1. Introduction
Grapes are widely consumed both in their fresh form and as processed products, and they represent one of the most extensively cultivated fruits worldwide. More than 80% of global grape production is destined for winemaking [1, 2, 3]. Despite its economic relevance, the wine industry traditionally follows a linear production model in which raw materials are processed, consumed, and subsequently discarded as waste [4]. It is estimated that the production of 750 L of wine requires approximately one ton of grapes, generating substantial amounts of by-products such as stems, skins, and seeds. Grape pomace alone accounts for nearly 60% of the total waste generated during winemaking [5, 6].
The accumulation of winery by-products reflects inadequate waste management practices and poses significant environmental concerns. Consequently, the sustainable valorization of these residues has gained increasing attention, as it offers an opportunity to transform production losses into value-added products. Within this context, grape pomace represents a promising resource for circular-economy strategies. Grape seeds constitute approximately 38% of the pomace and can be effectively reused as a raw material, which enables the recovery of valuable compounds and contributes to waste reduction while adding economic value to the wine industry [7, 8, 9, 10].
Grape seeds from different varieties exhibit diverse nutritional and functional properties, particularly due to their content in fatty acids and phenolic compounds. When discarded without proper treatment, these components represent a lost opportunity and may contribute to environmental pollution. The extraction of grape seed oil constitutes an effective strategy to exploit the full potential of these residues, allowing their reuse as raw materials for the development of products across various industrial sectors. Such an approach supports circular-economy principles by promoting resource efficiency and contributing to zero-waste objectives [11, 12].
Grape seeds are recognized as an important source of bioactive phytochemicals, with an oil content ranging from 8% to 20%. Grape seed oil is characterized by its high content in unsaturated fatty acids, including linoleic and oleic acids, as well as minor constituents such as tocopherols and carotenoids, which confer nutritional and functional benefits [13]. These properties make grape seed oil attractive not only for dietary applications but also for cosmetic formulations, where its antioxidant and emollient characteristics are particularly valued.
In addition, grape seed oil exhibits notable antioxidant properties associated with its phenolic compounds, vitamin E content, and oligomeric proanthocyanidins. These characteristics have driven growing interest in its application within the pharmaceutical, cosmetic, food, biofuel, and chemical industries. Beyond enhancing product quality, the utilization of grape seed oil contributes to the development of greener production pathways by mitigating environmental impacts and promoting sustainable resource management [14, 15].
While grape seed oil is valued for its favorable fatty acid composition and antioxidant properties, it is important to acknowledge that vegetable oils may also contain naturally occurring antinutritional factors, such as phytic acid or tannins, depending on the botanical source and processing conditions. These compounds may influence nutrient bioavailability and product stability, highlighting the importance of comprehensive characterization when considering food-related applications [16].
