glycoside-binding protein receptors such as the plant lectins ConA (D-mannose/D-glucose), the highly toxic mistletoe Viscum album agglutinin (D-Gal/D-GalNAc), and the bacterial lectin PA-IL from P. aeruginosa (D-Gal). The galactoside-containing glycodendrimersomes are of significant biomedical relevance since they were shown capable of binding to human adhesion/growth-regulatory galectin-3 and galectin-4. These results demonstrated the candidacy of glycodendrimersomes as new mimics of biological membranes with programmable glycan ligand presentations, as supramolecular lectin blockers, vaccines, and targeted delivery devices.
4. Conclusion
In this review, recent advances in the design of chemical architectures incorporating carbohydrate moieties are discussed. The multivalency of the resulting ligands significantly enhances lectin-binding avidity, thereby modulating the ability of carbohydrates to engage in improved supramolecular recognition processes. These interactions facilitate the transfer of biological information and enable targeted engagement with complementary lectin receptors.
However, current models often fail to account for the heterogeneity and dynamic nature of densely glycosylated cell membranes. They also overlook the potential influence of high-density, nanoscale arrangements on interactions with the broader spectrum of carbohydrate-binding proteins. For years, it has been tacitly assumed the following: (i) effective lectin recognition generally necessitates high densities of the primary ligand and (ii) the mechanisms underlying carbohydrate motif recognition by lectins versus glycosidases are fundamentally distinct. We proposed a more generalized multivalent construction strategy to better address issues related to the topographical chemistry of carbohydrate and glycomimetic structures in biological systems. Within this context, (hetero)multivalency functions as a multimodal switch, channeling encoded information through diverse molecular pathways. We firmly believe that further development would be essential to translate these insights into practical applications, particularly in the promising field of glycodendrimers.
Acknowledgments
This work was supported by grants from the Natural Science and Engineering Research Council of Canada (NSERC) to RR (grant no. 70732101). GlycoNet, supported by the federal government’s Strategic Science Fund, provided support for the research (CT-19). The Université du Québec à Montréal (UQAM) is profoundly acknowledged for conferring the status of Emeritus professor on RR. The authors also acknowledge funding from the Programme de recherche CNRST-FRQ (Maroc-Québec; No. 327250).
Declaration of interests
The authors do not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and have declared no affiliations other than their research organizations.





 CC-BY 4.0
CC-BY 4.0