The objective of this study was to evaluate the valorization potential of grape seed residues from the wine industry in the city of Arequipa by assessing the oil yield, chemical composition, antioxidant capacity, microbiological quality, and economic feasibility of cold-pressed grape seed oil obtained from different grape varieties.
2. Materials and methods
2.1. Grape seed samples
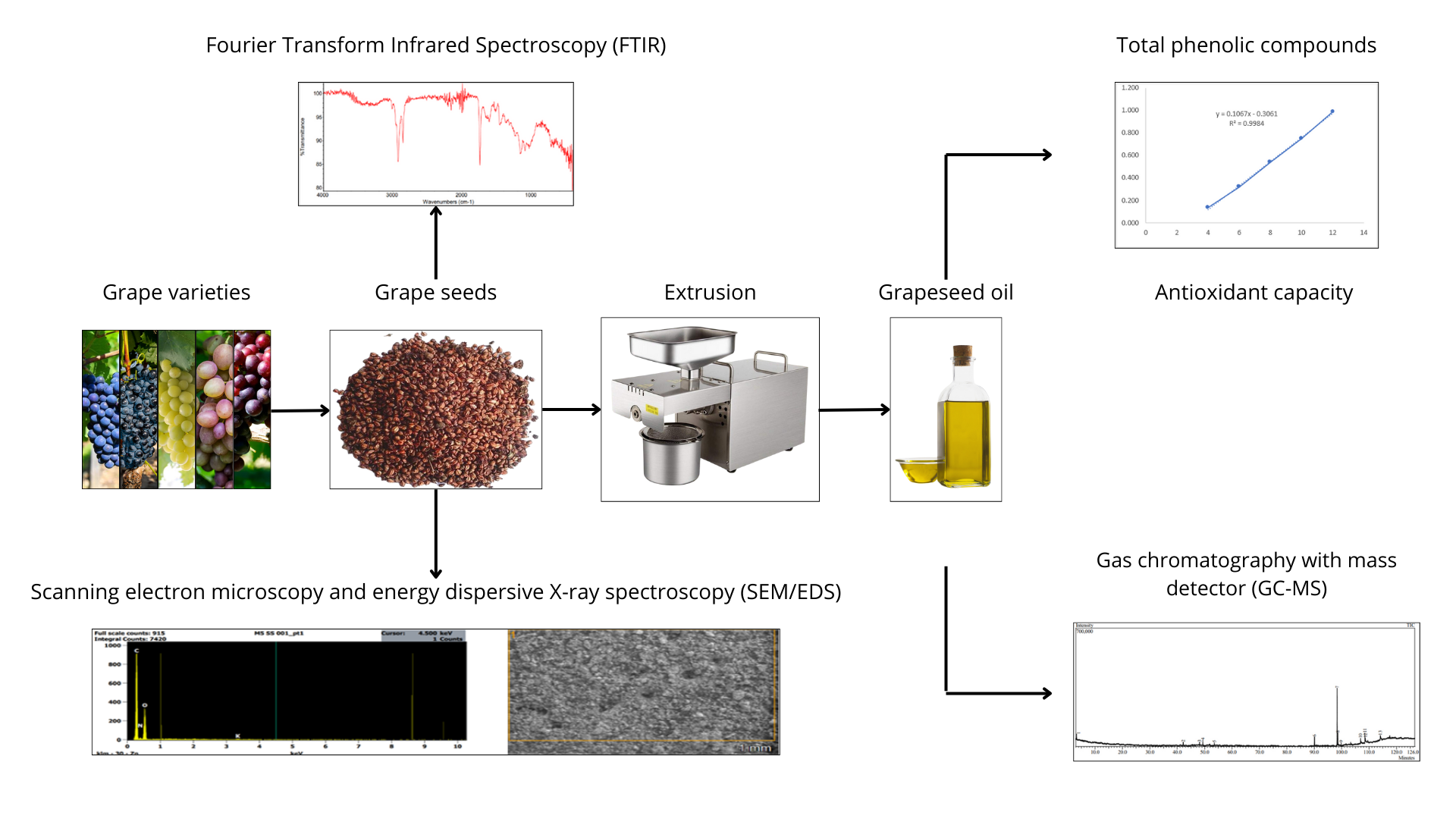
Seeds of five different varieties of Vitis vinifera were used: “Malbec”, “Syrah”, “Italia”, “Moscatel”, and “Criolla”, which were collected from the company Majes Tradición located in the city of Arequipa (Peru, 16°13′35.30′′S 72°27′04.49′′W) after the process of obtaining pisco and wine was completed. The seeds were dried at room temperature, cleaned, and stored in airtight bags protected from light until analysis. The seeds of each grape variety were ground and sifted for spectroscopic analyses.
2.2. Oil extraction from grape seeds
Oils were extracted from 500 g of each seed variety using a screw press driven by a variable speed electric motor with a screw rotating at 60 rpm. The oil was decanted, filtered, and stored at −10 °C in amber glass bottles.
2.3. Fourier-transform infrared spectroscopy (FTIR)
FTIR analyses were performed using a Nicolet iS10 mid-infrared spectrophotometer. Spectra were recorded in the 4000–400 cm−1 range in order to identify the main functional groups present in the grape seed samples.
2.4. Scanning electron microscopy–energy dispersive spectroscopy (SEM–EDS)
The surface morphology and pore diameter were evaluated using a Thermo Fisher FEG-SEM Scios 2 scanning electron microscope. The morphology of the samples was observed using a backscattered electron detector and a low vacuum detector at a pressure of 50 Pa and a voltage of 10 kV.
For chemical analysis, a Thermo Fischer Ultra-Dry energy dispersive spectroscopy (EDS) detector was used. The sifted seed samples were prepared as briquettes, mounted on an aluminum sample holder using conductive carbon tape, and subsequently coated with gold using a sample metallizer. They were blown with compressed air to prevent the release of dust particles, and then they were taken to the vacuum chamber of the electron microscope for observation.
2.5. Gas chromatography–mass spectrophotometry (GC–MS)
2.5.1. Sample Preparation
Once the oils from each grape seed variety were obtained, a pool was prepared with equal quantities f the five varieties. The oil sample (pool) was dried with anhydrous sodium sulfate and filtered using 0.45 μm syringe filters. Subsequently, 0.20 mL of the sample was diluted with n-hexane in a 10 mL vial. The diluted 10 mL sample was then transferred to a vial and placed in the GC–MS autosampler for reading.
2.5.2. GC–MS analysis
To determine the composition of the obtained oils, the following were used: a GC-2010 Plus gas chromatograph (from Shimadzu), a Shimadzu AOC-6000 autosampler, a GCMS-QP210 Ultra mass spectrometry detector (Shimadzu), GG Restek column, RTX-5MS, 30 m × 0.25 mm ID × 0.25 μm df, serial 1346249, carrier gas Helium UHP (5.0) ⩾99.999%, LS1-10 μL syringe, SPL1 injector, injection flow 100 μL/min, injection volume 1.0 μL, split type injection port, temperature of 220 °C, column flow of 0.80 mL/min, purge flow 3 mL/min. The temperature program applied was 50–150 °C with a heating rate of 3 °C/min, then from 150 to 250 °C with a heating rate of 3 °C/min, and finally 250 °C (total programmed time 126.67 min). The mass detector was set up as follows: ion source temperature of 250 °C, interface temperature of 290 °C, cut-off time of 2 min, MS start time 3 min, ionization energy of 70 eV, acquisition mode Scan and Scan range of 20–500 m/z. Identification of the components was achieved by comparing the retention indices and the respective mass spectra with those of the NIST mass spectra library (2014).
2.6. Total phenolic compounds (TPC)
Total phenolic compounds of the seed oil samples were determined using the Folin–Ciocalteu colorimetric method described by Singleton and Rossi (1965) with certain modifications [17]. For the extraction of phenolic compounds from each of the oils obtained, the procedures of Bail et al. and Pardo et al. [18, 19] were followed with some modifications. Each sample (1 g) was mixed with 5 mL of n-hexane and 1 mL of methanol:water (MetOH:H2O) (60:40). The mixture was vortexed, then centrifuged at 6000 rpm for 5 min, then extracted three times with MetOH:H2O (60:40). All methanolic extracts were combined and concentrated with heat until dry. Finally, the dry residue was dissolved in 1 mL MetOH:H2O (60:40) and stored at 4 °C in the dark. The same process was performed for the five oil varieties. For the determination of total phenolic compounds, 0.3 mL of the extract was mixed with 0.5 mL of 2N Folin–Ciocalteu reagent. After 5 min, 2 mL of a saturated sodium carbonate solution (20%) was added. The sample was incubated at 40 °C for 30 min in the dark. Finally, the absorbance was measured at 765 nm in a UV–Vis spectrophotometer (Thermo scientific). For the quantification of phenolic compounds, a calibration curve of gallic acid (4, 6, 8, 10, 12 mg/L) was prepared (R2 = 0.9984). The results were expressed in mg of gallic acid equivalent (GAE) per kilogram of oil (mg GAE/kg).
2.7. Antioxidant activity
The DPPH assay was selected as a rapid and widely used method to provide an initial estimation of the antioxidant capacity of grape seed oils. The method described by Brand-Williams et al. [20] used in different studies to determine the antioxidant capacity of different plant extracts [21, 22] was also followed as a reference. This method evaluates the antioxidant activity of the methanolic phase or polar fraction previously extracted from the grape seed oil samples.
The calibration curve was prepared with Trolox dilutions of 0.1, 0.2, 0.4, 0.6, 0.8, 1.0, and 1.2 mmol/L from a stock solution of 10 mmol/L Trolox in methanol. Then, 0.150 mL of the different concentrations of Trolox and 3 mL of the 1 mmol/L DPPH solution in methanol were mixed, stirred for 30 s, and left in the dark at room temperature. The absorbance at 520 nm was measured after 60 min. For the samples, 3 mL of the DPPH radical were mixed with 0.150 mL of the methanolic extract of the oil to be tested. The mixture was stirred and left in the dark at room temperature, proceeding to measure the absorbance after 60 min.
Antioxidant activity is expressed as the percentage of inhibition or percentage of free radical uptake, which corresponds to the amount of DPPH radical neutralized by the extract at a given concentration, as determined by the following equation:
| \begin {equation*} (\%~\mathrm {inhibition}) = \dfrac {A_{\mathrm {negative~control}} - A_{\mathrm {sample}}} {A_{\mathrm {negative~control}}} \times 100 \end {equation*} |
2.8. Microbiological analysis
Samples of grape seed oil from each variety were collected and stored in sterile amber glass bottles until analysis. For the microbiological evaluation, 1 mL of each oil sample was aseptically transferred using a micropipette and inoculated onto MC-Media Pad culture media. The inoculated pads were incubated at 37 °C for 24 h. After incubation, microbial growth was assessed by visual inspection and colony counting using a digital colony counter. The same procedure was applied to the pooled oil sample.
2.9. Cost analysis
A preliminary cost analysis was conducted to estimate the production cost of grape seed oil obtained by cold pressing under laboratory-scale conditions. A base quantity of 500 g of grape seeds was considered for the calculation. The total cost included raw material handling, labor, energy consumption, and equipment depreciation. Labor costs were estimated assuming one operator working for 2 h during the extraction process. Energy costs were calculated based on electricity consumption (kWh) of the equipment used. Equipment depreciation was estimated according to laboratory usage. The total cost was extrapolated to one liter of oil produced.
2.10. Statistical analysis
All experiments were performed in triplicate and results were expressed as averages with standard deviations. For this, ANOVA was used by employing a significance level of 𝛼 = 0.05.
3. Results and discussion
3.1. Extraction of oil from different varieties of grape seeds (Vitis vinifera L.)
Grape pomace collected from the Majes Tradición company consisted mainly of seeds, dried skins, and stems. The seeds were manually separated from the remaining pomace, thoroughly cleaned, packaged, labeled, weighed, and stored by variety in airtight bags prior to oil extraction. Grape seed oil was extracted in triplicate for each variety by cold pressing. Briefly, seeds from each variety were introduced into the hopper of the cold-pressing machine, and the extracted oil was collected. The oil was subsequently filtered using filter paper and sterile gauze to remove solid impurities and stored at 4 °C in amber glass containers to protect it from light and heat.
The extraction yield of grape seed oil was determined for each variety (Figure 1). Oil yields differed among grape varieties, with Malbec showing the highest yield (13.69%), followed by Criolla (9.51%), Moscatel (8.23%), Italia (7.75%), and Syrah, which exhibited the lowest yield (6.15%). The average oil yield was 9.07 ± 2.85%. These values fall within the oil content range of 8–20% reported in the literature for grape seeds [18, 23, 24, 25, 26, 27, 28].
Oil yield (%) from grape seeds of different Vitis vinifera varieties (mean values, n = 3).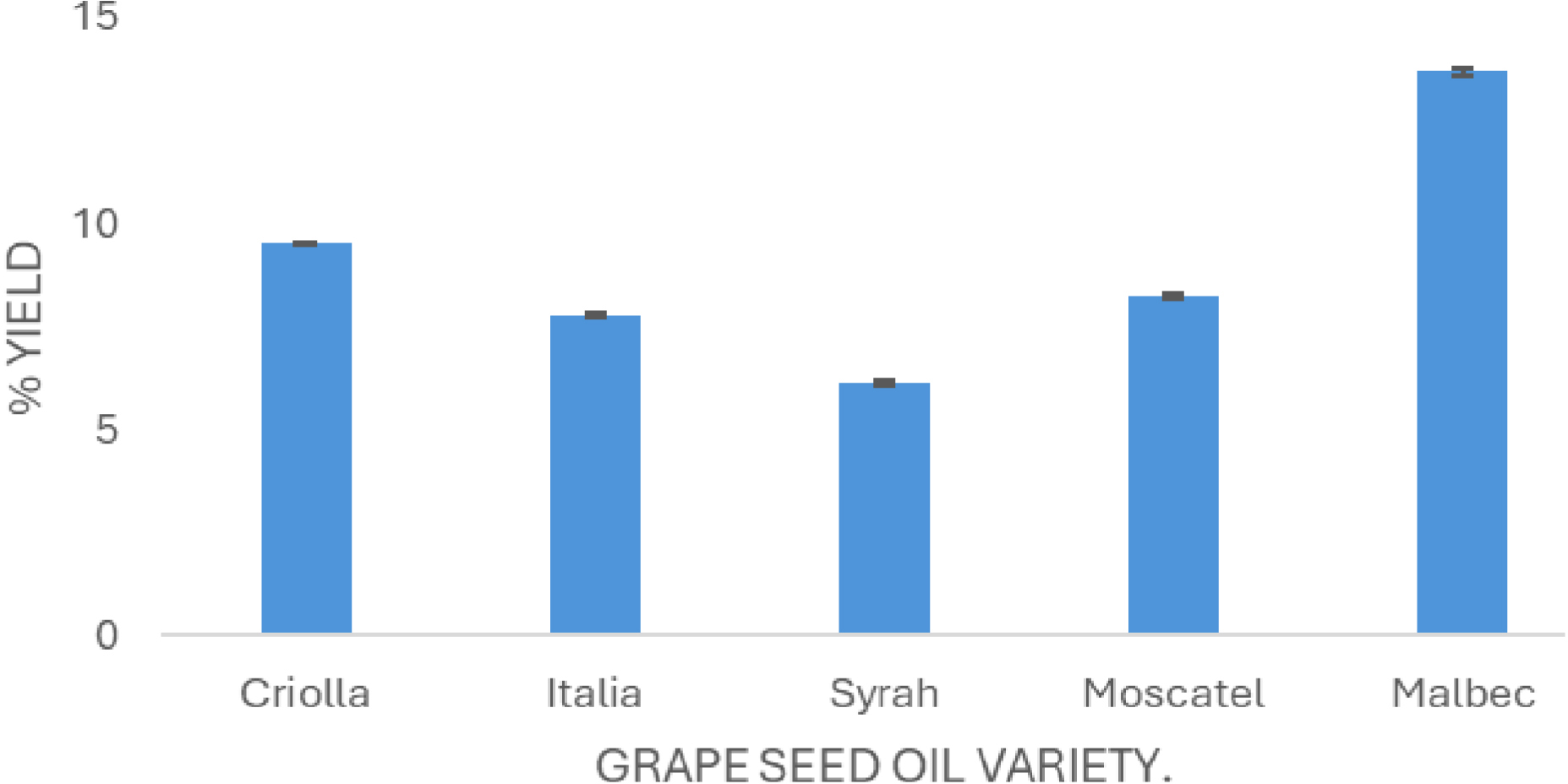
3.2. Fourier-transform infrared spectrophotometry (FTIR)
Fourier-transform infrared spectroscopy (FTIR) was used to obtain information about the functional groups present in the biomass (Figure 2).
FTIR spectra of the different grape seed varieties: (a) Malbec, (b) Syrah, (c) Italia, (d) Moscatel, (e) Criolla.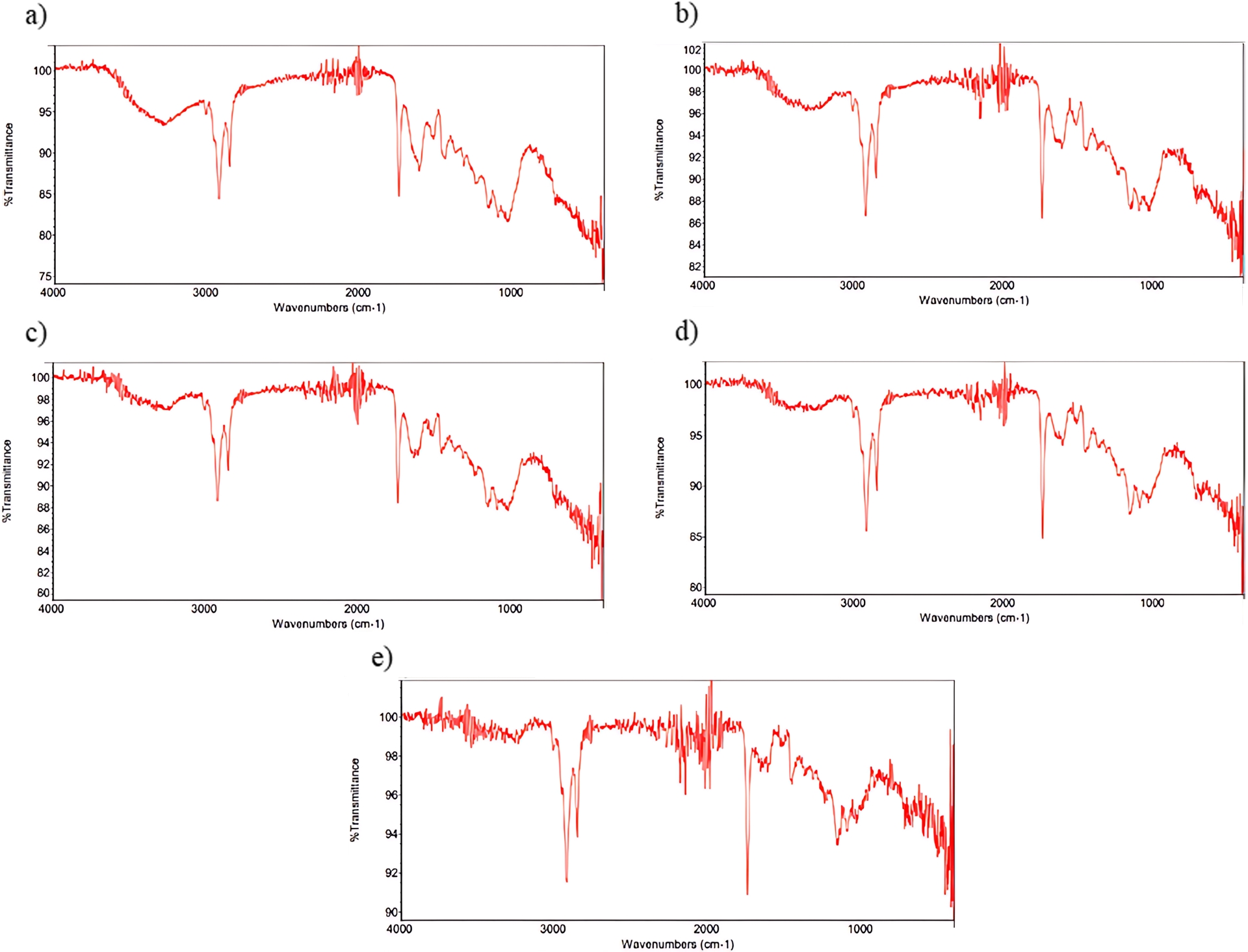
Figure 2 shows the FTIR spectra of grape seeds from the different Vitis vinifera varieties, which present very similar spectral profiles, indicating comparable chemical structures among the samples. In all analyzed varieties, a broad absorption band with a peak around 3260 cm−1 is observed, corresponding to O–H stretching vibrations present in polysaccharides and/or lignins, as previously reported [29, 30, 31]. A band around 3010 cm−1 is related to C–H stretching vibrations of cis-double bond groups (=CH), while the bands between 2953 and 2852 cm−1 are assigned to CH2 stretching vibrations associated with lipid chains or lignins [32].
The absorption band at approximately 1744 cm−1 corresponds to C=O stretching vibrations of ester functional groups, confirming the presence of fatty acids and their glycerides, as well as contributions from pectins and lignins [33, 34]. Bands observed around 1600 cm−1 are associated with aromatic C=C stretching vibrations and O–H bending modes, related to phenolic compounds and pectic substances [33, 35]. The fingerprint region between 1500 and 800 cm−1 exhibits multiple overlapping bands from complex organic constituents; however, due to its complexity, this region was not used for detailed compound differentiation.
The FTIR analysis confirms the presence of functional groups associated with lipids and phenolic compounds across all grape seed varieties, indicating their suitability as a raw material for oil extraction.
3.3. Scanning electron microscopy–energy dispersive spectroscopy (SEM–EDS) analysis
SEM micrographs of the grape seeds from the different Vitis vinifera varieties revealed a compact and irregular surface morphology with a heterogeneous and rough texture (Figure 3). Such features reflect the complex structural organization of the seed matrix. No pronounced morphological differences were observed among the analyzed varieties.
SEM micrographs and corresponding EDS spectra of grape seeds from different Vitis vinifera varieties: (a) Malbec, (b) Syrah, (c) Italia, (d) Moscatel, and (e) Criolla.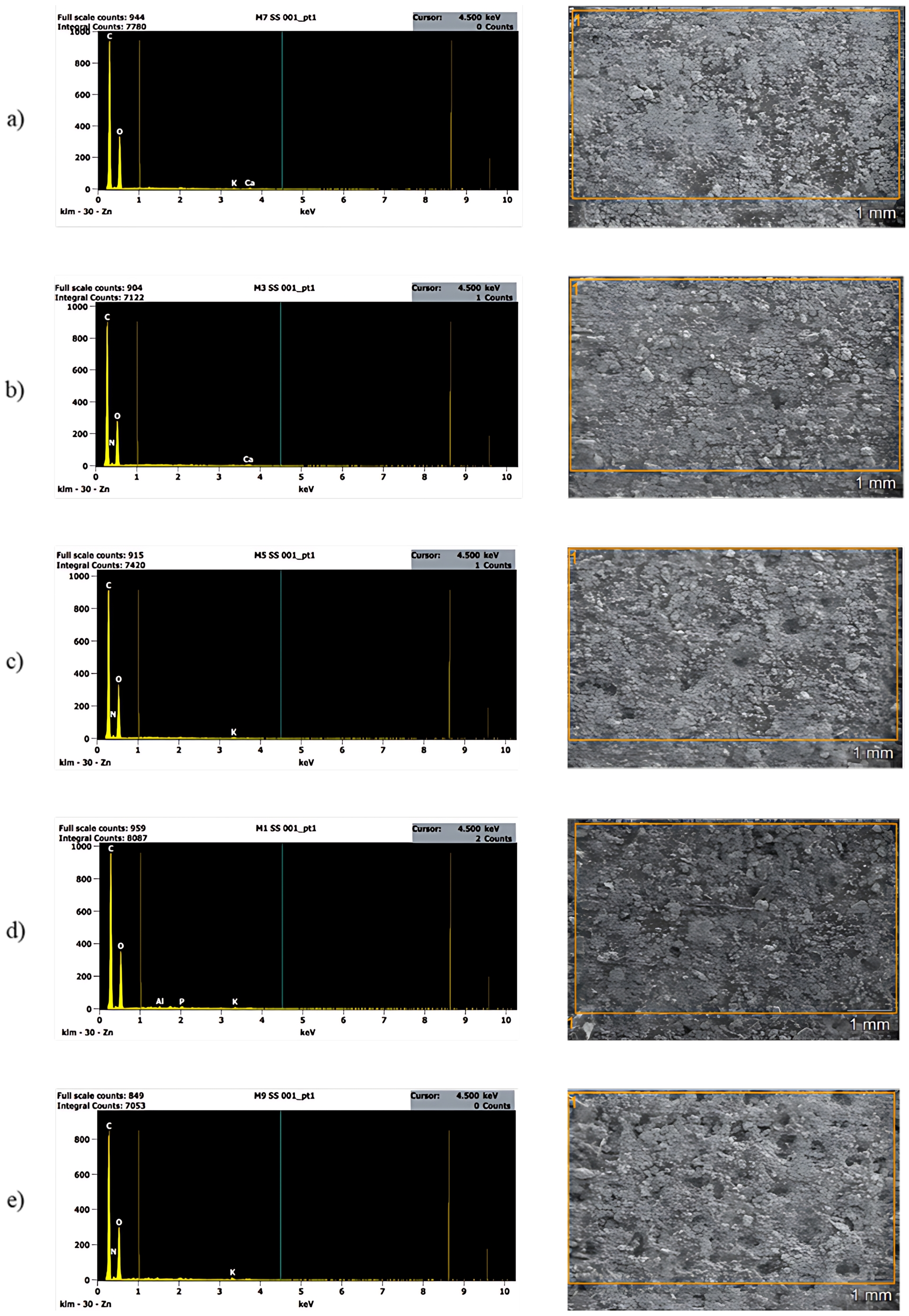
The corresponding EDS spectra indicated that carbon and oxygen were the predominant elements in all samples, confirming the organic nature of the grape seeds. Minor amounts of inorganic elements such as potassium, calcium, nitrogen, phosphorus, and aluminum were also detected, which can be attributed to naturally occurring mineral constituents of the plant material. The elemental composition determined by EDS is summarized in Table 1.
Elemental composition (wt%) of grape seeds from different Vitis vinifera varieties determined by SEM–EDS
| Weight by element (%) | |||||||
|---|---|---|---|---|---|---|---|
| Sample | C | O | K | Ca | N | Al | P |
| Malbec | 45.21 | 50.73 | 1.63 | 2.43 | - | - | - |
| Syrah | 38.10 | 46.95 | - | 2.18 | 12.77 | - | - |
| Italia | 37.51 | 47.42 | 1.62 | - | 13.45 | - | - |
| Moscatel | 47.43 | 49.04 | 1.40 | - | - | 0.74 | 1.39 |
| Criolla | 37.04 | 48.69 | 2.33 | - | 11.94 | - | - |
Note: C = carbon; O = oxygen; K = potassium; Ca = calcium; N = nitrogen; Al = aluminum; P = phosphorus.
3.4. Gas chromatography–mass spectrophotometry (GC–MS) analysis
The fatty acid composition of the pooled grape seed oil sample was analyzed by GC–MS, and the results are presented in Figure 4 and Table 2. The chromatographic analysis revealed the presence of saturated, monounsaturated, and polyunsaturated fatty acid derivatives, with a clear predominance of unsaturated compounds. The main constituents of the oil were linoleic acid ethyl ester (49.64%), ethyl oleate (10.26%), and hexadecanoic acid ethyl ester (10.00%), indicating that the oil is rich in essential and nutritionally relevant fatty acids. Compound identification was performed by comparing retention indices and mass spectra with data from the NIST mass spectral library, using similarity index values between 85 and 100, which indicates reliable identification of the detected components.
Chromatogram obtained for the pooled sample of grape oils from mixed seeds.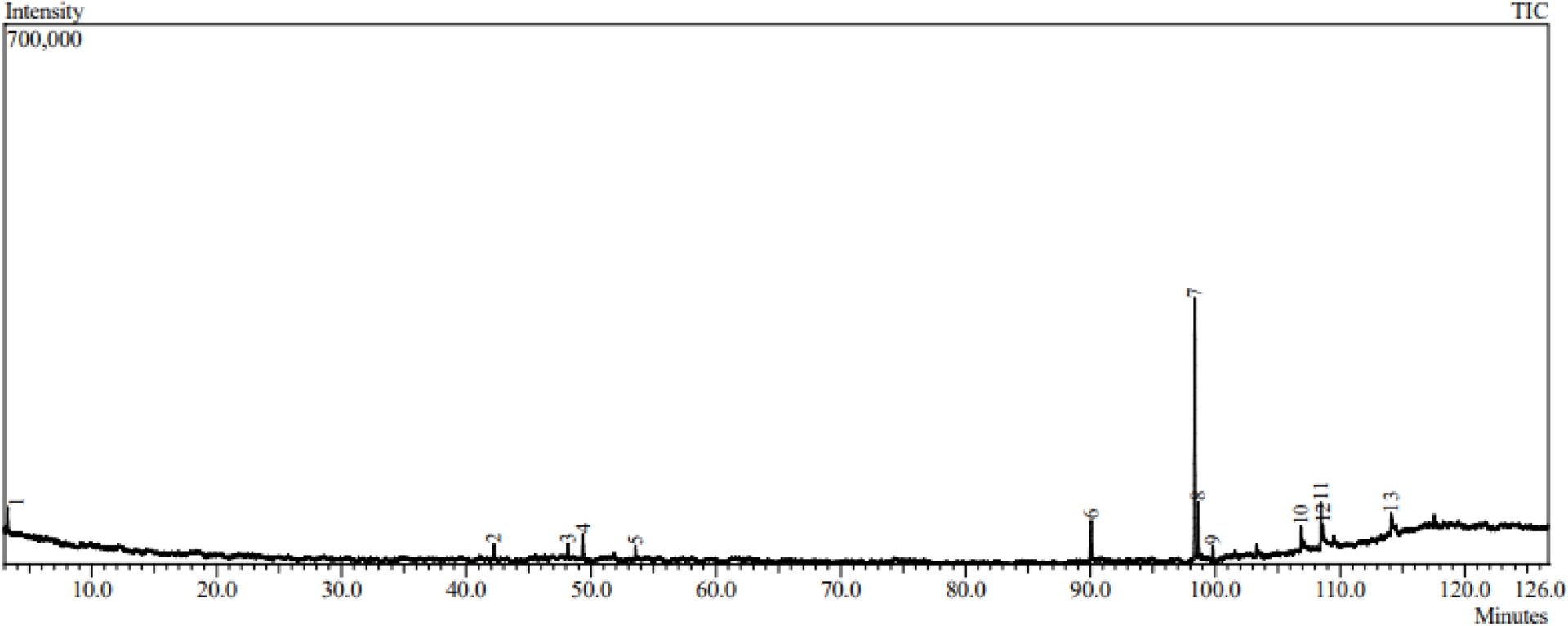
Chemical composition of the pooled sample of grape oils from mixed seeds
| Peak # | Name | Retention time (min) | Identificationa | Similarity index (SI) | Area | Area (%) | Height | Height (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | Hexanal | 3.266 | RI, MS | 92 | 56 209 | 1.76 | 26 138 | 3.95 |
| 2 | Octanoic acid, ethyl ester | 42.214 | RI, MS | 93 | 77 542 | 2.43 | 16 887 | 2.55 |
| 3 | 2,4-Decadienal | 48.148 | RI, MS | 92 | 79 097 | 2.48 | 16 871 | 2.55 |
| 4 | 2,4-Decadienal, (E,E)- | 49.351 | RI, MS | 95 | 152 739 | 4.79 | 30 162 | 4.56 |
| 5 | Decanoic acid, ethyl ester | 53.540 | RI, MS | 90 | 60 984 | 1.91 | 15 584 | 2.36 |
| 6 | Hexadecanoic acid, ethyl ester | 90.057 | RI, MS | 95 | 318 703 | 10.00 | 47 114 | 7.13 |
| 7 | Linoleic acid, ethyl ester | 98.351 | RI, MS | 95 | 1582 034 | 49.64 | 311 245 | 47.08 |
| 8 | Ethyl oleate | 98.626 | RI, MS | 94 | 327 122 | 10.26 | 66 012 | 9.98 |
| 9 | Octadecanoic acid, ethyl ester | 99.793 | RI, MS | 90 | 86 303 | 2.71 | 18 174 | 2.75 |
| 10 | Pentyl linoleate | 106.860 | RI, MS | 89 | 118 572 | 3.72 | 27 935 | 4.23 |
| 11 | Linolelaidic acid | 108.479 | RI, MS | 87 | 190 658 | 5.98 | 48 237 | 7.30 |
| 12 | 9-Octadecenoic acid, 1,2,3-propanetriyl ester, (E,E,E)- | 108.636 | RI, MS | 86 | 58 715 | 1.84 | 16 610 | 2.51 |
| 13 | Isopropyl linoleate | 114.091 | RI, MS | 87 | 78 268 | 2.46 | 20 177 | 3.05 |
| 3186 946 | 100.00 | 661 146 | 100.00 |
aMethod used for identification of compounds; RI: retention index data from literature; MS: mass spectrum.
The dominance of linoleic acid derivatives is consistent with previous reports on grape seed oil, which is known for its high content in polyunsaturated fatty acids [36]. The presence of oleic acid derivatives contributes to the overall fatty acid profile of the oil, complementing the predominance of polyunsaturated fatty acids. The identification of fatty acid ethyl esters can be attributed to the extraction and analytical conditions in GC–MS analyses of vegetable oils [37].
3.5. Microbiological analysis
No microbial growth was detected in any of the grape seed oil samples obtained from the five analyzed varieties after 24 h of incubation. Similarly, the pooled oil sample showed no detectable growth of colonies. These results indicate that the grape seed oils obtained by cold pressing met acceptable microbiological quality under the evaluated conditions. Grape seed oil has been shown to exhibit antimicrobial activity, which supports the relevance of including microbiological evaluation in this study [38].
3.6. Analysis of total phenolic compounds (TPC) and antioxidant activity
The results of TPC and antioxidant activity of the five varieties of grape seeds are shown in Table 3. The results are in a range of 112–144 mg GAE/kg of oil, with the highest amount in the Syrah variety and the lowest in the Criolla variety. These results are higher than those found by Rombaut et al. [39] for type 2 and 3 grape seeds with maximum values of approximately 90 mg GAE/kg. However, for type 1 grape (121 mg GAE/kg of oil), it is within the range found in this study. In Brazil, four varieties of oils obtained in the local market presented a maximum value of 28 mg GAE/100 g, a value lower than the samples of this study obtained by cold compression [40]. On the other hand, Kiralan et al. obtained a TPC value of 554 mg GAE/kg, which is higher than the values found in this study. These differences can be explained by factors such as the different varieties of seeds used, place and season of collection, the extraction conditions (temperature, time, speed of rotation of the equipment), oil conservation (temperature and conservation container used), and finally the method of extraction of the polyphenols from the sample because, being hydrophilic in nature, they are not very soluble in oil [41].
TPC and antioxidant activity of grape seed oils from different varieties
| Sample | TPC (mg GAE/kg of oil) | DPPH radical scavenging (%) |
|---|---|---|
| Syrah | 143.84 | 45.71 |
| Italia | 118.02 | 34.10 |
| Moscatel | 115.75 | 32.82 |
| Criolla | 112.35 | 36.19 |
| Malbec | 127.18 | 39.92 |
Likewise, the antioxidant capacity of the samples varies from 33% (Moscatel) to 46% (Syrah). Konuskan et al. reported lower antioxidant capacity values of 7–18% for six grape seed oil samples from different varieties obtained by cold pressing [8]. Meanwhile, other studies presents antioxidant capacity results of 19%–30% for the samples obtained by solvent extraction [42]. The values of the samples in this study are in this range, even though the extraction method is different. Therefore, it is established that the extraction method, whether by solvent or by cold compression, influences the antioxidant activity of grape seed oil. Akca and Akpinar found approximately 29% antioxidant capacity in a commercially available grape oil sample, showing that the one obtained by cold compression in our study has higher values [43].
3.7. Preliminary cost analysis
Based on the considered parameters, the estimated total production cost of grape seed oil obtained by cold pressing was 25.58 USD per liter. This cost includes labor associated with sample handling and oil extraction, energy consumption required for the operation of the cold-pressing equipment, and laboratory-related costs such as equipment depreciation. Raw material costs were not included, as grape seeds were considered an agro-industrial residue from the wine industry. The reported value reflects laboratory-scale conditions and provides an initial indication of the economic feasibility of valorizing grape seed residues. Although production costs may vary under industrial-scale processing due to differences in scale, efficiency, and operational conditions, the result obtained suggests that cold pressing represents a potentially viable and environmentally friendly approach for grape seed oil production.
Beyond oil extraction, the solid residue remaining after cold pressing (grape seed cake) represents an additional opportunity for valorization within a circular economy framework. Oilseed cakes have been widely recognized as promising sources of value-added food components, including dietary fiber, proteins, and bioactive compounds, depending on the raw material and processing conditions [44]. The integrated utilization of both grape seed oil and the residual cake could further enhance the sustainability and economic potential of winery by-products.
Although this study provides relevant insights into the valorization of grape seed residues from the wine industry, some limitations should be acknowledged. The fatty acid composition was determined using a pooled oil sample, which does not allow differentiation among individual grape seed varieties. Antioxidant activity was evaluated using the DPPH assay, which provides an initial estimation of radical scavenging capacity; complementary antioxidant assays could offer a more comprehensive evaluation. In addition, the cost analysis represents a preliminary estimation based on laboratory-scale conditions and may vary when applied to larger-scale or industrial processing.
4. Conclusions
This study demonstrates that grape seed residues from the wine industry constitute a valuable raw material for the sustainable production of cold-pressed grape seed oil. The grape seeds used in this work were obtained as by-products of local wine production in southern Peru, highlighting an opportunity to transform agro-industrial residues that are typically discarded into value-added products. The recovered oil exhibited a favorable fatty acid profile, significant levels of phenolic compounds, and notable antioxidant activity, particularly in oils obtained from the Syrah variety. The absence of microbiological growth further supports the quality and stability of the extracted oils.
In addition to the chemical and functional characterization, preliminary application tests were carried out using the obtained grape seed oil in the formulation of an exfoliating soap and a cosmetic cream. These exploratory formulations showed satisfactory performance, highlighting the suitability of grape seed oil as a functional ingredient with emollient and antioxidant properties. Such applications reinforce the potential use of this oil in cosmetic products, while its lipid composition and antioxidant capacity also support its applicability in food-related formulations.
Furthermore, the preliminary cost analysis indicates that cold pressing represents an economically viable and environmentally friendly approach for the valorization of winery residues. Overall, the integration of grape seed oil recovery into local wine production chains may contribute to waste reduction, promote circular economy strategies, and add value to agro-industrial by-products through practical and scalable applications.
Acknowledgments
The authors would like to thank Majes Tradición for providing the samples for this research study.
Declaration of interests
The authors do not work for, advise, own shares in, or receive funds from any organization that could benefit from this article, and have declared no affiliations other than their research organizations.






 CC-BY 4.0
CC-BY 4.0